Abstract
Fas-associated protein with death domain (FADD), procaspase-8, and cellular FLICE-inhibitory proteins (cFLIP) assemble through death-effector domains (DEDs), directing death receptor signaling towards cell survival or apoptosis. Understanding their three-dimensional regulatory mechanism has been limited by the absence of atomic coordinates for their ternary DED complex. By employing X-ray crystallography and cryogenic electron microscopy (cryo-EM), we present the atomic coordinates of human FADD-procaspase-8-cFLIP complexes, revealing structural insights into these critical interactions. These structures illustrate how FADD and cFLIP orchestrate the assembly of caspase-8-containing complexes and offer mechanistic explanations for their role in promoting or inhibiting apoptotic and necroptotic signaling. A helical procaspase-8-cFLIP hetero-double layer in the complex appears to promote limited caspase-8 activation for cell survival. Our structure-guided mutagenesis supports the role of the triple-FADD complex in caspase-8 activation and in regulating receptor-interacting protein kinase 1 (RIPK1). These results propose a unified mechanism for DED assembly and procaspase-8 activation in the regulation of apoptotic and necroptotic signaling across various cellular pathways involved in development, innate immunity, and disease.
Similar content being viewed by others
Introduction
The fundamental role of death receptor (DR) signaling pathways lies in making vital life-or-death decisions that are crucial for proper embryogenesis, tissue development, immune responses, and the maintenance of tissue homeostasis1,2. The formation of various signaling complexes through homotypic death domain (DD)-fold assembly is a pivotal process. Upon activation by death ligands (DL), receptors like Fas (CD95) and tumor necrosis factor-related apoptosis-inducing ligand receptors (TRAILR, including DR4 and DR5) recruit an adapter protein, Fas-associated death domain (FADD) via DD-DD interactions. In contrast, receptor-like tumor necrosis factor receptor 1 (TNFR1) recruits TNFR-associated death domain (TRADD). These DR initiate distinct signaling cascades by forming different signaling complexes3,4. Nevertheless, the precise mechanical underpinnings of this process remain unclear.
The CD95/TRAILR-FADD receptor complex assembles the death-inducing signaling complex (DISC), or complex I, by homotypic death-effector domain (DED) assembly with procaspase-8 (Casp-8) and antiapoptotic cellular FLICE-inhibitory proteins (cFLIP)5,6. This complex is subsequently dismantled into cytosolic secondary complexes, known as complex II7. Both complex I and II can activate caspases, driving apoptosis7,8. In DL-sensitive cells, the levels of cFLIP are often insufficient to block DR-induced apoptosis9,10,11.
The signaling complex formation within the TNFR1 pathway is more intricate. DD-mediated formation of TNFR1 complex I initiates receptor-interacting protein kinase 1 (RIPK1)-dependent NF-κB activation, leading to the expression of antiapoptotic proteins such as cFLIP for cell survival12,13,14,38,39,51,75,76,77,78. Furthermore, future investigations are necessary to investigate whether cFLIP could effectively impede full Casp-8 activation if cFLIP doesn’t target FADD and the first CBS on the FADD-Casp-8 intermediate complex.
In addition to our investigations into multiprotein FADD-Casp-8-cFLIP DED complexes, it is noteworthy to mention recently reported domain-swapped dimer formations in Casp-8 tDED under specific conditions, involving the F122A mutation or a two-residue truncation possibly at the C-terminus of DED2’s H651,77. The details of Casp-8 tDED domain-swap** merit consideration in the broader context of DED assembly dynamics, a topic that may be explored in future studies. In a broader context of DD-fold multiprotein assembly, we found the diverse use of various CSS in self-assembly in other DD-fold multiprotein structures. For example, the RIG-I tandem CARD assembly79 utilizes a type III-I-III CSS (comprising one type I and two type III surfaces) to self-assemble a tetramer (Supplementary Fig. 14d). In the human apoptosome79, the heterodimeric Apaf-1-Casp-9 CARD protomer employs a type III-II-III CSS to self-assemble a trimer (Supplementary Fig. 14e). In our study, the interaction of two single FADDDED molecules leads to a homodimer that creates a type-III-II-III CSS surface to bind Casp-8tDED, supporting our earlier hypothesis80.
The structural insights provided in this study, elucidating previously undocumented interactions such as FADDDED-FADDDED, FADDDED-Casp-8tDED, FADDDED-cFLIPtDED, Casp-8tDED-cFLIPtDED, and cFLIPtDED-cFLIPtDED within the ternary complex at a 3.7 Å resolution, mark a pivotal advancement in our comprehension of the intricate multiprotein DED/tDED assembly in 3D. By unveiling crucial determinants of specificity in assembling a complex with pseudo-helical symmetry, these structural data propel our insights to a higher dimension. The revelation underscores the imperative for further research to uncover the functional implications embedded in these intricate structural findings, especially in different types of cells with variations in receptor levels, signaling molecules, regulatory mechanisms, and unique genetic defects.
Methods
Protein expression and purification
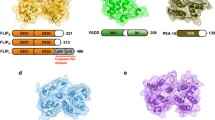
The FADD protein fused with a C-terminal 6xHis-tag (C-tag), Casp-8tDED mutant containing the residues 1–185 of Casp-8, and cFLIPtDED protein containing the residues 1–181 of cFLIP, were individually expressed in Escherichia coli (BL21-CodonPlus, Agilent). Ternary complexes were co-purified by using Ni-nitrilotriacetic acid affinity resin followed by size-exclusion chromatography (SEC) using a Superdex-200 (10/300) gel-filtration column (Cytiva) equilibrated with Buffer A (80 mM NaCl and 20 mM Tris, pH 8.0). The selenomethionine (SeMet, Carbosynth FS09881)-substituted protein complex was expressed and co-purified by the same method. The cloning primers are listed in Supplementary Table 4.
