Abstract
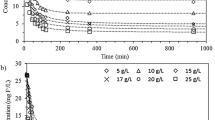
Cyanide is highly toxic and must be destroyed or removed before discharge into the environment. This study examined the ability of commercial anion-exchange resins to remove residual cyanide complexes from industrial plating wastewater as a complement to conventional treatment. Cyanide removal experiments were conducted with various initial concentrations, reaction times, and temperatures, and the presence of co-existing anions. The maximum cyanide removal capacity (Qm) of the Bonlite BAMB140 resin is 31.82 mg/g and effectively removes cyanide from aqueous solution within 30 min. The cyanide removal by the resin is an endothermic process and is affected by the presence of anions in industrial plating wastewater. The relative competitiveness observed in this study was sulfate > nitrate > chloride. A mixture of 0.05 M NaCl and NaOH regenerates resin for continuous reuse for 5 cycles. The Bonlite BAMB140 resin was able to remove residual cyanide complexes from industrial plating wastewater, but the removal capacity of the resin was reduced by more than three times in batch (9.94 mg/g) and column (6349.12 mg/L) systems. Based on the results, the anion-exchange resins are expected to be used as a complementary technique to remove residual cyanide complexes in industrial plating wastewater after conventional treatment.








Similar content being viewed by others
References
Akcil A, Mudder T (2003) Microbial destruction of cyanide wastes in gold mining: process review. Biotechnol Lett 25:445–450
Ali DS, Faizullah AT (2012) Combination of FIA-CL technique with ion-exchanger for determination of sulphate in various water resources in Erbil city. Arab J Chem 5:147–153. https://doi.org/10.1016/j.arabjc.2010.08.005
Arroyo F, Morillo J, Usero J, Rosado D, El Bakouri H (2019) Lithium recovery from desalination brines using specific ion-exchange resins. Desalination 468:114073. https://doi.org/10.1016/j.desal.2019.114073
Asci B, Kovenc E, Arar O, Arda M (2018) Kinetic, isotherm and thermodynamic investigations of nitrite (NO2-) removal from water by anion exchange resins. Glob Nest J 20:368–372
Bodalo-Santoyo A, Gómez-Carrasco J, Gomez-Gomez E, Maximo-Martin F, Hidalgo-Montesinos A (2003) Application of reverse osmosis to reduce pollutants present in industrial wastewater. Desalination 155:101–108
Cavaco SA, Fernandes S, Augusto CM, Quina MJ, Gando-Ferreira LM (2009) Evaluation of chelating ion-exchange resins for separating Cr(III) from industrial effluents. J Hazard Mater 169:516–523. https://doi.org/10.1016/j.jhazmat.2009.03.129
Chaudhary BK, Farrell J (2015) Understanding regeneration of arsenate-loaded ferric hydroxide-based adsorbents. Environ Eng Sci 32:353–360. https://doi.org/10.1089/ees.2014.0453
Chen R, Yang Q, Zhong Y, Li X, Liu Y, Li XM, du WX, Zeng GM (2014) Sorption of trace levels of bromate by macroporous strong base anion exchange resin: influencing factors, equilibrium isotherms and thermodynamic studies. Desalination 344:306–312. https://doi.org/10.1016/j.desal.2014.04.001
Dash RR, Balomajumder C, Kumar A (2009a) Removal of cyanide from water and wastewater using granular activated carbon. Chem Eng J 146:408–413. https://doi.org/10.1016/j.cej.2008.06.021
Dash RR, Gaur A, Balomajumder C (2009b) Cyanide in industrial wastewaters and its removal: a review on biotreatment. J Hazard Mater 163:1–11. https://doi.org/10.1016/j.jhazmat.2008.06.051
Dizge N, Demirbas E, Kobya M (2009) Removal of thiocyanate from aqueous solutions by ion exchange. J Hazard Mater 166:1367–1376. https://doi.org/10.1016/j.jhazmat.2008.12.049
Dutra AJ, Rocha GP, Pombo FR (2008) Copper recovery and cyanide oxidation by electrowinning from a spent copper-cyanide electroplating electrolyte. J Hazard Mater 152:648–655. https://doi.org/10.1016/j.jhazmat.2007.07.030
Elwakeel KZ (2010) Removal of Cr(VI) from alkaline aqueous solutions using chemically modified magnetic chitosan resins. Desalination 250:105–112. https://doi.org/10.1016/j.desal.2009.02.063
Fernando K, Tran T, Zwolak G (2005) The use of ion exchange resins for the treatment of cyanidation tailings. Part 2––pilot plant testing. Miner Eng 18:109–117. https://doi.org/10.1016/j.mineng.2004.05.002
