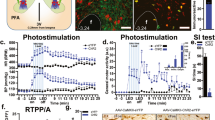
Abstract
Panic disorder is characterized by uncontrollable fear accompanied by somatic symptoms that distinguish it from other anxiety disorders. Neural mechanisms underlying these unique symptoms are not completely understood. Here, we report that the pituitary adenylate cyclase-activating polypeptide (PACAP)-expressing neurons in the lateral parabrachial nucleus projecting to the dorsal raphe are crucial for panic-like behavioral and physiological alterations. These neurons are activated by panicogenic stimuli but inhibited in conditioned fear and anxiogenic conditions. Activating these neurons elicits strong defensive behaviors and rapid cardiorespiratory increase without creating aversive memory, whereas inhibiting them attenuates panic-associated symptoms. Chemogenetic or pharmacological inhibition of downstream PACAP receptor-expressing dorsal raphe neurons abolishes panic-like symptoms. The pontomesencephalic PACAPergic pathway is therefore a likely mediator of panicogenesis, and may be a promising therapeutic target for treating panic disorder.







Similar content being viewed by others

Data availability
All data associated with this study are available in the supplementary information. Source data are provided with this paper.
References
Killgore, W. D. S. et al. Cortico-limbic responses to masked affective faces across PTSD, panic disorder, and specific phobia. Depress Anxiety 31, 150–159 (2014).
Crowe, R. R., Noyes, R., Pauls, D. L. & Slymen, D. A family study of panic disorder. Arch. Gen. Psychiatry 40, 1065–1069 (1983).
Katon, W. Panic disorder and somatization. Review of 55 cases. Am. J. Med. 77, 101–106 (1984).
Meuret, A. E., Kroll, J. & Ritz, T. Panic disorder comorbidity with medical conditions and treatment implications. Annu. Rev. Clin. Psychol. 13, 209–240 (2017).
Gorman, J. M., Kent, J. M., Sullivan, G. M. & Coplan, J. D. Neuroanatomical hypothesis of panic disorder, revised. Am. J. Psychiatry 157, 493–505 (2000).
Coplan, J. D. & Lydiard, R. B. Brain circuits in panic disorder. Biol. Psychiatry 44, 1264–1276 (1998).
Shekhar, A., Sajdyk, T. J., Gehlert, D. R. & Rainnie, D. G. The amygdala, panic disorder, and cardiovascular responses. Ann. N. Y. Acad. Sci. 985, 308–325 (2003).
Kim, J. E., Dager, S. R. & Lyoo, I. K. The role of the amygdala in the pathophysiology of panic disorder: evidence from neuroimaging studies. Biol. Mood Anxiety Disord. 2, 20 (2012).
Yoon, S. et al. Subregional shape alterations in the amygdala in patients with panic disorder. PLoS ONE 11, e0157856 (2016).
Feinstein, J. S., Adolphs, R., Damasio, A. & Tranel, D. The human amygdala and the induction and experience of fear. Curr. Biol. 21, 34–38 (2011).
Khalsa, S. S. et al. Panic anxiety in humans with bilateral amygdala lesions: pharmacological induction via cardiorespiratory interoceptive pathways. J. Neurosci. 36, 3559–3566 (2016).
Feinstein, J. S. et al. Fear and panic in humans with bilateral amygdala damage. Nat. Neurosci. 16, 270–272 (2013).
Chiang, M. C. et al. Parabrachial complex: a hub for pain and aversion. J. Neurosci. 39, 8225–8230 (2019).
Han, S., Soleiman, M. T., Soden, M. E., Zweifel, L. S. & Palmiter, R. D. Elucidating an affective pain circuit that creates a threat memory. Cell 162, 363–374 (2015).
Davern, P. J. A role for the lateral parabrachial nucleus in cardiovascular function and fluid homeostasis. Front. Physiol. 5, 436 (2014).
Yahiro, T., Kataoka, N., Nakamura, Y. & Nakamura, K. The lateral parabrachial nucleus, but not the thalamus, mediates thermosensory pathways for behavioural thermoregulation. Sci. Rep. 7, 5031 (2017).
