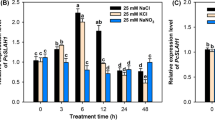
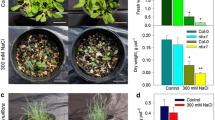
Abstract
Background and aims
Uncontrolled uptake of Na+ is the reason that many species are sensitive to salinity. Suberin is a protective barrier found in the walls of root endodermal cells that appears to be important for salt tolerance, yet its specific protective mechanism has not been fully elucidated.
Methods
Here we investigated the role of aliphatic suberin in protecting plants against salt stress by using a mutant of Arabidopsis, cyp86a1, which exhibits a significant reduction of root aliphatic suberin.
Results
We found that NaCl significantly increased suberization in roots of hydroponic-grown wild-type plants, but not in cyp86a1. Cyp86a1 exhibited a salt-sensitive phenotype. Compared with wild-type, Na+ accumulation in shoots was higher in cyp86a1. We provide evidence that increased Na+ uptake was via the root transcellular pathway. Furthermore, cyp86a1 accumulated less K+ in shoots than wild-type under NaCl stress, which was a consequence of increased K+ efflux from the root vasculature. Additionally, we provide evidence that aliphatic suberin reduces inflow of water across the root endodermis under non-stress conditions but reduces the backflow of water to the medium under salt stress.
Conclusions
Finally, we propose a model for the role of aliphatic suberin in restricting Na+ influx, K+ efflux and water backflow in plants under saline conditions.







Similar content being viewed by others
Abbreviations
- CTM:
-
Composite transport model
- GC-MS:
-
Gas chromatography-mass spectrometer
- Gs:
-
Stomatal conductance
- ICP-OES:
-
Inductively coupled plasma - optical emission spectrometer
- Jv r :
-
The volume flow of xylem sap per unit root surface area
- Lp r :
-
Root hydraulic conductivity
- NMT:
-
Non-invasive micro-test technology
- Pn:
-
Net photosynthesis rate
- PTS:
-
Trisodium-8-hydroxy-1,3,6-pyrenetrisulphonic acid
- SA:
-
Selective absorption
- ST:
-
Selective transport
- Tr:
-
Transpiration rate
- WUE:
-
Water use efficiency
References
Alassimone J, Naseer S, Geldner N (2010) A developmental framework for endodermal differentiation and polarity. Proc Natl Acad Sci U S A 107:5214–5219. https://doi.org/10.1073/pnas.0910772107
Andersen TG, Barberon M, Geldner N (2015) Suberization - the second life of an endodermal cell. Curr Opin Plant Biol 28:9–15. https://doi.org/10.1016/j.pbi.2015.08.004
Bajji M, Kinet JM, Lutts S (2002) The use of the electrolyte leakage method for assessing cell membrane stability as a water stress tolerance test in durum wheat. Plant Growth Regul 36:61–70. https://doi.org/10.1023/a:1014732714549
Barberon M, Geldner N (2014) Radial transport of nutrients: the plant root as a polarized epithelium. Plant Physiol 166:528–537. https://doi.org/10.1104/pp.114.246124
Barberon M, Vermeer JEM, Bellis DD, Wang P, Naseer S, Andersen TG, Humbel BM, Nawrath C, Takano J, Salt DE, Geldner N (2016) Adaptation of root function by nutrient-induced plasticity of endodermal differentiation. Cell 164:447–459. https://doi.org/10.1016/j.cell.2015.12.021
Barrowclough DE, Peterson CA, Steudle E (2000) Radial hydraulic conductivity along develo** onion roots. J Exp Bot 51:547–557. https://doi.org/10.1093/jexbot/51.344.547
Baxter I, Hosmani PS, Rus A, Lahner B, Borevitz JO, Muthukumar B, Mickelbart MV, Schreiber L, Franke RB, Salt DE (2009) Root suberin forms an extracellular barrier that affects water relations and mineral nutrition in Arabidopsis. PLoS Genet 5:e1000492. https://doi.org/10.1371/journal.pgen.1000492
Beisson F, Li-Beisson Y, Bonaventure G, Pollard M, Ohlrogge JB (2007) The acyltransferase GPAT5 is required for the synthesis of suberin in the seed coat and root of Arabidopsis. Plant Cell 19:351–368. https://doi.org/10.1105/tpc.106.048033
