Abstract
The potato steroidal glycoalkaloids (SGAs) are important components of plant resistance against pests and pathogens but can be toxic to humans at high levels. SGAs derive their toxicity from anticholinesterase activity affecting the central nervous system and the disruptive effects on cell membrane integrity affecting the digestive system and other organs. Accordingly, current safety regulations limit their content in the edible tuber to 20 mg per 100 g fresh weight. SGA composition and level are genetically determined, with unfavourable growth conditions and inappropriate postharvest management inducing the accumulation of SGAs at levels in the tubers of “safe” cultivars beyond the maximum level set by the industry. Hence, genetic alteration of potato to prevent toxic levels of SGAs in tubers is highly desirable. At the same time, maintaining high SGA levels in other plant organs will contribute to plant resistance against pathogen and pest attacks. To this end, SGA biosynthesis and degradation should be manipulated precisely to exploit tissue-specific expression rather than whole-plant suppression of SGA production, to produce potato cultivars with SGA content enriched in the foliage but diminished in the edible tubers. Only a few details are known about the SGA biosynthetic pathway, its genes and intermediates. Research on factors that regulate SGA biosynthesis and catabolism as well as searches for genetic markers linked to total and specific SGA levels have only recently been pursued. The present review summarizes current data on these issues to encourage further discussion on SGA manipulation for safer food products.



Similar content being viewed by others
Abbreviations
- FPP:
-
2-trans,6-trans-Farnesyl diphosphate
- HMGR:
-
3-hydroxy-3-methylglutaryl coenzyme A reductase
- SGA:
-
steroidal glycoalkaloid
- SS:
-
squalene synthase
References
Abreu P, Relva A, Matthew S, Gomes Z, Morais Z (2007) High-performance liquid chromatographic determination of glycoalkaloids in potatoes from conventional, integrated, and organic crop systems. Food Control 18:40–44
Arnqvist L, Dutta PC, Jonsson L, Sitbon F (2003) Reduction of cholesterol and glycoalkaloid levels in transgenic potato plants by overexpression of a type 1 sterol methyltransferase cDNA. Plant Physiol 131:1792–1799
Austin S, Lojkowska E, Ehlenfeldt MK, Kelman A, Helgeson JP (1988) Fertile interspecific somatic hybrids of Solanum: a novel source of resistance to Erwinia soft rot. Phytopathology 78:1216–1220
Bejarano L, Mignolet E, Devaux A, Espinola N, Carrasco E, Larondelle Y (2000) Glycoalkaloids in potato tubers: the effect of variety and drought stress on the alpha-solanine and alpha-chaconine contents of potatoes. J Sci Food Agric 80:2096–2100
Benveniste P (2004) Biosynthesis and accumulation of sterols. Annu Rev Plant Biol 55:429–457
Bergenstråhle A, Tillberg E, Jonsson L (1992) Regulation of glycoalkaloid accumulation in potato tuber disks. J Plant Physiol 140:269–275
Bergenstråhle A, Borga P, Jonsson MV (1996) Sterol composition and synthesis in potato tuber discs in relation to glycoalkaloid synthesis. Phytochemistry 41:155–161
Boluarte-Medina T, Fogelman E, Chani E, Miller AR, Levin I, Levy D, Veilleux RE (2002) Identification of molecular markers associated with leptine in reciprocal backcross families of diploid potato. Theor Appl Genet 105:1010–1018
Bouarab K, Melton R, Peart J, Baulcombe D, Osbourn A (2002) A saponin-detoxifying enzyme mediates suppression of plant defences. Nature 418:889–892
Bushway AA, Bushway RJ, Kim CH (1988) Isolation, partial purification and characterization of a potato peel glycoalkaloid glycosidase. Am Potato J 65:621–631
Choi D, Ward BL, Bostock RM (1992) Differential induction and suppression of potato 3-hydroxy-3-methylglutaryl coenzyme A reductase genes in response to Phytophthora infestans and to its elicitor arachidonic acid. Plant Cell 4:1333–1344