Liu, S. et al. Neural basis of opioid-induced respiratory depression and its rescue. Proc. Natl Acad. Sci. USA 118, e2022134118 (2021).
Bourin, M., Baker, G. B. & Bradwejn, J. Neurobiology of panic disorder. J. Psychosom. Res. 44, 163–180 (1998).
Singewald, N. & Sharp, T. Neuroanatomical targets of anxiogenic drugs in the hindbrain as revealed by Fos immunocytochemistry. Neuroscience 98, 759–770 (2000).
Brannan, S. et al. Neuroimaging of cerebral activations and deactivations associated with hypercapnia and hunger for air. Proc. Natl Acad. Sci. USA 98, 2029–2034 (2001).
Perna, G., Caldirola, D. & Bellodi, L. Panic disorder: from respiration to the homeostatic brain. Acta Neuropsychiatr. 16, 57–67 (2004).
Cardoso, J. C. R., Garcia, M. G. & Power, D. M. Tracing the origins of the pituitary adenylate-cyclase activating polypeptide (PACAP). Front. Neurosci. 14, 366 (2020).
Zhang, L. et al. Behavioral role of PACAP signaling reflects its selective distribution in glutamatergic and GABAergic neuronal subpopulations. eLife 10, e61718 (2021).
Stroth, N., Holighaus, Y., Ait-Ali, D. & Eiden, L. E. PACAP: a master regulator of neuroendocrine stress circuits and the cellular stress response. Ann. N. Y. Acad. Sci. 1220, 49–59 (2011).
Ressler, K. J. et al. Post-traumatic stress disorder is associated with PACAP and the PAC1 receptor. Nature 470, 492–497 (2011).
Cho, J.-H. et al. Pituitary adenylate cyclase-activating polypeptide induces postsynaptically expressed potentiation in the intra-amygdala circuit. J. Neurosci. 32, 14165–14177 (2012).
Iurato, S. et al. DNA methylation signatures in panic disorder. Transl. Psychiatry 7, 1287 (2017).
Erhardt, A., Lucae, S., Ising, M., Holsboer, F. & Binder, E. B. Association of PACAP and PACAPR1 gene variants with unipolar depression and panic disorder. Pharmacopsychiatry 46, A89 (2013).
Evans, A. K. & Lowry, C. A. Pharmacology of the β-carboline FG-7142, a partial inverse agonist at the benzodiazepine allosteric site of the GABAA receptor: neurochemical, neurophysiological, and behavioral effects. CNS Drug Rev. 13, 475–501 (2007).
Ye, J. & Veinante, P. Cell-type specific parallel circuits in the bed nucleus of the stria terminalis and the central nucleus of the amygdala of the mouse. Brain Struct. Funct. 224, 1067–1095 (2019).
Boucher, M. N., Aktar, M., Braas, K. M., May, V. & Hammack, S. E. Activation of lateral parabrachial nucleus (LPBn) PACAP-expressing projection neurons to the bed nucleus of the stria terminalis (BNST) enhances anxiety-like behavior. J. Mol. Neurosci. 72, 451–458 (2021).
Missig, G. et al. Parabrachial nucleus (PBn) pituitary adenylate cyclase activating polypeptide (PACAP) signaling in the amygdala: implication for the sensory and behavioral effects of pain. Neuropharmacology 86, 38–48 (2014).
Ziemann, A. E. et al. The amygdala is a chemosensor that detects carbon dioxide and acidosis to elicit fear behavior. Cell 139, 1012–1021 (2009).
Leibold, N. K. et al. CO2 exposure as translational cross-species experimental model for panic. Transl. Psychiatry 6, e885 (2016).
Perna, G. et al. Carbon dioxide/oxygen challenge test in panic disorder. Psychiatry Res. 52, 159–171 (1994).
Dorow, R., Horowski, R., Paschelke, G., Amin, M. & Braestrup, C. Severe anxiety induced by FG 7142, a β-carboline ligand for benzodiazepine receptors. Lancet 322, 98–99 (1983).