Protein crystallization
The protein complex was crystallized at a protein concentration of 10 mg/mL by the hanging-drop vapor-diffusion method at 20 °C with a solution of mixing 100 mM Na-HEPES at pH 7.0, 100 mM TBG at pH 9.0, 8% PEG8000, and 10 mM TCEP at pH 7.3. The SeMet-substituted protein complex was crystallized by the same hanging-drop vapor-diffusion method. The SeMet-substituted protein crystals was crystallized by using a solution containing 92 mM Na-HEPES at pH 7.0, 169 mM TBG at pH 9.0, 8% PEG8000, and 9 mM TCEP at pH 7.3.
Crystal structure determination and model building
Anomalous and native diffraction data was collected at the beamline TLS BL13B1 at NSRRC, using the wavelengths of 0.97939 and 0.99984 Å, respectively, at 110 K. The data sets were processed using the program HKL-2000. The structure of the FAFuL_H9G-C8FGLG-CFH7G complex was determined by using a SeMet-derivative dataset and the MR-SAD method in Phenix ver 1.14-3260. There was one 1:5:4 FAFuL_H9G-C8tDED_FGLG-CFtDED_H7G complex per asymmetric unit with a solvent content of 65%. The structural model was iteratively built and refined using the programs Coot ver 0.8.9.1 EL and Phenix ver 1.14-3260, respectively. There is no electron density for FADD DD. All residues belong to the Ramachandran favored and allowed regions with no outliers in both structures. Data collection and refinement statistics were summarized in Supplementary Table 1.
Structural analysis and molecular graphics
The structures were analyzed using the MUSTANG ver 3.2.3, MolProbity (http://kinemage.biochem.duke.edu), Coot ver 0.8.9.1 EL or WinCoot ver 0.8.2, PyMOL ver 1.8.2.1 and UCSF Chimera programs ver 1.13.1, and the qtPISA program in CCP4i2 ver 7.0.060. All interface residues and areas were identified and estimated, respectively, by qtPISA. All figures for presenting the structures were prepared by using the PyMOL ver 1.8.2.1 and UCSF Chimera program ver 1.13.1.
The ball-and-ribbon diagram
In Figs. 2b and d, the ball-and-ribbon diagrams show the spatial relationship of individual DEDs of the ternary complexes by using the PyMOL program ver 1.8.2.1 to draw ribbons that connect the spatially conserved Cα atoms (shown as balls) of every two adjacent DEDs. The ribbons show that DEDs assemble via the type I, II, and also III interfaces: blue, green, and cyan ribbons represent the DED assembly through the type I interface (called the type I connectivity), while gray ribbons represent the DED assembly through the type II interface (the type II connectivity). Red, yellow, and pink ribbons represent the DED assembly through the type III interface (the type III connectivity). The balls are the Cα atoms of the spatially conserved leucine residues among DEDs including FADD residue L63, Casp-8 residues L62 and L162, and cFLIP residues L55 and L152 (Supplementary Fig. 1c). In addition, the spatially conserved Cα atoms were also used to calculate the helical twist shown in Fig. 5c by the HELANAL-Plus program (https://dna.mbu.iisc.ac.in/resources.htm).
Multi-angle light scattering
In the SEC-MALS experiments, ternary DED complexes were injected onto a Superdex 200 (5/150) size-exclusion column and a Wyatt protein SEC column with the pore size of 300 Å, respectively. The SEC column was equilibrated with 20 mM Tris (pH 8.0) and 80 mM NaCl at room temperature, through a fast protein liquid chromatography system (Äkta purifier 10) coupled with a three-angle light-scattering detector (mini-DAWN TREOS) and a refractive index detector (Optilab T-rEX) (Wyatt Technology). Data analysis was carried out using the program ASTRA ver 6.0.5.3.
Small-angle X-ray scattering
In the SEC-SAXS experiments, the SAXS data was collected at the SWAXS beamline BL23A1 at NSRRC with an online size-exclusion high-performance liquid chromatographic system (Agilent chromatographic system 1260 series). Each protein sample was injected into an Agilent Bio SEC-5 HPLC column with a flow rate of 0.35 ml/min. Once the complex is eluted, the solution will go through a quartz capillary (2.0 mm diameter) with a flow rate decreased to 0.03 ml/min for the SAXS data collected with 30 s per frame using a Pilatus 1M-F area detector. All buffer solutions were measured under the same condition for background scattering subtraction. The SAXS data was analyzed by the ATSAS program suite ver 2.7 to 3.2.1. Data collection and structural parameters were summarized in Supplementary Table 2.
Treatment of protein precipitates for EM studies
The precipitates of overexpressed wild-type protein or complexes were resuspended in water for negative stain EM studies.
Negative staining
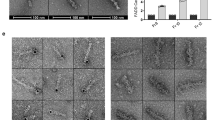
In all, 4 μl sample solution was applied onto the carbon-coated side of a TEM carbon type-B grid (Ted Pella, 01814-F) for 30 seconds and then the liquid was drawn off from the edge of grid with a filter paper (Whatman). Immediately, 4 μl of 1% uranyl acetate or uranyl formate was applied to the grid for 30 seconds and drawn off with a filter paper. Then the grid was placed into an electronic dry cabinet to air dry before imaging, which uses a FEI Tecnai G2 F20 S-TWIN at ICOB, Academia Sinica. To detect His-tagged targets, 5 nm Ni-NTA Nanogold was used according to Supplier’s instruction (Nanoprobes).
