Abstract
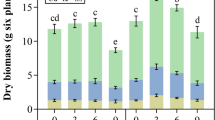
Pennisetum sp. was employed as a model species to detect the growth and physiological response to cadmium (Cd) stress at different Cd concentrations (0, 20, 50, and 100 mg kg−1) in three types of soils (yellow brown soil, yellow soil, and red soil). Results showed that the growth of Pennisetum sp. was not significantly influenced by Cd in 20 mg kg−1, but significantly inhibited at higher Cd concentrations in three types of soils. Besides, the higher Cd concentrations, the lower root, stem, and leaf biomass. With Cd concentration of soil increasing, Cd content of root, stem, and leaf increased. Compared with no Cd, high Cd concentrations (50 and 100 mg kg−1) induced the physiological indices (photosynthetic rate, stomatal conductance, transpiration rate) and biochemical indices (nitrate reductase, glutamine synthetase, and glutamate synthase activities) decreasing, but the concentration of NO3− and NH4+ increasing. The activity of antioxidative enzymes (SOD, POD, and CAT) was disrupted and the content of malondialdehyde (MDA) increasing. Pennisetum sp. could protect cells from damage and maintain normal physiological metabolism via increasing the production of soluble sugar and soluble protein, but soluble proteins and soluble sugars were limited in high concentrations of Cd (50 and 100 mg kg−1). Moreover, the growth and physiological response to Cd are different in the three types of soils. The growth of Pennisetum sp. in yellow brown soil was better than that in other two soils, and the gas exchange rate, antioxidant enzyme activity, and nitrogen metabolism in yellow soil and red soil were more affected by Cd stress than that in yellow brown soil. Overall, Pennisetum sp. had certain tolerance and biosorption ability to Cd in different Cd concentrations and different types of soil. Hence, Pennisetum sp. was a suitable choice for Cd remediation, especially in yellow brown soil.








Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Arriagada CA, Herrera MA, Ocampo JA (2005) Contribution of arbuscular mycorrhizal and saprobe fungi to the tolerance of eucalyptus globulus to Pb. Water Air Soil Pollut 166:31–47. https://doi.org/10.1007/s11270-005-7711-z
Azizollahi Z, Ghaderian SM, Ghotbi-Ravandi AA (2019) Cadmium accumulation and its effects on physiological and biochemical characters of summer savory (Satureja hortensis L.). Int J Phytoremediation 21:1241–1253. https://doi.org/10.1080/15226514.2019.1619163
Balestrasse KB, Benavides MP, Gallego SM, Tomaro ML (2003) Effect of cadmium stress on nitrogen metabolism in nodules and roots of soybean plants. Funct Plant Biol 30:57–64. https://doi.org/10.1071/FP02074
Bartolf M, Brennan E, Price CA (1980) Partial characterization of a cadmium-binding protein from the roots of cadmium-treated Tomato. Plant Physiol 66:438–441. https://doi.org/10.1104/pp.66.3.438
Baryla A, Carrier P, Franck F, Coulomb C, Sahut C, Havaux M (2001) Leaf chlorosis in oilseed rape plants (Brassica napus) grown on cadmium-polluted soil: causes and consequences for photosynthesis and growth. Planta 212:696–709. https://doi.org/10.1007/s004250000439
Bhaduri AM, Fulekar MH (2012) Antioxidant enzyme responses of plants to heavy metal stress. Environmental Science and Bio-Technology 11:55–69. https://doi.org/10.1007/s11157-011-9251-x
Boussama N, Ouariti O, Suzuki A, Ghorbal MH (1999) Cd-stress on nitrogen assimilation. J Plant Physiol 2:150–155. https://doi.org/10.1016/S0176-1617(99)80110-2
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Bräutigam A, Gagneul D, Weber APM (2006) High-throughput colorimetric method for the parallel assay of glyoxylic acid and ammonium in a single extract. Anal Biochem 362:151–153. https://doi.org/10.1016/j.ab.2006.12.033
Brown SL, Angle JS, Chaney RL (1995) Zinc and cadmium uptake by hyperaccumulator Thlaspi caerulescens grown in nutrient solution. Soil Sci Soc Am J 1:125–133. https://doi.org/10.2136/sssaj1995.03615995005900010020x
