Search
Search Results
-
Stability
After manufacturing or preparation and during use, medicines are subject to changes. Examples are a decline of the content, formation of degradation...
-
Co-Amorphous System (Paracetamol:Indomethacin): Investigations on Physical Stability and Intermolecular Interactions
To increase the performance of indomethacin (IND) by enhancing its water solubility and promoting its therapeutic efficiency, a ternary physically...

-
Impact of Ultrasonic Vibrations on the Stability of Bovhyaluronidase Azoximer in Solution
Changes in the quantitative content and specific activity of hyaluronidase enzyme after ultrasonic treatment of solutions of bovhyaluronidase...
-
The Stability of Azithromycin in a Reconstituted Oral Suspension Under Various In-Home Storage Conditions
The stability tests of an active compound or pharmaceutical product provide information about the degradation of the active compound or the...

-
Physical Stability of Ibuprofen Upon Co-Milling with Caffeine and Polyvinylpyrrolidone at Room Temperature
The purpose of this work is to investigate the physical stability of a ternary amorphous system (Ibuprofen, Caffeine, Polyvinylpyrrolidone (PVP K30))...

-
Effects of Surfactant and Polymer on Thermodynamic Solubility and Solution Stability of Carbamazepine–Cinnamic Acid Cocrystal
This study was aimed at evaluating the solubility and solution stability of a cocrystal form of carbamazepine with cinnamic acid in the presence of...

-
Development and Validation of a Stability-Indicating Related Substances RP-HPLC Method for Anagliptin and its Degradation Products
There is no stability-indicating specific related substances high-performance liquid chromatography (HPLC) method for anagliptin active...

-
Development and Validation of Stability Indicating RP-HPLC Method for Simultaneous Determination of Chlorthalidone and Amlodipine Besylate in Bulk and in Tablet Dosage Form
Rapid, simple, and novel isocratic reverse-phase high-performance liquid chromatography (RP-HPLC) method was developed with stability, indicating...

-
Response Surface Optimised Bioanalytical Stability Indicating LC-UV Method for the Estimation of Emtricitabine and Efavirenz with Internal Standard
This work was aimed at the development of response surface optimised bioanalytical stability indicating the LC-UV method for the estimation of drugs...

-
Determination of Paclitaxel Solubility and Stability in the Presence of Injectable Excipients
Due to poor aqueous solubility of paclitaxel, cremophor is one of the excipients used to improve solubility in Taxol while it is responsible for a...
-
A Green, Validated and Stability Indicating UV Spectrophotometric Determination And HPLC Comparison of Covid-19 Drug Favipiravir in Tablets
Favipiravir (FAV) is an antiviral drug in tablets, which has been originally intended for the treatment of influenza and determined by methods based...

-
New Stability Indicating RP-UPLC Method for Simultaneous Estimation of Telmisartan and Azelnidipine in Bulk and Combined Bilayer Film-Coated Tablet Dosage Form
An efficient, perceptive, sensitive, accurate and economical RP-UPLC method with tunable UV detector has been developed for the simultaneous...

-
Vortioxetine liposomes as a novel alternative to improve drug stability under stress conditions: toxicity studies and evaluation of antidepressant-like effect
BackgroundVortioxetine hydrobromide (VXT), a new therapeutic option in the treatment of major depressive disorder, is a poorly soluble drug, and...

-
Death-associated protein kinase 1 phosphorylates MDM2 and inhibits its protein stability and function
Breast cancer is one of the major malignancies in women, and most related deaths are due to recurrence, drug resistance, and metastasis. The...

-
A Rapid Stability Indicating HPLC Method for Determination of Quetiapine Fumarate in Tablets and Extemporaneous Formulations
Quetiapine fumarate is an atypical antipsychotic drug, which is clinically used for the treatment of depression and bipolar disorders. A stability...

-
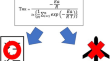
Use of Mean Kinetic Temperature for Pharmaceuticals in Japan and Stability Monitoring in the 21st Century
BackgroundThe guidelines of the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH), which was...
-
Stability of the Combined Antiallergic Gel Fludigel
Results from studies of the stability of the antiallergy gel for external application Fludigel on long-term storage in natural conditions are...

-
Stability Indicating RP-HPLC Simultaneous Estimation of Hyoscine Butylbromide and Paracetamol in Tablets
A stability indicating method was developed and validated for the quantitative simultaneous estimation of hyoscine butylbromide (HBB) and paracetamol...

-
Stability Indicating RP-HPLC-PDA Method for the Quantification of Solriamfetol in Bulk and Marketed Dosage Form
Solriamfetol is a dopamine and norepinephrine reuptake inhibitor used to treat excessive daytime sleep in patients with narcolepsy. The prime...

-
Neutral Lipids of Common Borage (Borago officinalis L.) Seeds: Stability to Oxidation During Long-Term Storage
The stability to oxidation during long-term storage of neutral lipids of common borage ( Borago officinalis L.) seeds was studied. The organoleptic...