Protein sample cross-linking
We utilized the gradient-based cross-linking method, also called the GraFix method, for protein sample cross-linking. Before cross-linking, purified protein complexes were dialyzed against a HEPES buffer and then concentrated to 1 mg/ml. For creating gradients of 10–30% glycerol with 0.016-0.05% glutaraldehyde, a centrifuge tube (14 × 89 mm, Beckman Coulter) containing a top solution (80 mM NaCl, 20 mM HEPES pH 8, 10% glycerol) and a bottom solution (80 mM NaCl, 20 mM HEPES pH 8, 30% glycerol, 0.05% glutaraldehyde) was placed into a gradient mixer (the Gradient Master 108, BioComp Instrument). Subsequently, a 200-μl buffering cushion (80 mM NaCl, 20 mM HEPES pH 8, 7% glycerol) was added on the top of the gradient, followed by loading a 200-μl protein sample on the top of the cushion. The ultracentrifugation was carried out at 4 °C in a swing-out rotor (SW-41, Beckmann) at a speed of 273,620 × g or 40,000 rpm for 18.5 h. The gradient was then fractionated from the bottom and each fraction was analyzed by SDS-PAGE (Bio-Rad). The peak fractions were further loaded onto a prewashed desalting column (ZebaTM Spin Desalting Columns, Thermo Scientific) to remove glycerol and excess cross linkers. The cross-linked complex was eluted in a TRIS buffer (80 mM NaCl, 20 mM Tris pH 8) and stored at 4 °C.
Cryo-EM grids preparation and data acquisition
The glutaraldehyde cross-linked protein samples (~250 μg/ml) (Complex B) were applied to glow-discharged holey carbon grids (Quantifoil Cu, R1.2/1.3, 300 mesh). Each grid was blotted for 3 ~ 4 s and flash-plunged into liquid ethane pre-cooled in liquid nitrogen using an FEI Vitrobot mark IV operated at 4 °C and 100% humidity. Cryo-EM data were acquired on a Titan Krios (FEI) operated at 300 kV, equipped with a Gatan K2 Summit detector at ASCEM, Academia Sinica. The EPU software was utilized for automated data collection. Movies were collected at a nominal magnification of 165,000× in counting mode resulting in a calibrated pixel size of 0.84 Å/pixel, with a defocus range of approximately –0.5 to –2.5 μm. Eighty frames were recorded over 6 s of exposure at a dose rate of 0.9 electrons per Å2 per frame. Data collection, refinement and validation statistics were summarized in Supplementary Table 3.
The non-crosslinked protein sample (Complex A) was absorbed onto freshly glow-discharged Quantifoil 2/1 grids with PELCO easiGlow glow discharger and flash frozen in liquid ethane using Leica GP Climate controlled sample plunger with a controlled temperature and humidity. Cryo-EM movies were collected with FEI Talos Arctica electron microscope equipped with a Gatan BioQuatum energy filter and a K2 Summit direct electron detector at the Institute for Quantitative Biomedicine at Rutgers University, USA using the software EPU for automated data collection. We collected 30 movie frames with a combined exposure time of 7.5 s corresponding to a total dose of 24 electrons per Å2 (0.8 electrons/frame). Defocus values were set from −0.5 to −3 μm at a magnification of 130,000 times resulting with a pixel size of 1 Å.
Cryo-EM image processing and model refinement
All datasets were processed using the CryoSparc V2 software package installed in our own computers. Movie frames were aligned by running the job Full-frame motion correction. The contrast transfer function parameters for each aligned micrograph were estimated by running CTF estimation (CTFFIND4). Particle templates were created from the particles picked manually from 15 to 20 micrographs. Particles were then picked automatically by running Template picker. All particles were local-aligned and extracted by running Local motion correction, and then subjected to 2D classification for several cycles to remove junk particles. Ab initio 3D classification was carried out by running Ab-initio Reconstruction. After removing junk particles, the jobs the heterogeneous refinement, homogeneous refinement, and non-uniform refinement were performed. All maps were sharpened and the resolution was estimated using the Fourier shell correlation (FSC) = 0.143 cutoff criterion (Supplementary Figs. 5 and 6).
The crystal structures of FADDDED, cFLIPtDED, and C8tDED, were fit into the Cryo-EM envelops using UCSF Chimera ver 1.13.1 and refined in real space using the Phenix ver 1.14-3260 software. Identification of Casp-8 and cFLIP is facilitated by the presence of envelops for helix H7 of tDED, with further confirmation of cFLIP identity through the observation of disordered helix H3 in both cFLIP DED1 and DED2 (Supplementary Figs. 1c and 9). Notably, given that tDED but not FADD DED has helix H7 (Supplementary Fig. 1a and Supplementary Figs. 8, 9, vs 7), the absence of an envelope for helix H7 of tDED distinctly indicates that in Complex B, the position typically occupied by the tDED of C8d is instead taken up by two FADD DED molecules (Supplementary Fig. 2e). The Casp-8 protease domain and FADD DD were not visible.
Mutagenesis and pulldown assays
All gene mutations were generated by using the KOD Plus Mutagenesis Kit (TOYOBO) or In-Fusion HD Cloning Plus kit (TaKaRa). The His-tagged proteins were expressed and captured by the resin (Qiagen). Excess proteins and impurities were removed by three times of wash with 40 mM imidazole. The resin was then mixed with non-tagged protein lysates at room temperature for 1 h, and then washed again with three times of 40 mM imidazole. The proteins were eluted with 1 M imidazole. All samples are subjected to SDS-PAGE analysis with Coomassie blue staining. The mutagenesis primers are listed in Supplementary Table 4.
