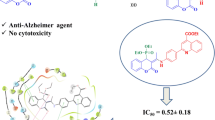
Abstract
A series of novel α-aminophosphonates bearing furan motif were designed and were synthesized under solvent-free condition and further evaluated for inhibition activity of acetylcholinesterase (AChE), butyrylcholinesterase (BuChE), and antioxidant property. All the synthesized compounds 4(a-h) showed good inhibition against AChE (IC50 = 0.88–4.46 µM) and BuChE (IC50 = 2.30–18.59 µM). Compound 4c was found to have maximum inhibitory potential against AChE with IC50 = 0.884 ± 0.002 µM while IC50 = 2.297 ± 0.091µM BuChE, suggesting that the synthesized compound are dual-inhibitors. Molecular docking studies suggested that the compound binds to both peripheral anionic site and catalytic anionic site which corroborate with the results obtained in kinetic inhibition suggesting a mixed-type inhibition. Predicted ADME showed good drug-likeness properties. Further, antioxidant activity using DPPH radical was performed; it was observed that all the compounds showed promising antioxidant activity. The highest antioxidant property was exhibited by compound 4c with IC50 value 30.04 when compared to ascorbic acid.
Graphical abstract







Similar content being viewed by others
References
S. Shaikh, P. Dhavan, P. Singh, J. Uparkar, S. P. Vaidya, B. L. Jadhav, and M. V. Ramana, J. Biomol. Struct. Dyn. 1–23 (2020)
P. Francis, A.M. Palmer, M. Snape, G.K. Wilcock, J. Neurol. Neurosurg. Psychiatry 66, 137 (1999)
G. Perry, A.D. Cash, M.A. Smith, J. Biomed. Biotechnol. 2, 120 (2002)
J. Nasica-Labouze, P.H. Nguyen, F. Sterpone, O. Berthoumieu, N.V. Buchete, S. Coté, A. De Simone, A.J. Doig, P. Faller, A. Garcia, A. Laio, M.S. Li, S. Melchionna, N. Mousseau, Y. Mu, A. Paravastu, S. Pasquali, D.J. Rosenman, B. Strodel, B. Tarus, J.H. Viles, T. Zhang, C. Wang, P. Derreumaux, Chem. Rev. 115, 3518 (2015)
L. Martin, X. Latypova, C.M. Wilson, A. Magnaudeix, M.L. Perrin, C. Yardin, F. Terro, Ageing Res. Rev. 12, 289 (2013)
F. Anwar, U. Saleem, B. Ahmad, M. Ashraf, A.U. Rehman, M. Froeyen, L.Y. Kee, I. Abdullah, M.U. Mirza, S. Ahmad, Comput. Biol. Chem. 89, 107378 (2020)
T.H. Ferreira-Vieira, I.M. Guimaraes, F.R. Silva, F.M. Ribeiro, Curr. Neuropharmacol. 14, 101 (2016)
J. Patocka, K. Kuca, D. Jun, Acta Medica (Hradec Kralove) 47, 215 (2004)
N. Kandiah, M.-C. Pai, V. Senanarong, I. Looi, E. Ampil, K. Karanam, A.K.W. Park, S. Christopher, Clin. Interv. Aging 12, 697 (2017)
J. Sussman, M. Hare, F. Frolow, C. Oefner, A. Goldman, L. Toker, I. Silman, Science 253, 872 (1991)
Y. Nicolet, O. Lockridge, P. Masson, J.C. Fontecilla-Camps, F. Nachon, J. Biol. Chem. 278, 41141 (2003)
R. Saini, A. Saxsena, Alzheimer’s Neurodegener. Dis. 4, 1 (2019)
Y. Pourshojaei, A. Abiri, K. Eskandari, Z. Haghighijoo, N. Edraki, A. Asadipour, Sci. Rep. 9, 1 (2019)
M. Bajda, A. Wiȩckowska, M. Hebda, N. Guzior, C.A. Sotriffer, B. Malawska, Int. J. Mol. Sci. 14, 5608 (2013)
