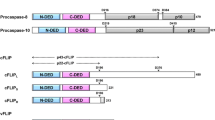
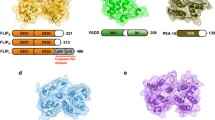
Abstract
Apoptosis is an important process to maintain cellular homeostasis. Deregulated apoptosis has linked to a number of diseases, such as inflammatory diseases, neurodegenerative disorder, and cancers. A major signaling complex in the death receptor signaling pathway leading to apoptosis is death-induced signaling complex (DISC), which is regulated mainly by death effector domain (DED)-containing proteins. There are seven DED-containing proteins in human, including FADD, c-FLIP, caspase-8, caspase-10, DEDD, DEDD2, and PEA-15. The main players in DISC formation employ tandem DEDs for regulating signaling complex formation. The regulatory mechanism of signaling complex formation is important and yet remains unclear. Interestingly, three caspase recruitment domain (CARD)-containing members, which belong to the same DD superfamily as DED-containing proteins, also contains similar tandem CARDs. Recent structural studies have shown that tandem CARDs are essential for the formation of a helical signaling complex. This review summarizes recent structural studies on DED-containing proteins and especially discusses the studies on tandem DEDs and tandem CARDs, which suggest new mechanisms of signaling complex assembly.







Similar content being viewed by others
References
Hanahan D, Weinberg RA (2000) The hallmarks of cancer. Cell 100:57–70
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144:646–674
Green DR, Evan GI (2002) A matter of life and death. Cancer Cell 1:19–30
Vaux DL, Korsmeyer SJ (1999) Cell death in development. Cell 96:245–254
Mattson MP (2000) Apoptosis in neurodegenerative disorders. Nat Rev Mol Cell Biol 1:120–129
Ramaswamy M, Deng M, Siegel RM (2011) Harnessing programmed cell death as a therapeutic strategy in rheumatic diseases. Nat Rev Rheumatol 7:152–160
Reed JC, Doctor KS, Godzik A (2004) The domains of apoptosis: a genomics perspective. Sci STKE 239:re9
Dempsey PW, Doyle SE, He JQ, Cheng G (2003) The signaling adaptors and pathways activated by TNF superfamily. Cytokine Growth Factor Rev 14:193–209
Kischkel FC, Lawrence DA, Chuntharapai A, Schow P, Kim KJ, Ashkenazi A (2000) Apo2L/TRAIL-dependent recruitment of endogenous FADD and caspase-8 to death receptors 4 and 5. Immunity 12:611–620
Holler N, Tardivel A, Kovacsovics-Bankowski M et al (2003) Two adjacent trimeric Fas ligands are required for Fas signaling and formation of a death-inducing signaling complex. Mol Cell Biol 23:1428–1440
Scott FL, Stec B, Pop C et al (2009) The Fas-FADD death domain complex structure unravels signalling by receptor clustering. Nature 457:1019–1022
Peter ME, Krammer PH (2003) The CD95(APO-1/Fas) DISC and beyond. Cell Death Differ 10:26–35
Bao Q, Shi Y (2007) Apoptosome: a platform for the activation of initiator caspases. Cell Death Differ 14:56–65
Fuentes-Prior P, Salvesen GS (2004) The protein structures that shape caspase activity, specificity, activation and inhibition. Biochem J 384:201–232
Riedl SJ, Shi Y (2004) Molecular mechanisms of caspase regulation during apoptosis. Nat Rev Mol Cell Biol 5:897–907
Weber CH, Vincenz C (2001) The death domain superfamily: a tale of two interfaces? Trends Biochem Sci 26:475–481
Park HH, Lo YC, Lin SC, Wang L, Yang JK, Wu H (2007) The death domain superfamily in intracellular signaling of apoptosis and inflammation. Annu Rev Immunol 25:561–586
Chinnaiyan AM, O’Rourke K, Tewari M, Dixit VM (1995) FADD, a novel death domain-containing protein, interacts with the death domain of Fas and initiates apoptosis. Cell 81:505–512
Kischkel FC, Hellbardt S, Behrmann I et al (1995) Cytotoxicity-dependent APO-1 (Fas/CD95)-associated proteins form a death-inducing signaling complex (DISC) with the receptor. EMBO J 14:5579–5588
Boldin MP, Goncharov TM, Goltsev YV, Wallach D (1996) Involvement of MACH, a novel MORT1/FADD-interacting protease, in Fas/APO-1- and TNF receptor-induced cell death. Cell 85:803–815