Cell culture
Immortalized mouse embryonic fibroblast (iMEF)81 and HEK293T cells (American Type Culture Collection (ATCC) CRL-3216, RRID:CVCL_0063) were cultured in Dulbecco’s Modified Eagle’s Medium (DMEM) high glucose supplied with 10% FBS, Sodium pyruvate and penicillin/streptomycin. HAP1 cells (human chronic myeloid leukemia CML-derived HAP1 cells) were purchased from Horizon Discovery Group plc (RRID:CVCL_SM74) and cultured Iscove’s Modified Dulbecco’s Medium (IMDM) with 10% FBS and penicillin/streptomycin (Gibico) according to Supplier’s instruction (Horizon Discovery). These cell lines were not authenticated by ourself in this study. No commonly misidentified cell lines were used in this study.
Antibodies used in the cell-based experiments
phospho-IKKα/β (Ser176/180)(Cell Signaling, #2697 L, 1:1000); IKKα/IKKβ (Santa Cruz, #sc-7607,1:1000); GAPDH (GeneTex, #GTX627408, 1:5000); cFLIP (dave-2) (Adipogen, #AG-20B-0005, 1:1000); Casp-8 (D35G2) (Cell Signaling, #4790, 1:1000); Cleaved Casp-3 (Asp175) (5A1E) (Cell Signaling, #9664, 1:1000); PARP (Cell Signaling, 930 #9532,1:1000); phospho-MLKL (S345) (Abcam, #ab196436, 1:2000); MLKL (clone 3H1) (Millipore, #MABC604, 1:1000); phospho-RIPK3 (Thr231/Ser232) (Cell Signaling, #57220, 1:1000); RIPK3 (Cell Signaling, #95702, 1:1000); RIPK1 (BD, #610459, 1:2000); Phospho-RIPK1 (Ser166) (Cell Signaling, #31122, 1:1000); phospho-p38 (Thr180/Tyr182) (Cell Signaling, #9211,1:1000); p38 MAPK (Cell Signaling, #9212, 1:1000); phospho-JNK/SAPK (Thr183/Tyr185) (Cell Signaling, #9251, 1:1000); JNK/SAPK (Cell Signaling, #9252, 1:1000); FADD (Abcam, #Ab124812, 1:1000); FADD (human specific) (Cell Signaling, #2782, 1:1000).
Reagents used in the cell-based experiments
Recombinant mouse TNFα (R&D Systems, #410-MT), Recombinant human TNFα (R&D Systems, #210-TA), SuperKillerTRAIL (Adipogen, #AG-40T-0004-C020), SM-406 (ApexBio, #A3019), z-VAD-fmk (ApexBio, #A1902), Nec-1s (Biovision, #2263), Doxycycline hyclate (Sigma, #D9891), Cyclohexylamine (Sigma, #C1988).
Generation of FADD knockout cells
SgRNA targeting on FADD and pAll-Cas9.pPuro plasmid were designed and purchased from the National RNAi Core Facility at Academia Sinica, Taiwan. iMEF was transfected with CRISPR plasmid using Turbofect (Thermo) according to manufacturer’s instructions. On the third day after transfection, iMEF underwent serial dilutions and antibiotic selection to remove unwanted cells. Surviving cell colonies were examined by western blotting to confirm the gene knockout (Supplementary Fig. 15a). The FADD-gene knockout colonies were expanded and stored for the following experiments. FADD-KO clone sg4.B9 was used for expansion because necroptotic signaling molecules MLKL and RIPK3 remain there (Supplementary Fig. 15a). TNF-induced NF-κB activation signaling remains normal in the FADD-KO iMEF cells (clone sg4.B9) with or without CHX (Supplementary Fig. 15g, h). The FADD-gene knockout HAP1 (RRID:CVCL_SM74) in Fig. 6g were purchased from Horizon Discovery.
FADD reconstitution
The human or mouse full-length FADD gene was cloned into the pAS4.1w.Pbsd-aOn vector. HEK293T cells (American Type Culture Collection (ATCC) CRL-3216, RRID:CVCL_0063) were transfected with FADD constructs and the packaging plasmids pMD.G and pCMVR8.91 (the National RNAi Core Facility at Academia Sinica) with Turbofect according to manufacturer’s instructions. Virus-containing mediums were harvested 48 and 72 h after transfection. FADD-gene knockout iMEF clone was incubated with the virus-containing medium overnight for infection. After infection, iMEF were selected with blasticidin (5 μg/ml) to eliminate uninfected cells. For HAP1 cells, 7.5 μg/ml blasticidin was used. Stable colonies were then saved for following assays. FADD protein expression was induced by doxycycline hyclate (0.1–1 μg/ml) for 16–24 h before additional treatments. The cloning primers are listed in Supplementary Table 4.
Cell-based mutagenesis assays
Tet-inducible plasmids containing FADD WT or mutant sequences were transfected into FADD-deficient immortalized mouse embryonic fibroblast (FADD-/- iMEF) using a lentivirus packaging system. Mouse FADD (mFADD)-reconstituted iMEF were treated with doxycycline overnight to induce FADD expression. iMEF were seeded and grew to reach 60-80% confluence before assays. To induce apoptosis, iMEF were pretreated with cycloheximide (CHX, 10 μg/ml) for 30 min, followed by the stimulation with recombinant mouse TNFα (10 ng/ml) for 6 h or the indicated time. For necroptosis induction, iMEF were pretreated with cycloheximide (10 μg/ml) and z-VAD-fmk (50 μM), before TNFα treatment. Human FADD (hFADD)-reconstituted HAP1 cells were treated with doxycycline overnight to induce FADD expression. To induce apoptosis in HAP1 cell, HAP1 cells were pretreated with cycloheximide (10 μg/ml) for 30 min, followed by the stimulation with SuperKillerTRAIL (50 ng/ml). All the mutants were created by KOD-based site-direct mutagenesis by using the primers listed in Supplementary Table 4. Uncropped data are included in a Source Data file.