S.A. Galal, A.S. AbdEl-All, M.M. Abdallah, H.I. El-Diwani, Bioorg. Med. Chem. Lett. 19, 2420 (2009)
P. Diana, A. Carbone, P. Barraja, G. Kelter, H.H. Fiebig, G. Cirrincione, Bioorg. Med. Chem. 18, 4524 (2010)
S. Malladi, R.V. Nadh, K.S. Babu, P.S. Babu, Beni-Suef Univ. J. Basic Appl. Sci. 6, 345 (2017)
S. Manfredini, S. Vertuani, B. Manfredi, G. Rossoni, G. Calviello, P. Palozza, Bioorg. Med. Chem. 8, 2791 (2000)
M.D. Delost, D.T. Smith, B.J. Anderson, J.T. Njardarson, J. Med. Chem. 61, 10996 (2018)
C.J.V. Simões, Z.L. Almeida, D. Costa, C.S.H. Jesus, A.L. Cardoso, M.R. Almeida, M.J. Saraiva, T.M.V.D. Pinho e Melo, R.M.M. Brito, Eur. J. Med. Chem. 121, 823 (2016)
D. Goyal, A. Kaur, B. Goyal, ChemMedChem 13, 1275 (2018)
B. Bulic, M. Pickhardt, E.M. Mandelkow, E. Mandelkow, Neuropharmacology 59, 276 (2010)
A. Kumar, F. Pintus, A. Di Petrillo, R. Medda, P. Caria, M.J. Matos, D. Viña, E. Pieroni, F. Delogu, B. Era, G.L. Delogu, A. Fais, Sci. Rep. 8, 4424 (2018)
B.S. Kumar, A.U.R. Sankar, C.S. Reddy, S.K. Nayak, C.N. Raju, ARKIVOC 2007, 155 (2007)
Z. Rezaei, S. Khabnadideh, K. Zomorodian, K. Pakshir, S. Nadali, N. Mohtashami, E. Faghih Mirzaei, Int. J. Med. Chem. 2011, 1 (2011)
R.R. Chinthaparthi, I. Bhatnagar, C.S.R. Gangireddy, S.C. Syama, S.R. Cirandur, Arch. Pharm. (Weinheim). 346, 667 (2013)
Y. Xu, K. Yan, B. Song, G. Xu, S. Yang, W. Xue, D. Hu, P. Lu, G. Ouyang, L. **, Z. Chen, Molecules 11, 666 (2006)
S. Shaikh, P. Dhavan, M. M. V. Ramana, and B. L. Jadhav, Mol. Divers. (2020)
S. Shaikh, G. Pavale, M.M.V. Ramana, Chem. Pap. 74, 2555 (2020)
S. Shaikh, P. Dhavan, G. Pavale, M.M.V. Ramana, B.L. Jadhav, Bioorg. Chem. 96, 103589 (2020)
S. Shaikh, P. Dhavan, P. Singh, J. Uparkar, S. P. Vaidya, B. L. Jadhav, and M. V. Ramana, J. Biomol. Struct. Dyn. (2021)
G.L. Ellman, K.D. Courtney, V. Andres, R.M. Featherstone, Biochem. Pharmacol. 7, 88 (1961)
A. Joshi, M. Gadhwal, U. Joshi, Med. Chem. Res. 23, 1414 (2014)
R. Prentis, Y. Lis, S. Walker, Br. J. Clin. Pharmacol. 25, 387 (1988)
P. Matsson, B.C. Doak, B. Over, J. Kihlberg, Adv. Drug Deliv. Rev. 101, 42 (2016)
J. Wang, T. Hou, Comb. Chem. High Throughput Screen. 14, 328 (2011)
Acknowledgements
We are thankful to Microanalytical Laboratory, University of Mumbai for Characterization facilities. We also thank Dr. Urmila Joshi and Miss Heena Bhojwani (K. M. Kundnani College of Pharmacy, Mumbai) for computer-aided drug design.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declared that there are no conflicts of interest.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Uparkar, J.J., Dhavan, P.P., Jadhav, B.L. et al. Design, synthesis and biological evaluation of furan based α-aminophosphonate derivatives as anti-Alzheimer agent. J IRAN CHEM SOC 19, 3103–3116 (2022). https://doi.org/10.1007/s13738-022-02515-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-022-02515-w