McGregor, I. S., Lee, A. M. & Westbrook, R. F. Stress-induced changes in respiratory quotient, energy expenditure and locomotor activity in rats: effects of midazolam. Psychopharmacology 116, 475–482 (1994).
Patki, G. et al. Tempol treatment reduces anxiety-like behaviors induced by multiple anxiogenic drugs in rats. PLoS ONE 10, e0117498 (2015).
Federici, L. M., Caliman, I. F., Fitz, S. D., Shekhar, A. & Johnson, P. L. Select panicogenic drugs and stimuli induce consistent increases in tail skin flushes and decreases in core body temperature. Behav. Pharmacol. 30, 376–382 (2019).
Salchner, P. et al. Airjet and FG-7142-induced Fos expression differs in rats selectively bred for high and low anxiety-related behavior. Neuropharmacology 50, 1048–1058 (2006).
Johnson, P. L. et al. Orexin 1 receptors are a novel target to modulate panic responses and the panic brain network. Physiol. Behav. 107, 733–742 (2012).
Palmiter, R. D. The parabrachial nucleus: CGRP neurons function as a general alarm. Trends Neurosci. 41, 280–293 (2018).
Kang, S. J. et al. A central alarm system that gates multi-sensory innate threat cues to the amygdala. Cell Rep. 40, 111222 (2022).
Iversen, S. D. 5-HT and anxiety. Neuropharmacology 23, 1553–1560 (1984).
Kahn, R. S., Asnis, G. M., Wetzler, S. & Praag, H. M. Neuroendocrine evidence for serotonin receptor hypersensitivity in panic disorder. Psychopharmacology 96, 360–364 (1988).
Kahn, R. S., Wetzler, S., Praag, H. M., Asnis, G. M. & Strauman, T. Behavioral indications for serotonin receptor hypersensitivity in panic disorder. Psychiatry Res. 25, 101–104 (1988).
Ren, J. et al. Anatomically defined and functionally distinct dorsal raphe serotonin sub-systems. Cell 175, 472–487.e20 (2018).
Paul, E. D. & Lowry, C. A. Functional topography of serotonergic systems supports the Deakin/Graeff hypothesis of anxiety and affective disorders. J. Psychopharmacol. 27, 1090–1106 (2013).
Luskin, A. T. et al. A diverse network of pericoerulear neurons control arousal states. Preprint at bioRxiv https://doi.org/10.1101/2022.06.30.498327 (2022).
Borkar, C. D. et al. Sex differences in behavioral responses during a conditioned flight paradigm. Behav. Brain Res. 389, 112623 (2020).
Dorofeikova, M. et al. Effects of footshock stress on social behavior and neuronal activation in the medial prefrontal cortex and amygdala of male and female mice. PLoS ONE 18, e0281388 (2023).
Eiden, L. E., Goosens, K. A., Jacobson, K. A., Leggio, L. & Zhang, L. Peptide-liganded G protein-coupled receptors as neurotherapeutics. ACS Pharmacol. Transl. Sci. 3, 190–202 (2020).
Spampanato, J., Polepalli, J. & Sah, P. Interneurons in the basolateral amygdala. Neuropharmacology 60, 765–773 (2011).
Gilpin, N. W., Herman, M. A. & Roberto, M. The central amygdala as an integrative hub for anxiety and alcohol use disorders. Biol. Psychiatry 77, 859–869 (2015).
Duvarci, S., Popa, D. & Paré, D. Central amygdala activity during fear conditioning. J. Neurosci. 31, 289–294 (2011).
Fogaça, M. V. & Duman, R. S. Cortical GABAergic dysfunction in stress and depression: new insights for therapeutic interventions. Front. Cell. Neurosci. 13, 87 (2019).
Brambilla, P., Perez, J., Barale, F., Schettini, G. & Soares, J. C. GABAergic dysfunction in mood disorders. Mol. Psychiatry 8, 721–737 (2003).
Maller, R. G. & Reiss, S. Anxiety sensitivity in 1984 and panic attacks in 1987. J. Anxiety Disord. 6, 241–247 (1992).