Choi D, Bostock RM, Avdiushko S, Hildebrand DF (1994) Lipid-derived signals that discriminate wound- and pathogen-responsive isoprenoid pathways in plants: methyl jasmonate and the fungal elicitor arachidonic acid induce different 3-hydroxy-3-methylglutaryl-coenzyme A reductase genes and antimicrobial isoprenoids in Solanum tuberosum L. Proc Natl Acad Sci USA 91:2329–2333
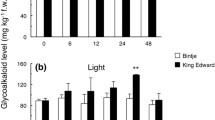
Dale MFB, Griffiths DW, Bain H, Todd D (1993) Glycoalkaloid increase in Solanum tuberosum on exposure to light. Ann Appl Biol 123:411–418
Deahl KL, Cantelo WW, Sinden SL, Sanford LL (1991) The effect of light intensity on Colorado potato beetle resistance and foliar glycoalkaloid concentration of four Solanum chacoense clones. Am Potato J 68:659–666
Deahl KL, Sinden SL, Young RJ (1993) Evaluation of wild tuber bearing Solanum accessions for foliar glycoalkaloid level and composition. Am Potato J 70:61–69
Eltayeb EA, Al-Ansari AS, Roddick JG (1997) Changes in the steroidal alkaloid solasodine during development of Solanum nigrum and Solanum incanum. Phytochemistry 46:489–494
Fewell AM, Roddick JG (1997) Potato glycoalkaloid impairment of fungal development. Mycol Res 101:597–603
Friedman M (2006) Potato glycoalkaloids and metabolites: roles in the plant and in the diet. J Agric Food Chem 54:8655–8681
Friedman M, Dao L (1992) Distribution of glycoalkaloids in potato plants and commercial potato products. J Agric Food Chem 40:419–423
Friedman M, McDonald GM (1997) Potato glycoalkaloids: chemistry, analysis, safety, and plant physiology. Crit Rev Plant Sci 16:55–132
Friedman M, Henika PR, Mackey BE (1996) Feeding of potato, tomato and eggplant alkaloids affects food consumption and body and liver weights in mice. J Nutr 126:989–999
Greenhagen BT, Schoenbeck MA, Yeo YS, Chappell J (2003) The chemical wizardry of isoprenoid metabolism in plants. In: Romeo JT (ed) Integrative phytochemistry: from ethnobotany to molecular ecology. Pergamon, Amsterdam, pp 231–251
Gregory P, Sinden SL, Tingey WM, Osman SF (1980) Glycoalkaloids of some wild potato species differing in insect resistance. Am Potato J 57:478–478
Gregory P, Sinden SL, Osman SF, Tingey WM, Chessin DA (1981) Glycoalkaloids of wild, tuber-bearing Solanum species. J Agric Food Chem 29:1212–1215
Grunenfelder LA, Knowles LO, Hiller LK, Knowles NR (2006) Glycoalkaloid development during greening of fresh market potatoes (Solanum tuberosum L.). J Agric Food Chem 54:5847–5854
Hajslova J, Schulzova V, Slanina P, Janne K, Hellenas KE, Andersson C (2005) Quality of organically and conventionally grown potatoes: four-year study of micronutrients, metals, secondary metabolites, enzymic browning and organoleptic properties. Food Addit Contam 22:514–534
Heftmann E (1983) Biogenesis of steroids in Solanaceae. Phytochemistry 22:1843–1860
Holland HL, Taylor GJ (1979) Transformations of steroids and the steroidal alkaloid, solanine, by Phytophthora infestans. Phytochemistry 18:437–440
Hutvágner G, Bánfalvi Z, Milánkovics I, Silhavy D, Polgár Z, Horváth S, Wolters P, Nap J-P (2001) Molecular markers associated with leptinine production are located on chromosome 1 in Solanum chacoense. Theor Appl Genet 102:1065–1071
Kaneko K, Watanabe M, Kawakoshi Y, Mitsuhashi H (1971) Etioline as important precursor in solanidine biosynthesis in Veratrum grandiflorum; (25S)-22,26-iminocholesta-5,22(n)-diene-3β,16α-diol. Tetrahedron Lett 12:4251–4254
Kaneko K, Tanaka MW, Mitsuhashi H (1976) Origin of nitrogen in biosynthesis of solanidine by Veratrum grandiflorum. Phytochemistry 15:1391–1393
Kaneko K, Tanaka MW, Mitsuhashi H (1977) Dormantinol, a possible precursor in solanidine biosynthesis, from budding Veratrum grandiflorum. Phytochemistry 16:1247–1251