Chen L, Han Y, Jiang H, Korpelainen H, Li C (2011) Nitrogen nutrient status induces sexual differences in responses to cadmium in Populus yunnanensis. J Exp Bot 62:5037–5050. https://doi.org/10.1093/jxb/err203
Chen L, Zhang D, Yang W, Liu Y, Zhang L, Gao S (2016) Sex-specific responses of Populus deltoides to Glomus intraradices colonization and Cd pollution. Chemosphere 155:196–206. https://doi.org/10.1016/j.chemosphere.2016.04.049
Cramer MD, Savidov NA, Lips SH (1996) The influence of enriched rhizosphere CO2 on N uptake and metabolism in wild-type and NR-deficient barley plants. Physiol Plant 97:47–54. https://doi.org/10.1034/j.1399-3054.1996.970108.x
Devriese M, Tsakaloudi V, Garbayo I, León R, Vílchez C, Vigara J (2001) Effect of heavy metals on nitrate assimilation in the eukaryotic microalga Chlamydomonas reinhardtii. Plant Physiol Biochem 39:443–448. https://doi.org/10.1016/s0981-9428(01)01257-8
Fang G, Ittersum MKV, Couëdel A, Yue Z, Werf WVD (2018) Intercrop** with wheat lowers nutrient uptake and biomass accumulation of maize, but increases photosynthetic rate of the ear leaf. Aob Plants 10:134–139. https://doi.org/10.1093/aobpla/ply010
Farquhar GD, Sharkey TD (1982) Stomatal conductance and photosynthesis. Annu Rev Plant Physiol 1:317–345
Fernández R, Bertrand A, Reis R, Mourato MP, Martins LL, González A (2013) Growth and physiological responses to cadmium stress of two populations of Dittrichia viscosa (L.) Greuter. J Hazard Mater 244-245:555–562. https://doi.org/10.1016/j.jhazmat.2012.10.044
Figueira MM, Volesky B, Ciminelli VST, Roddick FA (2000) Biosorption of metals in brown seaweed biomass. Water Res 34:196–204. https://doi.org/10.1016/s0043-1354(99)00120-7
Gallego SM, Pena LB, Barcia RA, Azpilicueta CE, Iannone MF, Rosales EP, Zawoznik MS, Groppa MD, Benavides MP (2012) Unravelling cadmium toxicity and tolerance in plants: Insight into regulatory mechanisms. Environ Exp Bot 83:33–46. https://doi.org/10.1016/j.envexpbot.2012.04.006
Gao ML, Qi Y, Song WH (2016) Effects of di-n-butyl phthalate and di (2-ethylhexyl) phthalate on the growth, photosynthesis, and chlorophyll fluorescence of wheat seedlings. Chemosphere 151:76–83. https://doi.org/10.1016/j.chemosphere.2016.02.061
Giannopolitis CN, Ries SK (1977) Superoxide dismutases: I. Occurrence in higher plants. Plant Physiol 59:309–314. https://doi.org/10.2307/4264724
Göbel C, Feussner I (2009) Methods for the analysis of oxylipins in plants. Phytochemistry 70:1485–1503. https://doi.org/10.1016/j.phytochem.2009.07.040
Gouia H, Suzuki A, Brulfert J, Ghorbal MH (2003) Effects of cadmium on the co-ordination of nitrogen and carbon metabolism in bean seedlings. J Plant Physiol 160:367–376. https://doi.org/10.1078/0176-1617-00785
Greef VMWA, Kemp B, Togt (1994) The effect of body weight and energy intake on the composition of deposited tissue in pigs. Anim Sci 58:234–239. https://doi.org/10.1017/S1357729800042582
Hodges DM, DeLong JM, Forney CF, Prange RK (1999) Improving the thiobarbituric acid-reactive-substances assay for estimating lipid peroxidation in plant tissues containing anthocyanin and other interfering compounds. Planta 207:604–611. https://doi.org/10.2307/23385611
Hou L, Huang R, Zhou L, Chen W, Liu C (2010) The accumulation of Cd in Huttuynia cordata and its promotion by rhizospheric microbes. Ecology and Environmental Sciences 6:641–646. https://doi.org/10.1016/S1872-5813(11)60001-7
Kalinowska R, Pawlik-Skowronska B (2010) Response of two terrestrial green microalgae (Chlorophyta, Trebouxiophyceae) isolated from Cu-rich and unpolluted soils to copper stress. Environ Pollut 158:2778–2785. https://doi.org/10.1016/j.envpol.2010.03.003
Karalija E, Selović A (2018) The effect of hydro and proline seed priming on growth, proline and sugar content, and antioxidant activity of maize under cadmium stress. Environ Sci Pollut Res 25:33370–33380. https://doi.org/10.1007/s11356-018-3220-7