Goodwin, R. D. et al. Panic attack as a risk factor for severe psychopathology. Am. J. Psychiatry 161, 2207–2214 (2004).
Coryell, W. et al. Depression and panic attacks: the significance of overlap as reflected in follow-up and family study data. Am. J. Psychiatry 145, 293–300 (1988).
Walker, D. L., Toufexis, D. J. & Davis, M. Role of the bed nucleus of the stria terminalis versus the amygdala in fear, stress, and anxiety. Eur. J. Pharmacol. 463, 199–216 (2003).
Lee, Y. & Davis, M. Role of the hippocampus, the bed nucleus of the stria terminalis, and the amygdala in the excitatory effect of corticotropin-releasing hormone on the acoustic startle reflex. J. Neurosci. 17, 6434–6446 (1997).
Waddell, J., Morris, R. W. & Bouton, M. E. Effects of bed nucleus of the stria terminalis lesions on conditioned anxiety: aversive conditioning with long-duration conditional stimuli and reinstatement of extinguished fear. Behav. Neurosci. 120, 324–336 (2006).
Taugher, R. J. et al. The bed nucleus of the stria terminalis is critical for anxiety-related behavior evoked by CO2 and acidosis. J. Neurosci. 34, 10247–10255 (2014).
Kaur, S. & Saper, C. B. Neural circuitry underlying waking up to hypercapnia. Front. Neurosci. 13, 401 (2019).
Song, G. & Poon, C.-S. Lateral parabrachial nucleus mediates shortening of expiration and increase of inspiratory drive during hypercapnia. Respir. Physiol. Neurobiol. 165, 9–12 (2009).
Breier, A., Charney, D. S. & Heninger, G. R. Agoraphobia with panic attacks: development, diagnostic stability, and course of illness. Arch. Gen. Psychiatry 43, 1029–1036 (1986).
Kessler, R. C. et al. The epidemiology of panic attacks, panic disorder, and agoraphobia in the National Comorbidity Survey replication. Arch. Gen. Psychiatry 63, 415–424 (2006).
Chiang, M. C. et al. Divergent neural pathways emanating from the lateral parabrachial nucleus mediate distinct components of the pain response. Neuron 106, 927–939.e5 (2020).
Sun, L. et al. Parabrachial nucleus circuit governs neuropathic pain-like behavior. Nat. Commun. 11, 5974 (2020).
Deakin, J. F. & Graeff, F. G. 5-HT and mechanisms of defence. J. Psychopharmacol. 5, 305–315 (1991).
Graeff, F. G. Serotonin, the periaqueductal gray and panic. Neurosci. Biobehav. Rev. 28, 239–259 (2004).
Pobbe, R. L. H., Zangrossi, H., Blanchard, D. C. & Blanchard, R. J. Involvement of dorsal raphe nucleus and dorsal periaqueductal gray 5-HT receptors in the modulation of mouse defensive behaviors. Eur. Neuropsychopharmacol. 21, 306–315 (2011).
Bago, M. & Dean, C. Sympathoinhibition from ventrolateral periaqueductal gray mediated by 5-HT1A receptors in the RVLM. Am. J. Physiol. 280, R976–R984 (2001).
Teissier, A. et al. Activity of raphé serotonergic neurons controls emotional behaviors. Cell Rep. 13, 1965–1976 (2015).
Correia, P. A. et al. Transient inhibition and long-term facilitation of locomotion by phasic optogenetic activation of serotonin neurons. eLife 6, e20975 (2017).
Seo, C. et al. Intense threat switches dorsal raphe serotonin neurons to a paradoxical operational mode. Science 363, 538–542 (2019).
Walsh, J. J. et al. 5-HT release in nucleus accumbens rescues social deficits in mouse autism model. Nature 560, 589–594 (2018).
Cathala, A. et al. Serotonin2B receptor blockade in the rat dorsal raphe nucleus suppresses cocaine-induced hyperlocomotion through an opposite control of mesocortical and mesoaccumbens dopamine pathways. Neuropharmacology 180, 108309 (2020).