Kolesnikova MD, **ong QB, Lodeiro S, Hua L, Matsuda SPT (2006) Lanosterol biosynthesis in plants. Arch Biochem Biophys 447:87–95
Korpan YI, Nazarenko EA, Skryshevskaya IV, Martelet C, Jaffrezic-Renault N, El’skaya AV (2004) Potato glycoalkaloids: true safety or false sense of security? Trends Biotechnol 22:147–151
Kozukue N, Misoo S, Yamada T, Kamijima O, Friedman M (1999) Inheritance of morphological characters and glycoalkaloids in potatoes of somatic hybrids between dihaploid Solanum acaule and tetraploid Solanum tuberosum. J Agric Food Chem 47:4478–4483
Krits P, Fogelman E, Ginzberg I (2007) Potato steroidal glycoalkaloid levels and the expression of key isoprenoid metabolic genes. Planta 227:143–150
Lafta AM, Lorenzen JH (2000) Influence of high temperature and reduced irradiance on glycoalkaloid levels in potato leaves. J Am Soc Hortic Sci 125:563–566
Laurila J, Laakso I, Valkonen JPT, Hiltunen R, Pehu E (1996) Formation of parental-type and novel glycoalkaloids in somatic hybrids between Solanum brevidens and S. tuberosum. Plant Sci 118:145–155
Lawson DR, Veilleux RE, Miller AR (1993) Biochemistry and genetics of S. chacoense steroidal alkaloids: natural resistance factors to the Colorado potato beetle. Curr Top Bot Res 1:335–352
Lee YY, Hashimoto F, Yahara S, Nohara T, Yoshida N (1994) Studies on the solanaceous plants 29. Steroidal glycosides from Solanum dulcamara. Chem Pharm Bull 42:707–709
Maga JA (1980) Potato glycoalkaloids. Crit Rev Food Sci Nutr 12:371–404
Maga JA (1994) Glycoalkaloids in Solanaceae. Food Rev Int 10:385–418
McCue KF, Shepherd LVT, Allen PV, Maccree MM, Rockhold DR, Corsini DL, Davies HV, Belknap WR (2005) Metabolic compensation of steroidal glycoalkaloid biosynthesis in transgenic potato tubers: using reverse genetics to confirm the in vivo enzyme function of a steroidal alkaloid galactosyltransferase. Plant Sci 168:267–273
McCue KF, Allen PV, Shepherd LVT, Blake A, Whitworth J, Maccree MM, Rockhold DR, Stewart D, Davies HV, Belknap WR (2006) The primary in vivo steroidal alkaloid glucosyltransferase from potato. Phytochemistry 67:1590–1597
McCue KF, Allen PV, Shepherd LVT, Blake A, Maccree MM, Rockhold DR, Novy RG, Stewart D, Davies HV, Belknap WR (2007) Potato glycosterol rhamnosyltransferase, the terminal step in triose side-chain biosynthesis. Phytochemistry 68:327–334
McGarvey DJ, Croteau R (1995) Terpenoid metabolism. Plant Cell 7:1015–1026
Moehs CP, Allen PV, Friedman M, Belknap WR (1997) Cloning and expression of solanidine UDP-glucose glucosyltransferase from potato. Plant J 11:227–236
Morris SC, Petermann JB (1985) Genetic and environmental effects of levels of glycoalkaloids in cultivars of potato (Solanum tuberosum L.). Food Chem 18:271–282
Osman SF, Herb SF, Fitzpatrick TJ, Sinden SL (1976) Commersonine, a new glycoalkaloid from two Solanum species. Phytochemistry 15:1065–1067
Osman SF, Johns TA, Price KR (1986) Sisunine, a glycoalkaloid found in hybrids between Solanum acaule and Solanum x ajanhuiri. Phytochemistry 25:967–968
Osman S, Sinden SL, Deahl K, Moreau R (1987) The metabolism of solanidine by microsomal fractions from Solanum chacoense. Phytochemistry 26:3163
Parnell A, Bhuva VS, Bintcliffe EJB (1984) The glycoalkaloid content of potato varieties. J Natl Inst Agric Bot UK 16:535–541
Pehu E, Gibson RW, Jones MGK, Karp A (1990) Studies on the genetic basis of resistance to potato leaf roll virus, potato virus Y and potato virus X in Solanum brevidens using somatic hybrids of Solanum brevidens and Solanum tuberosum. Plant Sci 69:95–101
Percival G, Dixon G, Sword A (1994) Glycoalkaloid concentration of potato tubers following continuous illumination. J Sci Food Agric 66:139–144
Percival GC, Karim MS, Dixon GR (1998) Influence of light enhanced glycoalkaloids on resistance of potato tubers to Fusarium sulphureum and Fusarium solani var. coeruleum. Plant Pathol 47:665–670