Fluorescence apoptosis and Necrosis assay
iMEFs were seeded in a 96-well plate (5000 cell per well) a day before the assay. To induce apoptosis, iMEF were pretreated with cycloheximide (CHX, 10 μg/ml) for 30 min, followed by the stimulation with recombinant mouse TNFα (10 ng/ml) for 6 h or the indicated time. For necroptosis induction, iMEF were pretreated with cycloheximide (10 μg/ml) and z-VAD-fmk (50 μM), before TNFα treatment. Nucleic acid and the Casp-3 activity were detected by using SYTO green (5 μM) and DEVD-AMC (20 μM), respectively. Fluorescent signal was detected by a 96-well plate reader (Victor X, PerkinElmer) at the time indicated in the figures and the data was analyzed. Bar chars were generated by GraphPad Prism 9.4.1. Notably, this assay was modified from a previous assay82.
Cell lysate-based mutagenesis study
In vitro mutagenesis experiments to test FADD-mediated signaling were performed by a HeLa cell lysate-based protein expression system (1-Step Human Coupled IVT kit, Thermo). The full-length wild type and mutant FADD proteins were expressed by PURExpress in vitro protein synthesis kit (NEB) using pET-26 vector. The reaction mixture with expressed FADD protein was added to the reaction component of the IVT kit to a final FADD concentration of about 4 μM (measured by UV absorbance), higher than ~0.33 μM of endogenous FADD83. The mixture was incubated at 30 °C for 6 h and then was analyzed by SDS-PAGE and western blotting. All the mutants were created by KOD-based site-direct mutagenesis by using the primers listed in Supplementary Table 4. Uncropped data are included in a Source Data file.
Antibodies used in the cell lysate-based experiments
FLIP (D5J1E) (Cell Signaling, #56343, 1:2000); Casp-8(D35G2) (Cell Signaling, #4790, 1:4000); Casp-3 antibody (Cell Signaling, #9662, 1:4000); Cleaved Casp-3 (Asp175) (5A1E) (Cell Signaling, #9664, 1:2000); Human FADD (Cell Signaling, #2782, 1:2000); PARP (46D11) (Cell Signaling, #9532,1:2000); RIPK1 (Cell Signaling, #4926, 1:2000); Phospho-RIPK1 (Ser166) (D1L3S) (Cell Signaling, #65746, 1:2000); GAPDH (14C10) (HRP Conjugate) (Cell Signaling, #3683, 1:4000); Goat Anti-Rabbit IgG H&L (HRP) (Abcam, #ab6721, 1:10,000).
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The atomic coordinates and reflection files for the crystal structures generated in this study have been deposited in the Worldwide Protein Data Bank (wwPDB) under accession codes 8YD7 (The SeMet derivative of the single-FADD-Casp-8-cFLIP DED complex) and 8YD8 (Native single-FADD-Casp-8-cFLIP DED complex). The cryo-EM structures generated in this study have been deposited in the Electron Microscopy Data Bank (EMDB) under accession codes EMD-39126 (The triple-FADD-Casp-8-cFLIP DED complex B) and EMD-39127 (The triple-FADD-Casp-8-cFLIP DED complex A). The atomic coordinates for the cryo-EM structure generated in this study have been deposited in the wwPDB under accession code 8YBX (The triple-FADD-Casp-8-cFLIP DED complex B). The cryo-EM structure used in this study are available in the EMDB under accession codes EMD-11939 (Central region of the Caspase-8-FADD complex), and EMD-11941 (The ternary complex of full-length Caspase-8 with FADD and FLIPs). The atomic coordinates for the cryo-EM structure used in this study are available in the wwPDB under accession codes 5L0838 (Casp-8 tDED filaments). All other data generated in this study are provided in the Supplementary Information or a Source Data file. Alternatively, any data that support this study are also available from the corresponding authors upon request. Source data are provided with this paper.
References
Tang, D., Kang, R., Berghe, T. V., Vandenabeele, P. & Kroemer, G. The molecular machinery of regulated cell death. Cell Res. 29, 347–364 (2019).
Singh, R., Letai, A. & Sarosiek, K. Regulation of apoptosis in health and disease: the balancing act of BCL-2 family proteins. Nat. Rev. Mol. Cell Biol. 20, 175–193 (2019).
Peter, M. E. et al. The CD95 receptor: apoptosis revisited. Cell 129, 447–450 (2007).
Wilson, N. S., Dixit, V. & Ashkenazi, A. Death receptor signal transducers: nodes of coordination in immune signaling networks. Nat. Immunol. 10, 348–355 (2009).
Kischkel, F. C. et al. Cytotoxicity-dependent APO-1 (Fas/CD95)-associated proteins form a death-inducing signaling complex (DISC) with the receptor. EMBO J. 14, 5579–5588 (1995).
Irmler, M. et al. Inhibition of death receptor signals by cellular FLIP. Nature 388, 190–195 (1997).
Lavrik, I. N. et al. CD95 stimulation results in the formation of a novel death effector domain protein-containing complex. J. Biol. Chem. 283, 26401–26408 (2008).
Peter, M. E. & Krammer, P. H. The CD95(APO-1/Fas) DISC and beyond. Cell Death Differ. 10, 26–35 (2003).
Rasper, D. M. et al. Cell death attenuation by ‘Usurpin’, a mammalian DED-caspase homologue that precludes caspase-8 recruitment and activation by the CD-95 (Fas, APO-1) receptor complex. Cell Death Differ. 5, 271–288 (1998).
Scaffidi, C. et al. Differential modulation of apoptosis sensitivity in CD95 type I and type II cells. J. Biol. Chem. 274, 22532–22538 (1999).
Scaffidi, C., Schmitz, I., Krammer, P. H. & Peter, M. E. The role of c-FLIP in modulation of CD95-induced apoptosis. J. Biol. Chem. 274, 1541–1548 (1999).