Kusljic, S. & Van Den Buuse, M. Differential role of serotonin projections from the dorsal and median raphe nuclei in phencyclidine-induced hyperlocomotion and fos-like immunoreactivity in rats. Synapse 66, 885–892 (2012).
Sukamoto, T., Yamamoto, T., Watanabe, S. & Ueki, S. Cardiovascular responses to centrally administered serotonin in conscious normotensive and spontaneously hypertensive rats. Eur. J. Pharmacol. 100, 173–179 (1984).
Gradin, K., Qadri, F., Nomikos, G. G., Hillegaart, V. & Svensson, T. H. Substance P injection into the dorsal raphe increases blood pressure and serotonin release in hippocampus of conscious rats. Eur. J. Pharmacol. 218, 363–367 (1992).
Lovick, T. A. Influence of the dorsal and median raphe nuclei on neurons in the periaqueductal gray matter: role of 5-hydroxytryptamine. Neuroscience 59, 993–1000 (1994).
Courtney, N. A. & Ford, C. P. Mechanisms of 5-HT1A receptor-mediated transmission in dorsal raphe serotonin neurons. J. Physiol. 594, 953–965 (2016).
Gantz, S. C., Levitt, E. S., Llamosas, N., Neve, K. A. & Williams, J. T. Depression of serotonin synaptic transmission by the dopamine precursor l-DOPA. Cell Rep. 12, 944–954 (2015).
Perna, G., Schruers, K., Alciati, A. & Caldirola, D. Novel investigational therapeutics for panic disorder. Expert Opin. Investig. Drugs 24, 491–505 (2015).
Kaur, S. et al. Role of serotonergic dorsal raphe neurons in hypercapnia-induced arousals. Nat. Commun. 11, 2769 (2020).
Cho, H.-Y., Kim, M. & Han, J.-H. Specific disruption of contextual memory recall by sparse additional activity in the dentate gyrus. Neurobiol. Learn. Mem. 145, 190–198 (2017).
Jeong, Y. et al. Synaptic plasticity-dependent competition rule influences memory formation. Nat. Commun. 12, 3915 (2021).
Acknowledgements
We thank members of the Han laboratory for critical discussion of the paper and D. O’Keefe for critical input on the manuscript. We also thank S. Park for proofreading the response letter to the reviewers. The study was funded by a National Institutes of Mental Health Biobehavioral Research Award for Innovative New Scientists (BRAINS) grant 1R01MH116203 (S.H.) and a Bridge to Independence award from the Simons Foundation Autism Research Initiative SFARI no. 388708 (S.H.).
Author information
Authors and Affiliations
Contributions
S.H., J.-H.K. and S.J.K. conceptualized the study. S.H., J.-H.K. and S.J.K. developed the methodology. J.-H.K. and S.J.K. performed the investigations and visualizations. S.H. acquired funding. D.-I.K. provided viruses. S.H., J.-H.K., S.J.K. and B.Z.R. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Neuroscience thanks Kerry Ressler and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Projections from PACAPPBL neurons.
a, Schematic and histological confirmation of Cre-dependent expression of EYFP in the PBL of an Adcyap1Cre/+ mouse. Scale bars: 100 µm. b, Representative images of the output regions of PACAPPBL neurons. Scale bars: 100 µm. Injections were repeated on three mice with similar results.
Extended Data Fig. 2 Behavioral changes of the PACAPPBL→DR::GCaMP6s mice during panicogenic conditions.
a, Heat map and distance traveled before- (normal air) and after CO2 exposure. Paired two-sided t-test, P < 0.001. n = 7 mice. b, Heat map and distance traveled following FG-7142 or control injection. Repeated measure two-way ANOVA with Sidak’s multiple comparisons test. n = 7 mice. Data are presented as the mean ± SEM; see also Supplementary Table 3 for statistical details. *P < 0.05, **P < 0.01.