Fleming C, Cromberge G (1984) The extraction of gold from cyanide solutions by strong-and weak-base anion-exchange resins. J South Afr Inst Min Metall 84:125–137
Gao Y, Deng S, Du Z, Liu K, Yu G (2017) Adsorptive removal of emerging polyfluoroalky substances F-53B and PFOS by anion-exchange resin: a comparative study. J Hazard Mater 323:550–557. https://doi.org/10.1016/j.jhazmat.2016.04.069
Guamán Guadalima MP, Nieto Monteros DA (2018) Evaluation of the rotational speed and carbon source on the biological removal of free cyanide present on gold mine wastewater, using a rotating biological contactor. J Water Process Eng 23:84–90. https://doi.org/10.1016/j.jwpe.2018.03.008
Gupta SS, Bhattacharyya KG (2011) Kinetics of adsorption of metal ions on inorganic materials: a review. Adv Colloid Interf Sci 162:39–58
Han B, Shen Z, Wickramasinghe SR (2005) Cyanide removal from industrial wastewaters using gas membranes. J Membr Sci 257:171–181. https://doi.org/10.1016/j.memsci.2004.06.064
Hutchins RA (1973) New method simplifies design of activated carbon systems. Chem Eng 80:133–138
Kepa U, Stanczyk-Mazanek E, Stepniak L (2008) The use of the advanced oxidation process in the ozone + hydrogen peroxide system for the removal of cyanide from water. Desalination 223:187–193. https://doi.org/10.1016/j.desal.2007.01.215
Kim SJ, Lim KH, Park YG, Kim JH, Cho SY (2001) Simultaneous removal and recovery of cadmium and cyanide ions in synthetic wastewater by ion exchange. Korean J Chem Eng 18:686–691
Kim S-J, Lim K-H, Joo K-H, Lee M-J, Kil S-G, Cho S-Y (2002) Removal of heavy metal-cyanide complexes by ion exchange. Korean J Chem Eng 19:1078–1084
Kim YJ, Qureshi T, Min KS (2003) Application of advanced oxidation processes for the treatement of cyanide containing effluent. Environ Technol 24:1269–1276
Lee C-G, Kim J-H, Kang J-K, Kim S-B, Park S-J, Lee S-H, Choi J-W (2014) Comparative analysis of fixed-bed sorption models using phosphate breakthrough curves in slag filter media. Desalin Water Treat 55:1795–1805. https://doi.org/10.1080/19443994.2014.930698
Lee CG, Song MK, Ryu JC, Park C, Choi JW, Lee SH (2016) Application of carbon foam for heavy metal removal from industrial plating wastewater and toxicity evaluation of the adsorbent. Chemosphere 153:1–9. https://doi.org/10.1016/j.chemosphere.2016.03.034
Lee CG, Alvarez PJ, Nam A, Park SJ, Do T, Choi US, Lee SH (2017) Arsenic (V) removal using an amine-doped acrylic ion exchange fiber: kinetic, equilibrium, and regeneration studies. J Hazard Mater 325:223–229. https://doi.org/10.1016/j.jhazmat.2016.12.003
Lee C-G, Hong S-H, Hong S-G, Choi J-W, Park S-J (2019) Production of biochar from food waste and its application for phenol removal from aqueous solution. Water Air Soil Pollut 230. https://doi.org/10.1007/s11270-019-4125-x
Li T, Guo C, **e T, Zhou C, Zhang Y (2015) Preparation of quaternary ammonium-Chlorella vulgaris and its adsorption of Ag(CN)2. Water Sci Technol 72:1437–1445. https://doi.org/10.2166/wst.2015.262
Li Q, Lu X, Shuang C, Qi C, Wang G, Li A, Song H (2019) Preferential adsorption of nitrate with different trialkylamine modified resins and their preliminary investigation for advanced treatment of municipal wastewater. Chemosphere 223:39–47. https://doi.org/10.1016/j.chemosphere.2019.02.008
Lito PF, Aniceto JPS, Silva CM (2012) Removal of anionic pollutants from waters and wastewaters and materials perspective for their selective sorption. Water Air Soil Pollut 223:6133–6155. https://doi.org/10.1007/s11270-012-1346-7
Lu X, Shao Y, Gao N, Chen J, Zhang Y, Wang Q, Lu Y (2016) Adsorption and removal of clofibric acid and diclofenac from water with MIEX resin. Chemosphere 161:400–411. https://doi.org/10.1016/j.chemosphere.2016.07.025
Mekuto L, Kim YM, Ntwampe SKO, Mewa-Ngongang M, N. Mudumbi JB, Dlangamandla N, Itoba-Tombo EF, Akinpelu EA (2018) Heterotrophic nitrification-aerobic denitrification potential of cyanide and thiocyanate degrading microbial communities under cyanogenic conditions. Environ Eng Res 24:254–262. https://doi.org/10.4491/eer.2018.147
Moussavi G, Khosravi R (2010) Removal of cyanide from wastewater by adsorption onto pistachio hull wastes: parametric experiments, kinetics and equilibrium analysis. J Hazard Mater 183:724–730. https://doi.org/10.1016/j.jhazmat.2010.07.086