Brenner, D., Blaser, H. & Mak, T. W. Regulation of tumour necrosis factor signalling: live or let die. Nat. Rev. Immunol. 15, 362–374 (2015).
Hsu, H., Huang, J., Shu, H. B., Baichwal, V. & Goeddel, D. V. TNF-dependent recruitment of the protein kinase RIP to the TNF receptor-1 signaling complex. Immunity 4, 387–396 (1996).
Shu, H. B., Takeuchi, M. & Goeddel, D. V. The tumor necrosis factor receptor 2 signal transducers TRAF2 and c-IAP1 are components of the tumor necrosis factor receptor 1 signaling complex. Proc. Natl Acad. Sci. USA 93, 13973–13978 (1996).
Hsu, H., **ong, J. & Goeddel, D. V. The TNF receptor 1-associated protein TRADD signals cell death and NF-kappa B activation. Cell 81, 495–504 (1995).
Liu, Z. G., Hsu, H., Goeddel, D. V. & Karin, M. Dissection of TNF receptor 1 effector functions: JNK activation is not linked to apoptosis while NF-kappaB activation prevents. Cell Death Cell 87, 565–576 (1996).
Hsu, H., Shu, H. B., Pan, M. G. & Goeddel, D. V. TRADD-TRAF2 and TRADD-FADD interactions define two distinct TNF receptor 1 signal transduction pathways. Cell 84, 299–308 (1996).
Wang, C. Y., Mayo, M. W., Korneluk, R. G., Goeddel, D. V. & Baldwin, A. S. Jr. NF-kappaB antiapoptosis: induction of TRAF1 and TRAF2 and c-IAP1 and c-IAP2 to suppress caspase-8 activation. Science 281, 1680–1683 (1998).
Micheau, O., Lens, S., Gaide, O., Alevizopoulos, K. & Tschopp, J. NF-kappaB signals induce the expression of c-FLIP. Mol. Cell. Biol. 21, 5299–5305 (2001).
Kreuz, S., Siegmund, D., Scheurich, P. & Wajant, H. NF-kappaB inducers upregulate cFLIP, a cycloheximide-sensitive inhibitor of death receptor signaling. Mol. Cell. Biol. 21, 3964–3973 (2001).
Micheau, O. & Tschopp, J. Induction of TNF receptor I-mediated apoptosis via two sequential signaling complexes. Cell 114, 181–190 (2003).
Varfolomeev, E. E. & Ashkenazi, A. Tumor necrosis factor: an apoptosis JuNKie? Cell 116, 491–497 (2004).
Karin, M. & Lin, A. NF-kappaB at the crossroads of life and death. Nat. Immunol. 3, 221–227 (2002).
Beg, A. A. & Baltimore, D. An essential role for NF-kappaB in preventing TNF-alpha-induced cell death. Science 274, 782–784 (1996).
Van Antwerp, D. J., Martin, S. J., Kafri, T., Green, D. R. & Verma, I. M. Suppression of TNF-alpha-induced apoptosis by NF-kappaB. Science 274, 787–789 (1996).
Harper, N., Hughes, M., MacFarlane, M. & Cohen, G. M. Fas-associated death domain protein and caspase-8 are not recruited to the tumor necrosis factor receptor 1 signaling complex during tumor necrosis factor-induced apoptosis. J. Biol. Chem. 278, 25534–25541 (2003).
Wang, L., Du, F. & Wang, X. TNF-alpha induces two distinct caspase-8 activation pathways. Cell 133, 693–703 (2008).
Safa A. R. Roles of c-FLIP in apoptosis, necroptosis, and autophagy. J. Carcinog. Mutagen Suppl 6, 003 (2013).
Tsuchiya, Y., Nakabayashi, O. & Nakano, H. FLIP the switch: regulation of apoptosis and necroptosis by cFLIP. Int. J. Mol. Sci. 16, 30321–30341 (2015).
Humphreys, L., Espona-Fiedler, M. & Longley, D. B. FLIP as a therapeutic target in cancer. FEBS J. 285, 4104–4123 (2018).
Krueger, A., Schmitz, I., Baumann, S., Krammer, P. H. & Kirchhoff, S. Cellular FLICE-inhibitory protein splice variants inhibit different steps of caspase-8 activation at the CD95 death-inducing signaling complex. J. Biol. Chem. 276, 20633–20640 (2001).
Krueger, A., Baumann, S., Krammer, P. H. & Kirchhoff, S. FLICE-inhibitory proteins: regulators of death receptor-mediated apoptosis. Mol. Cell. Biol. 21, 8247–8254 (2001).
Oberst, A. et al. Catalytic activity of the caspase-8-FLIP(L) complex inhibits RIPK3-dependent necrosis. Nature 471, 363–367 (2011).
Lalaoui, N. et al. Mutations that prevent caspase cleavage of RIPK1 cause autoinflammatory disease. Nature 577, 103–108 (2020).
Newton, K. et al. Cleavage of RIPK1 by caspase-8 is crucial for limiting apoptosis and necroptosis. Nature 574, 428–431 (2019).
Zhang, X., Dowling, J. P. & Zhang, J. RIPK1 can mediate apoptosis in addition to necroptosis during embryonic development. Cell Death Dis. 10, 245 (2019).
Tao, P. et al. A dominant autoinflammatory disease caused by non-cleavable variants of RIPK1. Nature 577, 109–114 (2020).
Fu, T. M. et al. Cryo-EM structure of caspase-8 tandem DED filament reveals assembly and regulation mechanisms of the death-inducing signaling complex. Mol. Cell 64, 236–250 (2016).
Fox, J. L. et al. Cryo-EM structural analysis of FADD:Caspase-8 complexes defines the catalytic dimer architecture for co-ordinated control of cell fate. Nat. Commun. 12, 819 (2021).