Blumwald E (2000) Sodium transport and salt tolerance in plants. Curr Opin Cell Biol 12:431–434. https://doi.org/10.1016/S0955-0674(00)00112-5
Britto DT, Ebrahimi-Ardebili S, Hamam AM, Coskun D, Kronzucker HJ (2010) 42K analysis of sodium-induced potassium efflux in barley: mechanism and relevance to salt tolerance. New Phytol 186:373–384. https://doi.org/10.1111/j.1469-8137.2009.03169.x
Buschmann PH, Vaidyanathan R, Gassmann W, Schroeder JI (2000) Enhancement of Na+ uptake currents, time-dependent inward rectifying K+ channel currents, and K+ channel transcripts by K+ starvation in wheat root cells. Plant Physiol 122:1387–1397. https://doi.org/10.1104/pp.122.4.1387
Byrt CS, Munns R, Burton RA, Gilliham M, Wege S (2018) Root cell wall solutions for crop plants in saline soils. Plant Sci 269:47–55. https://doi.org/10.1016/j.plantsci.2017.12.012
Chen Y, Cao C, Guo Z, Zhang Q, Li S, Zhang X, Gong J, Shen Y (2020) Herbivore exposure alters ion fluxes and improves salt tolerance in a desert shrub. Plant Cell Environ 43:400–419. https://doi.org/10.1111/pce.13662
Coskun D, Britto DT, Kronzucker HJ (2010) Regulation and mechanism of potassium release from barley roots: an in planta 42K analysis. New Phytol 188:1028–1038. https://doi.org/10.2307/40960856
Coskun D, Britto DT, Jean Y-K, Kabir I, Tolay I, Torun AA, Kronzucker HJ (2013) K+ efflux and retention in response to NaCl stress do not predict salt tolerance in contrasting genotypes of rice (Oryza sativa L.). PLoS One 8:e57767. https://doi.org/10.1371/journal.pone.0057767
Delude C, Fouillen L, Bhar P, Cardinal MJ, Pascal S, Santos P, Kosma DK, Joubès J, Rowland O, Domergue F (2016) Primary fatty alcohols are major components of suberized root tissues of Arabidopsis in the form of alkyl hydroxycinnamates. Plant Physiol 171:1934–1950. https://doi.org/10.1104/pp.16.00834
Ding L, Zhu JK (1997) Reduced Na+ uptake in the NaCl-hypersensitive sos1 mutant of Arabidopsis thaliana. Plant Physiol 113:795–799. https://doi.org/10.2307/4277593
Doblas VG, Geldner N, Barberon M (2017) The endodermis, a tightly controlled barrier for nutrients. Curr Opin Plant Biol 39:136–143. https://doi.org/10.1016/j.pbi.2017.06.010
Domergue F, Vishwanath SJ, Joubès J, Ono J, Lee J, Bourdon M, Alhattab R, Lowe C, Pascal S, Lessire R, Rowland O (2010) Three Arabidopsis fatty acyl-CoA reductases, FAR1, FAR4, and FAR5, generate primary fatty alcohols associated with suberin deposition. Plant Physiol 153:1539–1554. https://doi.org/10.1104/pp.110.158238
Enstone DE, Peterson CA, Ma F (2002) Root endodermis and exodermis: structure, function, and responses to the environment. J Plant Growth Regul 21:335–351. https://doi.org/10.1007/s00344-003-0002-2
Faiyue B, Al-Azzawi MJ, Flowers TJ (2010) The role of lateral roots in bypass flow in rice (Oryza sativa L.). Plant Cell Environ 33:702–716. https://doi.org/10.1111/j.1365-3040.2009.02078.x
Flowers TJ (2004) Improving crop salt tolerance. J Exp Bot 55:307–319. https://doi.org/10.1093/jxb/erh003
Flowers TJ, Munns R, Colmer TD (2015) Sodium chloride toxicity and the cellular basis of salt tolerance in halophytes. Ann Bot 115:419–431. https://doi.org/10.1093/aob/mcu217
Franke R, Schreiber L (2007) Suberin—a biopolyester forming apoplastic plant interfaces. Curr Opin Plant Biol 10:252–259. https://doi.org/10.1016/j.pbi.2007.04.004
Franke R, Briesen I, Wojciechowski T, Faust A, Yephremov A, Nawrath C, Schreiber L (2005) Apoplastic polyesters in Arabidopsis surface tissues - a typical suberin and a particular cutin. Phytochemistry 66:2643–2658. https://doi.org/10.1016/j.phytochem.2005.09.027
