Abstract
Background
Aging may affect the efficacy of Helicobacter pylori eradication. The aim of our study was to assess the efficacy and safety of 7-day non-bismuth concomitant quadruple therapy as a first-line H. pylori infection eradication regimen in elderly individuals.
Methods
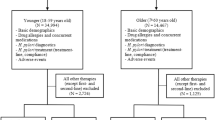
We retrospectively analyzed a cohort with prospectively collected data from January 2013 to December 2019 at Chang Gung Memorial Hospital in Kaohsiung. There were 408 naive infected subjects aged 20 years or older who were treated with 7 days of concomitant therapy as a first-line H. pylori eradication regimen. We divided the patients into an elderly group (aged ≥ 65 years) and a control group (aged < 65 years). Two patients were lost during follow-up in the elderly group and 29 patients were lost in the control group, resulting in 56 in the ≥ 65-year age group and 321 in the control group. The patients were asked to perform urea breath tests 8 weeks later.
Results
The eradication rates for the elderly and control groups were 93.1% (95% confidence interval (CI): 83.3–98.1) and 84.0% (95% CI 79.7–87.7) (p = 0.070), respectively, in the intention-to-treat analysis, and 96.4% (95% CI 87.6–99.6) and 91.6% (95% CI 88.0–94.4) (p = 0.210), respectively, in the per-protocol (PP) analysis. The adverse event rates were 8.9% in the elderly group and 12.8% in the control group (p = 0.417). The compliance was 100% in both groups. No significant difference was seen in antibiotic resistance in either group. Multivariate analysis revealed that metronidazole resistance (odds ratio (OR) 6.870, 95% CI 1.182–39.919, p = 0.032) and dual-therapy resistance (OR 7.188, 95% CI 1.326–38.952, p = 0.022) were independent factors for eradication failure.
Conclusions
The efficacy of non-bismuth concomitant quadruple therapy in the elderly cohort was comparable with that in the non-elderly cohort for first-line H. pylori eradication with acceptable adverse effects.

Similar content being viewed by others
References
Malfertheiner P, Megraud F, O’Morain CA, Gisbert JP, Kuipers EJ, Axon AT, et al. Management of Helicobacter pylori infection-the Maastricht V/Florence Consensus Report. Gut. 2017;66(1):6–30.
Soenen S, Rayner CK, Jones KL, Horowitz M. The ageing gastrointestinal tract. Curr Opin Clin Nutr Metab Care. 2016;19(1):12–8. https://doi.org/10.1097/MCO.0000000000000238.
Cizginer S, Ordulu Z, Kadayifci A. Approach to Helicobacter pylori infection in geriatric population. World J Gastrointest Pharmacol Ther. 2014;5(3):139–47. https://doi.org/10.4292/wjgpt.v5.i3.139.
Lee YC, Chen TH, Chiu HM, Shun CT, Chiang H, Liu TY, et al. The benefit of mass eradication of Helicobacter pylori infection: a community-based study of gastric cancer prevention. Gut. 2013;62(5):676–82.
Kong YJ, Yi HG, Dai JC, Wei MX. Histological changes of gastric mucosa after Helicobacter pylori eradication: a systematic review and meta-analysis. World J Gastroenterol. 2014;20(19):5903–11. https://doi.org/10.3748/wjg.v20.i19.5903.
Chan FK, Ching JY, Suen BY, Tse YK, Wu JC, Sung JJ. Effects of Helicobacter pylori infection on long-term risk of peptic ulcer bleeding in low-dose aspirin users. Gastroenterology. 2013;144(3):528–35.
Malfertheiner P, Megraud F, Rokkas T, Gisbert JP, Liou JM, Schulz C, et al. European Helicobacter and Microbiota Study group. Management of Helicobacter pylori infection: the Maastricht VI/Florence consensus report. Gut. 2022. https://doi.org/10.1136/gutjnl-2022-327745.
Tomita N, Mizokami F, Kisara S, Arai H. Transdisciplinary approach for potentially harmful drug-drug interaction detection as a part of a comprehensive medication review and geriatric assessment. Geriatr Gerontol Int. 2019;19(5):462–3.
Sheu BS, Wu MS, Chiu CT, Lo JC, Wu DC, Liou JM, et al. Consensus on the clinical management, screening-to-treat, and surveillance of Helicobacter pylori infection to improve gastric cancer control on a nationwide scale. Helicobacter. 2017;22(3): e12368. https://doi.org/10.1111/hel.12368.
Kao SS, Chen WC, Hsu PI, Lai KH, Yu HC, Cheng HH, et al. 7-day nonbismuth-containing concomitant therapy achieves a high eradication rate for Helicobacter pylori in Taiwan. Gastroenterol Res Pract. 2012. https://doi.org/10.1155/2012/463985.