Naushad M, Khan MR, ALOthman ZA, Awual MR (2016) Bromate removal from water samples using strongly basic anion exchange resin Amberlite IRA-400: kinetics, isotherms and thermodynamic studies. Desalin Water Treat 57:5781–5788
Noroozi R, Al-Musawi TJ, Kazemian H, Kalhori EM, Zarrabi M (2018) Removal of cyanide using surface-modified Linde Type-A zeolite nanoparticles as an efficient and eco-friendly material. J Water Process Eng 21:44–51. https://doi.org/10.1016/j.jwpe.2017.11.011
Osathaphan K, Boonpitak T, Laopirojana T, Sharma VK (2008) Removal of cyanide and zinc–cyanide complex by an ion-exchange process. Water Air Soil Pollut 194:179–183. https://doi.org/10.1007/s11270-008-9706-z
Parga JR, Shukla SS, Carrillo-Pedroza FR (2003) Destruction of cyanide waste solutions using chlorine dioxide, ozone and titania sol. Waste Manag 23:183–191. https://doi.org/10.1016/s0956-053x(02)00064-8
Ravuru SS, Jana A, De S (2019) Synthesis of NiAl-layered double hydroxide with nitrate intercalation: application in cyanide removal from steel industry effluent. J Hazard Mater 373:791–800. https://doi.org/10.1016/j.jhazmat.2019.03.122
Schoeman E, Bradshaw SM, Akdogan G, Snyders CA, Eksteen JJ (2017) The elution of platinum and palladium cyanide from strong base anion exchange resins. Int J Miner Process 162:19–26. https://doi.org/10.1016/j.minpro.2017.03.002
Shi T, Wang Z, Liu Y, Jia S, Changming D (2009) Removal of hexavalent chromium from aqueous solutions by D301, D314 and D354 anion-exchange resins. J Hazard Mater 161:900–906. https://doi.org/10.1016/j.jhazmat.2008.04.041
Shuang C, Pan F, Zhou Q, Li A, Li P, Yang W (2012) Magnetic polyacrylic anion exchange resin: preparation, characterization and adsorption behavior of humic acid. Ind Eng Chem Res 51:4380–4387. https://doi.org/10.1021/ie201488g
Simsek H, Kobya M, Khan E, Bezbaruah AN (2015) Removal of aqueous cyanide with strongly basic ion-exchange resin. Environ Technol 36:1612–1622. https://doi.org/10.1080/09593330.2014.999829
Song M, Li M (2019) Adsorption and regeneration characteristics of phosphorus from sludge dewatering filtrate by magnetic anion exchange resin. Environ Sci Pollut Res Int 26:34233–34247. https://doi.org/10.1007/s11356-018-4049-9
Tandorn S, Arqueropanyo O-a, Naksata W, Sooksamiti P, Chaisri I (2019) Adsorption of arsenate from aqueous solution by ferric oxide-impregnated Dowex Marathon MSA anion exchange resin: application of non-linear isotherm modeling and thermodynamic studies. Environ Earth Sci 78. https://doi.org/10.1007/s12665-019-8138-y
Teixeira LAC, Andia JPM, Yokoyama L, da Fonseca Araújo FV, Sarmiento CM (2013) Oxidation of cyanide in effluents by Caro’s acid. Miner Eng 45:81–87. https://doi.org/10.1016/j.mineng.2013.01.008
Vapur H, Bayat O (2007) Prediction of cyanide recovery from silver leaching tailings with AVR using multivariable regression analysis. Miner Eng 20:729–737. https://doi.org/10.1016/j.mineng.2007.01.012
Víctor-Ortega MD, Ochando-Pulido JM, Martínez-Férez A (2016) Phenols removal from industrial effluents through novel polymeric resins: kinetics and equilibrium studies. Sep Purif Technol 160:136–144. https://doi.org/10.1016/j.seppur.2016.01.023
Yu X, Xu R, Wei C, Wu H (2016) Removal of cyanide compounds from coking wastewater by ferrous sulfate: improvement of biodegradability. J Hazard Mater 302:468–474. https://doi.org/10.1016/j.jhazmat.2015.10.013
Zhu Y, Gao N, Wang Q, Wei X (2015) Adsorption of perchlorate from aqueous solutions by anion exchange resins: effects of resin properties and solution chemistry. Colloid Surf A-Physicochem Eng Asp 468:114–121. https://doi.org/10.1016/j.colsurfa.2014.11.062
Funding
This work was supported by the National Research Foundation (NRF) of Korea (Grant no. NRF-2018R1C1B5044937).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Angeles Blanco
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chu, JH., Kang, JK., Park, SJ. et al. Application of the anion-exchange resin as a complementary technique to remove residual cyanide complexes in industrial plating wastewater after conventional treatment. Environ Sci Pollut Res 27, 41688–41701 (2020). https://doi.org/10.1007/s11356-020-10162-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-10162-y