Extended Data Fig. 3 Calcium activity changes in PACAPPBL→DR neurons do not respond as a general multi-modal aversive circuit.
a, Average calcium trace during “Conditioning” phase of the fear conditioning test. Blue shading indicates “Tone ON” periods and orange arrows indicate when the foot shock was given. n = 3 mice. b, Average calcium trace during the looming test with corresponding AUC analysis. Pink shading indicates the 2-s looming exposure. Repeated measure one-way ANOVA with Sidak’s multiple comparisons test, F (2, 6) = 240.127, P < 0.001. n = 4 mice. Data are presented as the mean ± SEM; see also Supplementary Table 3 for statistical details. **P < 0.01.
Extended Data Fig. 4 Calcium activity changes in PACAPPBL→DR neurons during anxiogenic conditions.
a, Schematic of the elevated platform test. b, Average calcium trace during elevated platform assessment, and corresponding area under curve (AUC) analysis. Paired two-sided t-test, P = 0.0017. n = 4 mice. c, Schematic of the elevated plus maze. d, Average calcium trace during the seconds immediately preceding and following open arm entry and exit, with corresponding AUC analysis. Paired two-sided t-test, P < 0.0001. n = 4 mice. e, Calcium traces from individual animals. Pink shading indicates when mice were in the open arm. n = 4 mice. Data are presented as the mean ± SEM; see also Supplementary Table 3 for statistical details. **P < 0.01, ***P < 0.001.
Extended Data Fig. 5 Inputs to PACAPPBL→DR neurons.
a, Schematics of Cre- and Flp-dependent retrograde tracing in PBL of an Adcyap1Cre/+ mouse for identification of inputs to PACAPPBL→DR neurons. b, Histology of TVA and RVdG expression in PBL. Scale bars: 50 µm. Injections were repeated on three mice with similar results. c, Representative images of cTRIO tracing in BNST and CeA. Scale bars: 50 µm. Injections were repeated on three mice with similar results. d, Schematic of inputs to PACAPPBL→DR neurons. Repeated measure one-way ANOVA with Sidak’s multiple comparisons test, F (4, 8) = 21.871, P = 0.0073. n = 3 mice. e,f, Histological images of RVdG and VGAT expression, and graphical representation of the percentage of VGAT colocalization in BNST (e) and CeA (f). Scale bars: 50 µm. n = 3 mice. Data are presented as the mean ± SEM; see also Supplementary Table 3 for counting and statistical details.
Extended Data Fig. 6 Inhibition of PACAPPBL→DR neurons attenuates panic-like symptoms.
a, Distance traveled before (normal air) and after CO2 exposure during chemogenetic inhibition of PACAPPBL→DR neurons. Mixed-effects analysis with Sidak’s multiple comparisons test. n = 9 control mice, n = 6 hM4Di mice. b,c, Distance traveled (b, repeated measures two-way ANOVA with Sidak’s multiple comparisons test) and velocity (c, repeated measures two-way ANOVA with Sidak’s multiple comparisons test) after FG-7142 injection during chemogenetic inhibition of PACAPPBL→DR neurons. n = 9 control mice, n = 5 hM4Di mice. Data are presented as the mean ± SEM; see also Supplementary Table 3 for statistical details. *P < 0.05, **P < 0.01.
Extended Data Fig. 7 PAC1RDR neurons are monosynaptically connected with PACAPPBL neurons.
a, Single channel histological images of Fig.7g. Presynaptic PACAPPBL neurons are also VGluT2 (Slc17a6) positive. b, Single channel histological images of Fig.7h. Presynaptic PACAPPBL neurons do not overlap with VGAT (Slc32a1). c, Histological images of DR after perfusing the retrograde rabies tracing mice. TVA (green), RVdG (red). Starter cells are observable. d, Representative images of presynaptic cells (RVdG, red) in PBL. Scale bars: 100 µm. This experiment was repeated on three mice with similar results.
Extended Data Fig. 8 Inhibition of PAC1RDR neurons blocks panic-like symptoms.
a, Heat map of mouse activity before (air) and after CO2 exposure during chemogenetic inhibition of PAC1RDR neurons. n = 7 mice per group. b,c, Heat map of mouse activity (b) and velocity (c, mixed-effects analysis with Sidak’s multiple comparisons test and adjusted P values) after FG-7142 injection during chemogenetic inhibition of PAC1RDR neurons. n = 7 mice per group. Data are presented as the mean ± SEM; see also Supplementary Table 3 for statistical details. **P < 0.01, ***P < 0.001.