Yip, K. M., Fischer, N., Paknia, E., Chari, A. & Stark, H. Atomic-resolution protein structure determination by cryo-EM. Nature 587, 157–161 (2020).
Chari, A. & Stark, H. Prospects and limitations of high-resolution single-particle cryo-electron microscopy. Annu. Rev. Biophys. 52, 391–411 (2023).
Park, H. H. et al. The death domain superfamily in intracellular signaling of apoptosis and inflammation. Annu. Rev. Immunol. 25, 561–586 (2007).
Lin, S. C., Lo, Y. C. & Wu, H. Helical assembly in the MyD88-IRAK4-IRAK2 complex in TLR/IL-1R signalling. Nature 465, 885–890 (2010).
Park, H. H. et al. Death domain assembly mechanism revealed by crystal structure of the oligomeric PIDDosome core complex. Cell 128, 533–546 (2007).
Majkut, J. et al. Differential affinity of FLIP and procaspase 8 for FADD’s DED binding surfaces regulates DISC assembly. Nat. Commun. 5, 3350 (2014).
Schleich, K. et al. Stoichiometry of the CD95 death-inducing signaling complex: experimental and modeling evidence for a death effector domain chain model. Mol. Cell 47, 306–319 (2012).
Dickens, L. S. et al. A death effector domain chain DISC model reveals a crucial role for caspase-8 chain assembly in mediating apoptotic cell death. Mol. Cell 47, 291–305 (2012).
Hughes, M. A. et al. Co-operative and hierarchical binding of c-FLIP and caspase-8: a unified model defines how c-FLIP isoforms differentially control cell fate. Mol. Cell 61, 834–849 (2016).
Humphreys, L. M. et al. A revised model of TRAIL-R2 DISC assembly explains how FLIP(L) can inhibit or promote apoptosis. EMBO Rep. 21, e49254 (2020).
Singh, N., Hassan, A. & Bose, K. Molecular basis of death effector domain chain assembly and its role in caspase-8 activation. FASEB J. 30, 186–200 (2016).
Shen, C. et al. Structural basis for dimerization of the death effector domain of the F122A mutant of Caspase-8. Sci. Rep. 8, 16723 (2018).
Muzio, M. et al. FLICE, a novel FADD-homologous ICE/CED-3-like protease, is recruited to the CD95 (Fas/APO-1) death-inducing signaling complex. Cell 85, 817–827 (1996).
Boldin, M. P., Goncharov, T. M., Goltsev, Y. V. & Wallach, D. Involvement of MACH, a novel MORT1/FADD-interacting protease, in Fas/APO-1- and TNF receptor-induced cell death. Cell 85, 803–815 (1996).
Fricker, N. et al. Model-based dissection of CD95 signaling dynamics reveals both a pro- and antiapoptotic role of c-FLIPL. J. Cell Biol. 190, 377–389 (2010).
Jeong, E. J. et al. The solution structure of FADD death domain. Structural basis of death domain interactions of Fas and FADD. J. Biol. Chem. 274, 16337–16342 (1999).
Yu, J. W., Jeffrey, P. D. & Shi, Y. Mechanism of procaspase-8 activation by c-FLIPL. Proc. Natl Acad. Sci. USA 106, 8169–8174 (2009).
Pop, C. et al. FLIP(L) induces caspase 8 activity in the absence of interdomain caspase 8 cleavage and alters substrate specificity. Biochem. J. 433, 447–457 (2011).
Boatright, K. M., Deis, C., Denault, J. B., Sutherlin, D. P. & Salvesen, G. S. Activation of caspases-8 and -10 by FLIP(L). Biochem J. 382, 651–657 (2004).
Dillon, C. P. et al. Survival function of the FADD-CASPASE-8-cFLIP(L) complex. Cell Rep. 1, 401–407 (2012).
Lin, Y. et al. Tumor necrosis factor-induced nonapoptotic cell death requires receptor-interacting protein-mediated cellular reactive oxygen species accumulation. J. Biol. Chem. 279, 10822–10828 (2004).
Irrinki, K. M. et al. Requirement of FADD, NEMO, and BAX/BAK for aberrant mitochondrial function in tumor necrosis factor alpha-induced necrosis. Mol. Cell Biol. 31, 3745–3758 (2011).
Holler, N. et al. Fas triggers an alternative, caspase-8-independent cell death pathway using the kinase RIP as effector molecule. Nat. Immunol. 1, 489–495 (2000).
Cho, Y. S. et al. Phosphorylation-driven assembly of the RIP1-RIP3 complex regulates programmed necrosis and virus-induced inflammation. Cell 137, 1112–1123 (2009).
Osborn, S. L. et al. Fas-associated death domain (FADD) is a negative regulator of T-cell receptor-mediated necroptosis. Proc. Natl Acad. Sci. USA 107, 13034–13039 (2010).
Muppidi, J. R. et al. Homotypic FADD interactions through a conserved RXDLL motif are required for death receptor-induced apoptosis. Cell Death Differ. 13, 1641–1650 (2006).
Sandu, C. et al. FADD self-association is required for stable interaction with an activated death receptor. Cell Death Differ. 13, 2052–2061 (2006).
Yang, J. K. et al. Crystal structure of MC159 reveals molecular mechanism of DISC assembly and FLIP inhibition. Mol. Cell 20, 939–949 (2005).
Lin, Y., Devin, A., Rodriguez, Y. & Liu, Z. G. Cleavage of the death domain kinase RIP by caspase-8 prompts TNF-induced apoptosis. Genes Dev. 13, 2514–2526 (1999).
Chan, F. K. et al. A role for tumor necrosis factor receptor-2 and receptor-interacting protein in programmed necrosis and antiviral responses. J. Biol. Chem. 278, 51613–51621 (2003).