Muzio M, Chinnaiyan AM, Kischkel FC et al (1996) FLICE, a novel FADD-homologous ICE/CED-3-like protease, is recruited to the CD95 (Fas/APO-1) death–inducing signaling complex. Cell 85:817–827
Shi Y (2004) Caspase activation: revisiting the induced proximity model. Cell 117:855–858
Medema JP, Scaffidi C, Kischkel FC et al (1997) FLICE is activated by association with the CD95 death-inducing signaling complex (DISC). EMBO J 16:2794–2804
Salvesen GS, Dixit VM (1999) Caspase activation: the induced-proximity model. Proc Natl Acad Sci USA 96:10964–10967
Thome M, Schneider P, Hofmann K et al (1997) Viral FLICE-inhibitory proteins (FLIPs) prevent apoptosis induced by death receptors. Nature 386:517–521
Thome M, Tschopp J (2001) Regulation of lymphocyte proliferation and death by FLIP. Nat Rev Immunol 1:50–58
Searles RP, Bergquam EP, Axthelm MK, Wong SW (1999) Sequence and genomic analysis of a Rhesus macaque rhadinovirus with similarity to Kaposi’s sarcoma-associated herpesvirus/human herpesvirus 8. J Virol 73:3040–3053
Hu S, Vincenz C, Buller M, Dixit VM (1997) A novel family of viral death effector domain-containing molecules that inhibit both CD-95- and tumor necrosis factor receptor-1-induced apoptosis. J Biol Chem 272:9621–9624
Bertin J, Armstrong RC, Ottilie S et al (1997) Death effector domain-containing herpesvirus and poxvirus proteins inhibit both Fas- and TNFR1-induced apoptosis. Proc Natl Acad Sci USA 94:1172–1176
Irmler M, Thome M, Hahne M et al (1997) Inhibition of death receptor signals by cellular FLIP. Nature 388:190–195
Golks A, Brenner D, Fritsch C, Krammer PH, Lavrik IN (2005) c-FLIPR, a new regulator of death receptor-induced apoptosis. J Biol Chem 280:14507–14513
Safa AR (2012) c-FLIP, a master anti-apoptotic regulator. Exp Oncol 34:176–184
Ozturk S, Schleich K, Lavrik IN (2012) Cellular FLICE-like inhibitory proteins (c-FLIPs): fine-tuners of life and death decisions. Exp Cell Res 318:1324–1331
Han DK, Chaudhary PM, Wright ME et al (1997) MRIT, a novel death-effector domain-containing protein, interacts with caspases and BclXL and initiates cell death. Proc Natl Acad Sci USA 94:11333–11338
Goltsev YV, Kovalenko AV, Arnold E, Varfolomeev EE, Brodianskii VM, Wallach D (1997) CASH, a novel caspase homologue with death effector domains. J Biol Chem 272:19641–19644
Rasper DM, Vaillancourt JP, Hadano S et al (1998) Cell death attenuation by ‘Usurpin’, a mammalian DED-caspase homologue that precludes caspase-8 recruitment and activation by the CD-95 (Fas, APO-1) receptor complex. Cell Death Differ 5:271–288
Inohara N, Koseki T, Hu Y, Chen S, Nunez G (1997) CLARP, a death effector domain-containing protein interacts with caspase-8 and regulates apoptosis. Proc Natl Acad Sci USA 94:10717–10722
Srinivasula SM, Ahmad M, Ottilie S et al (1997) FLAME-1, a novel FADD-like anti-apoptotic molecule that regulates Fas/TNFR1-induced apoptosis. J Biol Chem 272:18542–18545
Matta H, Chaudhary PM (2004) Activation of alternative NF-kappa B pathway by human herpes virus 8-encoded Fas-associated death domain-like IL-1 beta-converting enzyme inhibitory protein (vFLIP). Proc Natl Acad Sci USA 101:9399–9404
Desbarats J, Birge RB, Mimouni-Rongy M, Weinstein DE, Palerme JS, Newell MK (2003) Fas engagement induces neurite growth through ERK activation and p35 upregulation. Nat Cell Biol 5:118–125
Schneider P, Thome M, Burns K et al (1997) TRAIL receptors 1 (DR4) and 2 (DR5) signal FADD-dependent apoptosis and activate NF-kappaB. Immunity 7:831–836
Neumann L, Pforr C, Beaudouin J et al (2010) Dynamics within the CD95 death-inducing signaling complex decide life and death of cells. Mol Syst Biol 6:352
Kataoka T, Budd RC, Holler N et al (2000) The caspase-8 inhibitor FLIP promotes activation of NF-kappaB and Erk signaling pathways. Curr Biol 10:640–648
Kreuz S, Siegmund D, Rumpf JJ et al (2004) NFkappaB activation by Fas is mediated through FADD, caspase-8, and RIP and is inhibited by FLIP. J Cell Biol 166:369–380