Petersen HW, Molgaard P, Nyman U, Olsen CE (1993) Chemotaxonomy of the tuber bearing Solanum species, subsection potato (Solanaceae). Biochem Syst Ecol 21:629–644
Rayburn JR, Bantle JA, Friedman M (1994) Role of carbohydrate side-chains of potato glycoalkaloids in developmental toxicity. J Agric Food Chem 42:1511–1515
Rayburn JR, Friedman M, Bantle JA (1995) Synergistic interaction of glycoalkaloids α-chaconine and α-solanine on developmental toxicity in Xenopus embryos. Food Chem Toxicol 33:1013–1019
Reeve RM, Hautala E, Weaver ML (1969) Anatomy and compositional variation within potatoes 1. Developmental histology of the tuber. Am Potato J 46:361–373
Ripperge H, Moritz W, Schreibe K (1971) Solanum alkaloids: biosynthesis of Solanum alkaloids from cycloartenol or lanosterol. Phytochemistry 10:2699–2704
Rokka V-M, Xu Y-S, Kankila J, Kuusela A, Pulli S, Pehu E (1994) Identification of somatic hybrids of dihaploid Solanum tuberosum lines and S. brevidens by species specific RAPD patterns and assessment of disease resistance of the hybrids. Euphytica 80:207
Ronning CM, Sanford LL, Kobayashi RS, Kowalski SP (1998) Foliar leptine production in segregating F1, inter-F1, and backcross families of Solanum chacoense Bitter. Am J Potato Res 75:137–143
Ronning CM, Stommel JR, Kowalski SP, Sanford LL, Kobayashi RS, Pineada O (1999) Identification of molecular markers associated with leptine production in a population of Solanum chacoense Bitter. Theor Appl Genet 98:39–46
Ronning CM, Kowalski SP, Sanford LL, Stommel JR (2000) Geographical variation of solanidane aglycone glycoalkaloids in the wild potato species Solanum chacoense Bitter. Genet Res Crop Evol 47:359–369
Sagredo B, Lafta A, Casper H, Lorenzen J (2006) Map** of genes associated with leptine content of tetraploid potato. Theor Appl Genet 114:131–142
Sanford LL, Sinden SL (1972) Inheritance of potato glycoalkaloids. Am Potato J 49:209–217
Sanford LL, Deahl KL, Sinden SL, Ladd TL (1992) Glycoalkaloid contents in tubers from Solanum tuberosum populations selected for potato leafhopper resistance. Am Potato J 69:693–703
Sanford LL, Deahl KL, Sinden SL (1994) Glycoalkaloid content in foliage of hybrid and backcross populations from a Solanum tuberosum × S. chacoense cross. Am Potato J 71:225–235
Sanford LL, Deahl KL, Sinden SL, Kobayashi RS (1995) Glycoalkaloid content in tubers of hybrid and backcross populations from a Solanum tuberosum × S. chacoense cross. Am Potato J 72:261–271
Sanford LL, Kobayashi RS, Deahl KL, Sinden SL (1996) Segregation of leptines and other glycoalkaloids in Solanum tuberosum (4x) × S. chacoense (4x) crosses. Am Potato J 73:21–33
Sanford LL, Kobayashi RS, Deahl KL, Sinden SL (1997) Diploid and tetraploid Solanum chacoense genotypes that synthesize leptine glycoalkaloids and deter feeding by Colorado potato beetle. Am Potato J 74:15–21
Sarquis JI, Coria NA, Aguilar I, Rivera A (2000) Glycoalkaloid content in Solanum species and hybrids from a breeding program for resistance to late blight (Phytophthora infestans). Am J Potato Res 77:295–302
Schaeffer A, Bronner R, Benveniste P, Schaller H (2001) The ratio of campesterol to sitosterol that modulates growth in Arabidopsis is controlled by STEROL METHYLTRANSFERASE 2;1. Plant J 25:605–615
Schaller H (2004) New aspects of sterol biosynthesis in growth and development of higher plants. Plant Physiol Biochem 42:465–476
Schreiber K (1968) Steroid alkaloids: Solanum group. In: Manske RHF (ed) The alkaloids. Academic, New York, pp 1–192
Schuler MA, Werck-Reichhart D (2003) Functional genomics of P450s. Annu Rev Plant Biol 54:629–667
Shih M-J, Kuc J (1974) α- and β-Solamarine in Kennebec Solanum tuberosum leaves and aged tuber slices. Phytochemistry 13:997–1000