Extended Data Fig. 9 The majority of PAC1R (Adcyap1r1) neurons are SERT-positive.
a, Representative in situ hybridization images of PAC1R- (Adcyap1r1) and SERT-positive neurons in the DR (left). b, Graphical representation of percentages of fluorescent neurons. n = 7 mice. Scale bars: 50 µm. Data are presented as the mean ± SEM.; see also Supplementary Table 2 for counting details.
Extended Data Fig. 10 Projection targets of PAC1RDR neurons.
a, Histological images of output regions from PAC1RDR neurons expressing ChR2. Abbreviations: lateral septal nucleus (LS), BNST, CeA, paraventricular nucleus of the thalamus (PVT), nucleus of reuniens (RE), paraventricular hypothalamic nucleus (PVH), lateral hypothalamus (LHA), dorsomedial nucleus of the hypothalamus (DMH), lateral geniculate complex (LGd), subparafascicular nucleus parviceullar part (SPFp), peripeduncular nucleus (PP), substantia nigra compact part (SNc), ventral tegmental area (VTA), ifterfascicular nucleus raphe (IF), rostral linear uncleus raphe (RL), Peri-locus ceruleus (Peri-LC), LC, nucleus raphe magnus (RM), and pontine reticular nucleus (PRN). Scale bars: 100 µm. n = 5 mice.
Supplementary information
Supplementary Information
Supplementary Tables 1–3.
Supplementary Video 1
Photostimulation of PACAPPBL→DR neurons induced immediate panic-like behaviors. ChR2 was expressed in PBL of an Adcyap1Cre/+ mouse Cre- and Flp-dependently for optogenetic activation of PACAPPBL→DR neurons. The mouse immediately immobilized during the 20-Hz photostimulation.
Supplementary Video 2
Photostimulation of PACAPPBL→DR neurons in the control group has no effect. EYFP was expressed in the PBL of an Adcyap1Cre/+ mouse Cre- and Flp-dependently as a control for optogenetic activation of PACAPPBL→DR neurons. The mouse showed no behavioral change during photostimulation.
Supplementary Video 3
Optogenetic conditioning of PACAPPBL→DR neurons did not form contextual fear memory. Optogenetically conditioned PACAPPBL→DR::ChR2 mice did not show freezing behaviors during the context test.
Supplementary Video 4
Optogenetic conditioning of PACAPPBL→DR neurons did not form cued fear memory. Optogenetically conditioned PACAPPBL→DR::ChR2 mice did not show freezing behaviors during the cue test.
Supplementary Video 5
Photostimulation of PACAPPBL→DR neurons induced tail-rattling in the initial phase of stimulation. Optogenetic 20-Hz photostimulation of PACAPPBL→DR::ChR2 mice sometimes induced tail-rattling in the initial phase of the stimulation.
Supplementary Video 6
Photostimulation of PAC1RDR neurons induced immediate immobilization. Optogenetic 40-Hz photostimulation of PAC1RDR::ChR2 mice induced immediate immobilization.
Source data
Source Data Fig. 1
Statistical source data.
Source Data Fig. 2
Statistical source data.
Source Data Fig. 3
Statistical source data.
Source Data Fig. 4
Statistical source data.
Source Data Fig. 5
Statistical source data.
Source Data Fig. 6
Statistical source data.
Source Data Fig. 7
Statistical source data.
Source Data Extended Data Fig. 2
Statistical source data.
Source Data Extended Data Fig. 3
Statistical source data.
Source Data Extended Data Fig. 4
Statistical source data.
Source Data Extended Data Fig. 6
Statistical source data.
Source Data Extended Data Fig. 8
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kang, S.J., Kim, JH., Kim, DI. et al. A pontomesencephalic PACAPergic pathway underlying panic-like behavioral and somatic symptoms in mice. Nat Neurosci 27, 90–101 (2024). https://doi.org/10.1038/s41593-023-01504-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41593-023-01504-3
- Springer Nature America, Inc.