Khan ZS, Rizwan M, Hafeez M, Ali S, Adrees M, Qayyum MF, Khalid S, Ur Rehman MZ, Sarwar MA (2020) Effects of silicon nanoparticles on growth and physiology of wheat in cadmium contaminated soil under different soil moisture levels. Environ Sci Pollut Res 27:4958–4968. https://doi.org/10.1007/s11356-019-06673-y
Kieffer P, Schröder P, Dommes J, Hoffmann L, Renaut J, Hausman J (2009) Proteomic and enzymatic response of poplar to cadmium stress. J Proteome 72:379–396. https://doi.org/10.1016/j.jprot.2009.01.014
Kültz D (2005) Molecular and evolutionary basis of the cellular stress response. Annu Rev Physiol 67:225–257. https://doi.org/10.1146/annurev.physiol.67.040403.103635
Kwinta J, Kolik D (2006) Glutamine synthetase and glutamate dehydrogenase in cadmium-stressed triticale seedlings. Acta Physiol Plant 28:339–347. https://doi.org/10.1007/s11738-006-0030-1
Li F, Qi JM, Zhou GY, Liu LH (2013) Effect of cadmium stress on the growth, antioxidative enzymes and lipid peroxidation in two kenaf (Hibiscus cannabinus L.) plant seedlings. J Integr Agric 12:610–620. https://doi.org/10.1016/S2095-3119(13)60279-8
Li S, Yang W, Yang T, Chen Y, Ni W (2015) Effects of cadmium stress on leaf chlorophyll fluorescence and photosynthesis of Elsholtzia argyi-A cadmium accumulating plant. Int J Phytoremediat 17:85–92. https://doi.org/10.1080/15226514.2013.828020
Li W, Chen Z, Lin J, Aihua X, Jie D, Luyi M (2018) A novel gene of Kalanchoe daigremontiana confers plant drought resistance. Sci Rep-Uk 8:2547. https://doi.org/10.1038/s41598-018-20687-5
Liang WJ, Hai Q, Fu J (2006) Research advance of biomass and carbon storage of poplar in China. J Forestry Res 17:75–79
Lin X, Liu ZX, Liu DM (2012) Effects of different years of planting Pennisetum sp. on the plant and insect diversity in Pennisetum sp. communities. Chin J Appl Ecol 10:2849–2854
Liu Z, He X, Chen W, Yuan F, Yan K, Tao D (2009) Accumulation and tolerance characteristics of cadmium in a potential hyperaccumulator—Lonicera japonica Thunb. J Hazard Mater 169:170–175. https://doi.org/10.1016/j.jhazmat.2009.03.090
Margoshes M, Vallee BL (1979) A cadmium protein from equine kidney cortex. J Am Chem Soc 17:4813–4814. https://doi.org/10.1021/ja01574a064
Martins LL, Mourato MP, Cardoso AI, Pinto AP, Mota AM, Gonçalves M d LS, de Varennes A (2011) Oxidative stress induced by cadmium in Nicotiana tabacum L.: effects on growth parameters, oxidative damage and antioxidant responses in different plant parts. Acta Physiol Plant 33:1375–1383. https://doi.org/10.1007/s11738-010-0671-y
McBride MB (2002) Cadmium uptake by crops estimated from soil total Cd and pH. Soil Sci 167:62–67. https://doi.org/10.1097/00010694-200201000-00006
Meng D, Zhao Y, Lei C, Zhou X (2013) Physiological mechanism of Cd-tolerance of Artemisia Selengensis grown in Dongting Lake Wetland. Asian Journal of Ecotoxicology
Miflin BJ, Habash DZ (2002) The role of glutamine synthetase and glutamate dehydrogenase in nitrogen assimilation and possibilities for improvement in the nitrogen utilization of crops. J Exp Bot 1:370. https://doi.org/10.1093/jexbot/53.370.979
Mishra P, Dubey RS (2011) Nickel and Al-excess inhibit nitrate reductase but upregulate activities of aminating glutamate dehydrogenase and aminotransferases in growing rice seedlings. Plant Growth Regul 64:251–261. https://doi.org/10.1007/s10725-011-9566-1
Nayyar H, Walia DP (2003) Water stress induced proline accumulation in contrasting wheat genotypes as affected by calcium and abscisic acid. Biol Plant 46:275–279. https://doi.org/10.1023/A:1022867030790
Oral O, Kutlu T, Aksoy E, Fıçıcıoğlu C, Uslu H, Tuğrul S (2006) The effects of oxidative stress on outcomes of assisted reproductive techniques. J Assist Reprod Genet 23:81–85. https://doi.org/10.1007/s10815-005-9010-4
Patterson K, Cakmak T, Cooper A, Lager I, Rasmusson AG, Escobar MA (2010) Distinct signalling pathways and transcriptome response signatures differentiate ammonium- and nitrate-supplied plants. Plant Cell Environ 33. https://doi.org/10.1111/j.1365-3040.2010.02158.x