Tai WC, Liang CM, Lee CH, Chiu CH, Hu ML, Lu LS, et al. Seven-day Nonbismuth containing quadruple therapy could achieve a grade “A” success rate for first-line Helicobacter pylori Eradication. Biomed Res Int. 2015. https://doi.org/10.1155/2015/623732.
Megraud FH. pylori antibiotic resistance: prevalence, importance, and advances in testing. Gut. 2004;53:1374–784.
Wu DC, Hsu PI, Wu JY, Opekun AR, Kuo CH, Wu IC, et al. Sequential and concomitant therapy with 4 drugs are equally effective for eradication of H. pylori Infection. Clin Gastroenterol Hepatol. 2010;8:36–41.
Chuah SK, Tai WC, Hsu PI, Wu DC, Wu KL, Kuo CM, et al. The efficacy of second-line anti-Helicobacter pylori therapy using an extended 14-day levofloxacin/amoxicillin/proton-pump inhibitor treatment–a pilot study. Helicobacter. 2012;17(5):374–81.
Hsu PI, Wu DC, Chen A, et al. Quadruple rescue therapy for Helicobacter pylori infection after two treatment failures. Eur J Clin Invest. 2008;38:404–9.
Hsu PI, Hwang IR, Cittelly D, Lai KH, El-Zimaity HM, Gutierrez O, et al. Clinical presentation in relation to diversity within the Helicobacter pylori cag pathogenicity island. Am J Gastroenterol. 2002;97:2231–8.
Gray SF, Wyatt JI, Rathbone BJ. Simplified techniques for identifying Campylobacter pyloridis. J Clin Pathol. 1986;39(11):1279.
Tai WC, Liang CM, Bi KW, Kuo CM, Lu LS, Wu CK, et al. A comparison between dexlansoprazole modified release-based and lansoprazole-based nonbismuth quadruple (concomitant) therapy for first-line Helicobacter pylori eradication: a prospective randomized trial. Infect Drug Resist. 2019;12:2923–31.
The European Committee on Antimicrobial Susceptibility Testing—EUCAST, https://www.eucast.org/clinical_breakpoints (EUCAST Clinical Breakpoint Tables v. 12.0, valid from 2022-01-01.
Lee BE, Kim JS, Kim BW, Kim JH, Kim JI, Chung JW, et al. Consistency of Helicobacter pylori eradication rates of first-line concomitant and sequential therapies in Korea: A nationwide multicenter retrospective study for the last 10 years. Helicobacter. 2021;26(2): e12780.
Kobayashi S, Joshita S, Yamamoto C, Yanagisawa T, Miyazawa T, Miyazawa M, et al. Efficacy and safety of eradication therapy for elderly patients with Helicobacter pylori infection. Medicine (Baltimore). 2019;98(30):e16619. https://doi.org/10.1097/MD.0000000000016619.
Boyanova L, Gergova G, Markovska R, Kandilarov N, Davidkov L, Spassova Z, et al. Primary Helicobacter pylori resistance in elderly patients over 20 years: a Bulgarian study. Diagn Microbiol Infect Dis. 2017;88(3):264–7. https://doi.org/10.1016/j.diagmicrobio.2017.05.001.
Lyu T, Cheung KS, Ni L, Guo J, Mu P, Li Y, et al. High prevalence and risk factors of multiple antibiotic resistance in patients who fail first-line Helicobacter pylori therapy in southern China: a municipality-wide, multicentre, prospective cohort study. J Antimicrob Chemother. 2020;75(11):3391–4.
Liang CM, Tai WC, Hsu PI, Wu DC, Kuo CH, Tsay FW, et al. Trend of changes in antibiotic resistance in Helicobacter pylori from 2013 to 2019: a multicentre report from Taiwan. Therap Adv Gastroenterol. 2020;10(13):1756284820976990. https://doi.org/10.1177/1756284820976990.
Chuah SK, Liang CM, Lee CH, Chiou SS, Chiu YC, Hu ML, et al. A randomized control trial comparing 2 levofloxacin-containing second-line therapies for Helicobacter pylori eradication. Medicine (Baltimore). 2016;95: e3586.
Fischbach L, Evans EL. Meta-analysis: the effect of antibiotic resistance status on the efficacy of triple and quadruple first-line therapies for Helicobacter pylori. Aliment Pharmacol Ther. 2007;26:343–57.
Moon JY, Kim GH, You HS, Lee BE, Ryu DY, Cheong JH, et al. Levofloxacin, metronidazole, and lansoprazole triple therapy compared to quadruple therapy as a second-line treatment of Helicobacter pylori Infection in Korea. Gut and liver. 2013;7(4):406–10.