Djerbi M, Darreh-Shori T, Zhivotovsky B, Grandien A (2001) Characterization of the human FLICE-inhibitory protein locus and comparison of the anti-apoptotic activity of four different flip isoforms. Scand J Immunol 54:180–189
Schickling O, Stegh AH, Byrd J, Peter ME (2001) Nuclear localization of DEDD leads to caspase-6 activation through its death effector domain and inhibition of RNA polymerase I dependent transcription. Cell Death Differ 8:1157–1168
Roth W, Stenner-Liewen F, Pawlowski K, Godzik A, Reed JC (2002) Identification and characterization of DEDD2, a death effector domain-containing protein. J Biol Chem 277:7501–7508
Lv Q, Wang W, Xue J et al (2012) DEDD interacts with PI3KC3 to activate autophagy and attenuate epithelial-mesenchymal transition in human breast cancer. Cancer Res 72:3238–3250
Sharif A, Canton B, Junier MP, Chneiweiss H (2003) PEA-15 modulates TNFalpha intracellular signaling in astrocytes. Ann NY Acad Sci 1010:43–50
Fiory F, Formisano P, Perruolo G, Beguinot F (2009) Frontiers: PED/PEA-15, a multifunctional protein controlling cell survival and glucose metabolism. Am J Physiol Endocrinol Metab 297:E592–E601
Garvey T, Bertin J, Siegel R, Lenardo M, Cohen J (2002) The death effector domains (DEDs) of the molluscum contagiosum virus MC159 v-FLIP protein are not functionally interchangeable with each other or with the DEDs of caspase-8. Virology 300:217–225
Eberstadt M, Huang B, Chen Z et al (1998) NMR structure and mutagenesis of the FADD (Mort1) death-effector domain. Nature 392:941–945
Hill JM, Vaidyanathan H, Ramos JW, Ginsberg MH, Werner MH (2002) Recognition of ERK MAP kinase by PEA-15 reveals a common docking site within the death domain and death effector domain. EMBO J 21:6494–6504
Twomey EC, Wei Y (2012) High-definition NMR structure of PED/PEA-15 death effector domain reveals details of key polar side chain interactions. Biochem Biophys Res Commun 424:141–146
Yang JK, Wang L, Zheng L et al (2005) Crystal structure of MC159 reveals molecular mechanism of DISC assembly and FLIP inhibition. Mol Cell 20:939–949
Bagneris C, Ageichik AV, Cronin N et al (2008) Crystal structure of a vFlip-IKKgamma complex: insights into viral activation of the IKK signalosome. Mol Cell 30:620–631
Mace PD, Wallez Y, Egger MF et al (2013) Structure of ERK2 bound to PEA-15 reveals a mechanism for rapid release of activated MAPK. Nat Commun 4:1681
Arnold K, Bordoli L, Kopp J, Schwede T (2006) The SWISS-MODEL workspace: a web-based environment for protein structure homology modelling. Bioinformatics 22:195–201
Park HH, Logette E, Raunser S et al (2007) Death domain assembly mechanism revealed by crystal structure of the oligomeric PIDDosome core complex. Cell 128:533–546
Lin SC, Lo YC, Wu H (2010) Helical assembly in the MyD88-IRAK4-IRAK2 complex in TLR/IL-1R signalling. Nature 465:885–890
Wu B, Peisley A, Tetrault D et al (2014) Molecular Imprinting as a Signal-Activation Mechanism of the Viral RNA Sensor RIG-I. Mol Cell 55:511–523
Dickens LS, Boyd RS, Jukes-Jones R et al (2012) A death effector domain chain DISC model reveals a crucial role for caspase-8 chain assembly in mediating apoptotic cell death. Mol Cell 47:291–305
Schleich K, Warnken U, Fricker N et al (2012) Stoichiometry of the CD95 death-inducing signaling complex: experimental and modeling evidence for a death effector domain chain model. Mol Cell 47:306–319
Acknowledgments
This work is supported by Ministry of Science and Technology Grant MOST 101-2311-B-006-008-MY3 and Academia Sinica Thematic Research Program AS-102-TP-B14-1 (to Y.C.L.), and Ministry of Science and Technology Grant MOST 101-2320-B-001-034-MY3 and Academia Sinica Thematic Research Program AS-102-TP-B14-2 (to S.C.L.), and Academia Sinica Postdoc Fellowship (to C.Y.Y.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lo, YC., Lin, SC., Yang, CY. et al. Tandem DEDs and CARDs suggest novel mechanisms of signaling complex assembly. Apoptosis 20, 124–135 (2015). https://doi.org/10.1007/s10495-014-1054-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-014-1054-4