Salt DE, Blaylock M, Kumar NPBA, Dushenkov V, Ensley BD, Chet I, Raskin I (1995) Phytoremediation: a novel strategy for the removal of toxic metals from the environment using plants. Biotechnology 13:468–474. https://doi.org/10.1038/nbt0595-468
Sánchez-Rodríguez E, Rubio-Wilhelmi MDM, Ríos JJ, Blasco B, Rosales MÁ, Melgarejo R, Romero L, Ruiz JM (2010) Ammonia production and assimilation: Its importance as a tolerance mechanism during moderate water deficit in tomato plants. J Plant Physiol 168:816–823. https://doi.org/10.1016/j.jplph.2010.11.018
Saoussen B, Helmi H, Shino M, Yoshiro O (2010) Response of antioxidative enzymes and apoplastic bypass transport in Thlaspi caerulescens and Raphanus sativus to cadmium stress. Int J Phytoremediat 12:733–744. https://doi.org/10.1080/15226514.2010.483262
Seregin IV , Shpigun LK , Ivanov VB (2004) Distribution and Toxic Effects of Cadmium and Lead on Maize Roots. Russ J Plant Physiol 51(4):525–533. https://doi.org/10.1023/B:RUPP.0000035747.42399.84
Shafi M, Bakht J, Razuddin HY, Zhang GP (2011) Genotypic difference in the inhibition of photosynthesis and chlorophyll fluorescence by salinity and cadmium stresses in wheat. J Plant Nutr 34:315–323. https://doi.org/10.1080/01904167.2011.536874
Tang H, Li T, Yu H, Zhang X (2016) Cadmium accumulation characteristics and removal potentials of high cadmium accumulating rice line grown in cadmium-contaminated soils. Environ Sci Pollut Res Int 23:15351–15357
Wang X, Zheng X, Ding Y, Zheng S, Shi R (2016) Absorption rules and threshold value of amaranth cadmium under different soil types. Environ Sci Technol
Xu ZZ, Zhou GS (2006) Nitrogen metabolism and photosynthesis in Leymus chinensisin response to long-term soil drought. J Plant Growth Regul 25:252–266. https://doi.org/10.1007/s00344-006-0043-4
Xu L, **ng X, Liang J, Peng J, Zhou J (2019) In situ phytoremediation of copper and cadmium in a co-contaminated soil and its biological and physical effects. RSC Adv 21:275–279. https://doi.org/10.1039/C8RA07645F
Yemm EW, Willis AJ (1954) The estimation of carbohydrates in plant extracts by anthrone. Biochem J 57:508–514. https://doi.org/10.1042/bj0570508
Yi T, **ao YD, Sun GC (2018) Cadmium-accumulator straw application alleviates cadmium stress of lettuce (Lactuca sativa) by promoting photosynthetic activity and antioxidative enzyme activities. Environ Sci Pollut Res 25:30671–30679. https://doi.org/10.1007/s11356-018-2822-4
Yin G, Lulin B, **aowang S, Haoyu L (2019) Adsorption of Cd(II) from aqueous solution by Pennisetum sp. straw biochars derived from different modification methods. Environ Sci Pollut Res Int 35:421–426
Zhang F, Wang Y, Lou Z, Dong J (2006) Effect of heavy metal stress on antioxidative enzymes and lipid peroxidation in leaves and roots of two mangrove plant seedlings (Kandelia candel and Bruguiera gymnorrhiza). Chemosphere 67:44–50. https://doi.org/10.1016/j.chemosphere.2006.10.007
Zhang M, ** Z, Zhao J, Zhang G, Wu F (2015) Physiological and biochemical responses to drought stress in cultivated and Tibetan wild barley. Plant Growth Regul 75:567–574. https://doi.org/10.1007/s10725-014-0022-x
Zhou C, Zhang K, Lin J, Li Y (2015) Physiological responses and tolerance mechanisms to cadmium in conyza canadensis. Int J Phytoremediat 17:280–289. https://doi.org/10.1080/15226514.2014.898021
Funding
National Natural Science Foundation of China (CN) (31300513).
Author information
Authors and Affiliations
Contributions
Senlin Yang conducted the experiments to obtain the analytical data and contributed to the manuscript. Jian Zhang modified some experimental methods and helped to collect experimental data. Lianghua Chen interpreted the results, and was a major contributor in writing the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Responsible Editor: Gangrong Shi
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yang, S., Zhang, J. & Chen, L. Growth and physiological responses of Pennisetum sp. to cadmium stress under three different soils. Environ Sci Pollut Res 28, 14867–14881 (2021). https://doi.org/10.1007/s11356-020-11701-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-11701-3