Zou Y, Qian X, Liu X, Song Y, Song C, Wu S, et al. The effect of antibiotic resistance on Helicobacter pylori eradication efficacy: a systematic review and meta-analysis. Helicobacter. 2020;25(4): e12714.
Xu H, Wang W, Ma X, Feng R, Su Y, Cheng L, et al. Comparative efficacy and safety of high-dose dual therapy, bismuth-based quadruple therapy and non-bismuth quadruple therapies for Helicobacter pylori infection: a network meta-analysis. Eur J Gastroenterol Hepatol. 2021;33(6):775–86.
Tai WC, Liang CM, Kuo CM, Huang PY, Wu CK, Yang SC, et al. A 14 day esomeprazole- and amoxicillin-containing high-dose dual therapy regimen achieves a high eradication rate as first-line anti-Helicobacter pylori treatment in Taiwan: a prospective randomized trial. J Antimicrob Chemother. 2019;74(6):1718–24. https://doi.org/10.1093/jac/dkz046.
Durazzo M, Ferro A, Fagoonee S, Staiano MT, Saracco GM, Pellicano R. Helicobacter pylori eradication with a clarithromycin-based triple therapy in elderly patients. Panminerva Med. 2021;63(3):332–5. https://doi.org/10.23736/S0031-0808.21.04500-6.
Zullo A, Gatta L, De Francesco V, Hassan C, Ricci C, Bernabucci V, et al. High rate of Helicobacter pylori eradication with sequential therapy in elderly patients with peptic ulcer: a prospective controlled study. Aliment Pharmacol Ther. 2005;21(12):1419–24. https://doi.org/10.1111/j.1365-2036.2005.02519.x.
Moj D, Hanke N, Britz H, Frechen S, Kanacher T, Wendl T, et al. Clarithromycin, midazolam, and digoxin: application of PBPK modeling to gain new insights into drug-drug interactions and co-medication regimens. AAPS J. 2017;19(1):298–312. https://doi.org/10.1208/s12248-016-0009-9.
Huang Q, Jia X, Chu Y, Zhang X, Ye H. Helicobacter pylori infection in geriatric patients: current situation and treatment regimens. Front Med (Lausanne). 2021;30(8): 713908. https://doi.org/10.3389/fmed.2021.713908.
Goossens H, Glupczynski Y, Burette A, Van den Borre C, DePrez C, Bodenmann J, et al. Evaluation of a commercially available complement fixation test for diagnosis of Helicobacter pylori infection and for follow-up after antimicrobial therapy. J Clin Microbiol. 1992;30(12):3230–3. https://doi.org/10.1128/jcm.30.12.3230-3233.1992.
Perets TT, Gingold-Belfer R, Leibovitzh H, Itskoviz D, Schmilovitz-Weiss H, et al. Optimization of 13 C-urea breath test threshold levels for the detection of Helicobacter pylori infection in a national referral laboratory. J Clin Lab Anal. 2019;33(2): e22674. https://doi.org/10.1002/jcla.22674.
Kwon YH, Kim N, Yoon H, Shin CM, Park YS, Lee DH. Effect of citric acid on accuracy of 13C-urea breath test after Helicobacter pylori eradication therapy in a region with a high prevalence of atrophic gastritis. Gut Liver. 2019;13(5):506–14. https://doi.org/10.5009/gnl18398.
Uotani T, Graham DY. Diagnosis of Helicobacter pylori using the rapid urease test. Ann Transl Med. 2015;3(1):9. https://doi.org/10.3978/j.issn.2305-5839.2014.12.04.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
T-LM, W-CT, S-SL, C-CY, C-ML, S-KC declare that they have no commercial association, such as consultancies, stock ownership or other equity interests or patent-licensing arrangements.
Funding
No external funding was used in the conduct of this study or the preparation of this manuscript.
Author contributions
S-K Chuah and C-M Liang participated in the design of the study, coordinated the study, and performed the statistical analysis. T-L Ma wrote the manuscript. S-S Loke, C-C Yao, and W-C Tai contributed with administrative, technical, and/or material support. All authors have read and approved the manuscript.
Data availability statement
The data that support the findings of this study are available from the corresponding author on request. The data are not publicly available due to privacy or ethical restrictions.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Code availability
Not applicable.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ma, TL., Tai, WC., Loke, SS. et al. Efficacy and Safety of 7-Day Non-Bismuth Concomitant Quadruple Therapy for First-Line Helicobacter pylori Eradication in the Elderly. Drugs Aging 40, 71–79 (2023). https://doi.org/10.1007/s40266-022-00990-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40266-022-00990-7