Tummers, B. et al. Caspase-8-dependent inflammatory responses are controlled by its adaptor, FADD, and necroptosis. Immunity 52, 994–1006 (2020).
Edelmann, B. et al. Caspase-8 and caspase-7 sequentially mediate proteolytic activation of acid sphingomyelinase in TNF-R1 receptosomes. EMBO J. 30, 379–394 (2011).
Boucher, D., Blais, V. & Denault, J. B. Caspase-7 uses an exosite to promote poly(ADP ribose) polymerase 1 proteolysis. Proc. Natl Acad. Sci. USA 109, 5669–5674 (2012).
Desroches, A. & Denault, J. B. Caspase-7 uses RNA to enhance proteolysis of poly(ADP-ribose) polymerase 1 and other RNA-binding proteins. Proc. Natl Acad. Sci. USA 116, 21521–21528 (2019).
Henry, C. M. & Martin, S. J. Caspase-8 acts in a non-enzymatic role as a scaffold for assembly of a pro-inflammatory “FADDosome” complex upon TRAIL stimulation. Mol. Cell 65, 715–729 e715 (2017).
Watt, W. et al. The atomic-resolution structure of human caspase-8, a key activator of apoptosis. Structure 7, 1135–1143 (1999).
Eberstadt, M. et al. NMR structure and mutagenesis of the FADD (Mort1) death-effector domain. Nature 392, 941–945 (1998).
Park, H. H. Molecular basis of dimerization of initiator caspase was revealed by crystal structure of caspase-8 pro-domain. Cell Death Differ. 26, 1213–1220 (2019).
Shen, C. et al. Crystal structure of the death effector domains of caspase-8. Biochem. Biophys. Res. Commun. 463, 297–302 (2015).
Su, T. W. et al. Structural insights into DD-fold assembly and caspase-9 activation by the Apaf-1 apoptosome. Structure 25, 407–420 (2017).
Lo, Y. C., Lin, S. C., Yang, C. Y. & Tung, J. Y. Tandem DEDs and CARDs suggest novel mechanisms of signaling complex assembly. Apoptosis 20, 124–135 (2015).
Lin, Y. S. et al. The Src-ZNRF1 axis controls TLR3 trafficking and interferon responses to limit lung barrier damage. J. Exp. Med. 220, e20220727 (2023).
Grootjans, S. et al. A real-time fluorometric method for the simultaneous detection of cell death type and rate. Nat. Protoc. 11, 1444–1454 (2016).
Laussmann, M. A. et al. Proteasome inhibition can impair caspase-8 activation upon submaximal stimulation of apoptotic tumor necrosis factor-related apoptosis inducing ligand (TRAIL) signaling. J. Biol. Chem. 287, 14402–14411 (2012).
Acknowledgements
This work was supported by Academia Sinica (AS) Thematic Program (AS-TP-107-L16, AS-TP-107-L16-1, AS-102-TP-B14 and AS-102-TP-B14-2 to S.-C.L.; AS-TP-107-L16-2 and AS-102-TP-B14-1 to Y.-C.L; AS-TP-107-L16-3 to L.-C.H.; AS Postdoc Fellowships to C.-Y.Y. and T.-W.S.), Ministry of Science and Technology (MoST) (MoST 107-2320-B-001-018-, 108-2311-B-001-018-, 109-2311-B-001-016-, and 110-2311-B-001-015- to S.-C.L.; MoST 107-2320-B-006-062-MY3, and 111-2311-B-006-005-MY3 to Y.-C.L.; MoST 108-2320-B-002-020-MY3, 111-2320-B-002-048-MY3, and 112-2326-B-002-007- to L.-C.H.), and Taiwan Protein Project (TPP) (Grant No. AS-KPQ-105-TPP to Y.-C.L and S.-C.L.). We thank the Protein X-ray Diffractometer, Chameleon, and Vitrobot facilities of GRC for crystallization screening and sample preparation for cryo-EM, DNA Sequencing Core Facility of IBMS (AS CFII Project AS-CFII-113-A12) for DNA sequencing, AS Biological EM Core Facility of ICOB (AS-CFII-111-203) for negative stain EM data, AS Cryo-EM Facility (ASCEM) (AS-CFII-111-210 and TPP AS-KPQ-109-TPP2) for cryo-EM data. We thank the beamline BL13B1 and BL23A1 at NSRRC, Taiwan, for X-ray diffraction and SAXS data, respectively. We thank Prof. Ming-Daw Tsai and Prof. Chih-Hao Lee for their insightful feedback.
Author information
Authors and Affiliations
Contributions
C.-Y.Y. expressed, purified and crystallized the complexes, collected the SAXS, X-ray, and cryo-EM data and solved the structures. C.-I.L. did the cell-based experiments. Y.-C.T. did the cell lysate-based experiments. Y.-C.T., Y.-F.T. and Y.-C.Lu did the pulldown experiments. A.W.K. collected the cryo-EM data of the complex A and helped to solve the structure. C.-Y.Y., T.-W.S. and Y.-T.W. help analyzing the data. C.-Y.Y., C.-I.L., Y.-C.T., Y.-F.T., Y.-C.Lu and Y.-T.W. made the constructs. L.-C.H., Y.-C.Lo and S.-C.L. supervised the project. S.-C.L. and Y.-C.Lo analyzed and interpreted the data, made the final figures, and wrote the manuscript. A.W.K., Y.-C.Lo and L.-C.H. provided comments and revised the manuscript. S.-C.L. initiated the project.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Hyun Ho Park and the other, anonymous, reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yang, CY., Lien, CI., Tseng, YC. et al. Deciphering DED assembly mechanisms in FADD-procaspase-8-cFLIP complexes regulating apoptosis. Nat Commun 15, 3791 (2024). https://doi.org/10.1038/s41467-024-47990-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-024-47990-2
- Springer Nature Limited