Sinden SL, Sanford LL, Osman SF (1980) Glycoalkaloids and resistance to the Colorado potato beetle in Solanum chacoense Bitter. Am Potato J 57:331–343
Sinden SL, Sanford LL, Webb RE (1984) Genetic and environmental control of potato glycoalkaloids. Am Potato J 61:141–156
Sinden SL, Sanford LL, Deahl KL (1986) Segregation of leptine glycoalkaloids in Solanum chacoense Bitter. J Agric Food Chem 34:372–377
Sinden SL, Sanford LL, Cantelo WW, Deahl KL (1988) Bioassays of segregating plants. A strategy for studying chemical defences. J Chem Ecol 14:1941–1950
Smith DB, Roddick JG, Jones JL (1996) Potato glycoalkaloids: some unanswered questions. Trends Food Sci Technol 7:126–131
Stankovic M, Stojanovic O, Kobilarov N (1990) Unsaponifiable lipids from haulm and tuber sprouts of potato (Solanum tuberosum L). Potato Res 33:459–464
Suzuki M, Muranaka T (2007) Molecular genetics of plant sterol backbone synthesis. Lipids 42:47–54
Suzuki M, **ang T, Ohyama K, Seki H, Saito K, Muranaka T, Hayashi H, Katsube Y, Kushiro T, Shibuya M, Ebizuka Y (2006) Lanosterol synthase in dicotyledonous plants. Plant Cell Physiol 47:565–571
Swain AP, Fitzpatrick TJ, Talley EA, Herb SF, Osman SF (1978) Enzymatic hydrolysis of α-chaconine and α-solanine. Phytochemistry 17:800–801
Väänänen D, Ikonen T, Rokka V-M, Kuronen P, Serimaa R, Ollilainen V (2005) Influence of incorporated wild Solanum genomes on potato properties in terms of starch nanostructure and glycoalkaloid content. J Agric Food Chem 53:5313–5325
Väänänen T, Ikonen T, Rokka V-M, Kuronen P, Serimaa R, Ollilainen V (2006) Correction. Influence of incorporated wild Solanum genomes on potato properties in terms of starch nanostructure and glycoalkaloid content. J Agric Food Chem 2005, 53, 5313. J Agric Food Chem 54:4496–4497
Valkonen JPT, Keskitalo M, Vasara T, Pietilä L (1996) Potato glycoalkaloids: a burden or a blessing? Crit Rev Plant Sci 15:1–20
van Dam J, Levin I, Struik PC, Levy D (2003) Identification of epistatic interaction affecting glycoalkaloid content in tubers of tetraploid potato (Solanum tuberosum L.). Euphytica 134:353–360
van Gelder WMJ, Scheffer JJC (1991) Transmission of steroidal glycoalkaloids from Solanum vernei to the cultivated potato. Phytochemistry 30:165–168
Veilleux RE, Miller AR (1998) Hybrid breakdown in the F1, between Solanum chacoense and S. phureja and gene transfer for leptine biosynthesis. J Am Society Hortic Sci 123:854–858
Weltring KM, Wessels J, Geyer R (1997) Metabolism of the potato saponins α-chaconine and α-solanine by Gibberella pulicaris. Phytochemistry 46:1005–1009
Winkel BSJ (2004) Metabolic channeling in plants. Annu Rev Plant Biol 55:85–107
Wszelaki AL, Delwiche JF, Walker SD, Liggett RE, Scheerens JC, Kleinhenz MD (2005) Sensory quality and mineral and glycoalkaloid concentrations in organically and conventionally grown redskin potatoes (Solanum tuberosum). J Sci Food Agric 85:720–726
Yencho GC, Kowalski SP, Kobayashi RS, Sinden SL, Bonierbale MW, Deahl KL (1998) QTL map** of foliar glycoalkaloid aglycones in Solanum tuberosum × S. berthaultii potato progenies: quantitative variation and plant secondary metabolism. Theor Appl Genet 97:563–574
Yoshioka H, Yamada N, Doke N (1999) cDNA cloning of sesquiterpene cyclase and squalene synthase, and expression of the genes in potato tuber infected with Phytophthora infestans. Plant Cell Physiol 40:993–998
Zook MN, Kuc JA (1991) Induction of sesquiterpene cyclase and suppression of squalene synthetase activity in elicitor treated or fungal infected potato tuber tissue. Physiol Mol Plant Pathol 39:377–390
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ginzberg, I., Tokuhisa, J.G. & Veilleux, R.E. Potato Steroidal Glycoalkaloids: Biosynthesis and Genetic Manipulation. Potato Res. 52, 1–15 (2009). https://doi.org/10.1007/s11540-008-9103-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11540-008-9103-4