Franke R, Hofer R, Briesen I, Emsermann M, Efremova N, Yephremov A, Schreiber L (2009) The DAISY gene from Arabidopsis encodes a fatty acid elongase condensing enzyme involved in the biosynthesis of aliphatic suberin in roots and the chalaza-micropyle region of seeds. Plant J 57:80–95. https://doi.org/10.1111/j.1365-313X.2008.03674.x
Geldner N (2013) The endodermis. Annu Rev Plant Biol 64:631–558. https://doi.org/10.1146/annurev-arplant-050312-120050
Gierth M, Mäser P (2007) Potassium transporters in plants involvement in K+ acquisition, redistribution and homeostasis. FEBS Lett 581:2348–2356. https://doi.org/10.1016/j.febslet.2007.03.035
Guo Q, Wang P, Ma Q, Zhang JL, Bao AK, Wang SM (2012) Selective transport capacity for K+ over Na+ is linked to the expression levels of PtSOS1 in halophyte Puccinellia tenuiflora. Funct Plant Biol 39:1047–1057. https://doi.org/10.1071/FP12174
Hasegawa PM (2013) Sodium (Na+) homeostasis and salt tolerance of plants. Environ Exp Bot 92:19–31. https://doi.org/10.1016/j.envexpbot.2013.03.001
Hasegawa PM, Bressan RA, Zhu JK, Bohnert HJ (2000) Plant cellular and molecular responses to high salinity. Annu Rev Plant Physiol Plant Mol Biol 1:463–499. https://doi.org/10.1146/annurev.arplant.51.1.463
Höfer R, Briesen I, Beck M, Pinot F, Schreiber L, Franke R (2008) The Arabidopsis cytochrome P450 CYP86A1 encodes a fatty acid omega-hydroxylase involved in suberin monomer biosynthesis. J Exp Bot 59:2347–2360. https://doi.org/10.1093/jxb/ern101
Jenkin S, Molina I (2015) Isolation and compositional analysis of plant cuticle lipid polyester monomers. JOVE-J Vis Exp 105:e53386. https://doi.org/10.3791/53386
Kamiya T, Borghi M, Wang P, Danku JMC, Kalmbach L, Hosmani PS, Naseer S, Fujiwara T, Geldner N, Salt DE (2015) The MYB36 transcription factor orchestrates Casparian strip formation. Proc Natl Acad Sci U S A 112:10533–10538. https://doi.org/10.1073/pnas.1507691112
Kim YX, Ranathunge K, Lee S, Lee Y, Lee D, Sung J (2018) Composite transport model and water and solute transport across plant roots: an update. Front Plant Sci 9:193. https://doi.org/10.3389/fpls.2018.00193
Kolattukudy PE (1981) Structure, biosynthesis and biodegradation of cutin and suberin. Annu Rev Plant Physiol 32:539–567. https://doi.org/10.1146/annurev.pp.32.060181.002543
Kreszies T, Schreiber L, Ranathunge K (2018) Suberized transport barriers in Arabidopsis, barley and rice roots: from the model plant to crop species. J Plant Physiol 227:75–83. https://doi.org/10.1016/j.jplph.2018.02.002
Krishnamurthy P, Ranathunge K, Franke R, Prakash HS, Schreiber L, Mathew MK (2009) The role of root apoplastic transport barriers in salt tolerance of rice (Oryza sativa L.). Planta 230:119–134. https://doi.org/10.2307/23390598
Krishnamurthy P, Ranathunge K, Nayak S, Schreiber L, Mathew MK (2011) Root apoplastic barriers block Na+ transport to shoots in rice (Oryza sativa L.). J Exp Bot 62:4215–4228. https://doi.org/10.1093/jxb/err135
Krishnamurthy P, Jyothi-Prakash PA, Qin L, He J, Lin Q, Loh CS, Kumar PP (2014) Role of root hydrophobic barriers in salt exclusion of a mangrove plant Avicennia officinalis. Plant Cell Environ 37:1656–1671. https://doi.org/10.1111/pce.12272
Lagarde D, Basset M, Lepetit M, Conejero G, Gaymard F, Astruc S, Grignon C (1996) Tissue-specific expression of Arabidopsis AKT1 gene is consistent with a role in K+ nutrition. Plant J 9:195–203. https://doi.org/10.1046/j.1365-313X.1996.09020195.x
Lee SH, Singh AP, Chung GC, Ahn SJ, Noh EK, Steudle E (2004) Exposure of roots of cucumber (Cucumis sativus) to low temperature severely reduces root pressure, hydraulic conductivity and active transport of nutrients. Physiol Plant 120:413–420. https://doi.org/10.1111/j.0031-9317.2004.00248.x
Li Y, Beisson F, Koo AJ, Molina I, Pollard M, Ohlrogge J (2007) Identification of acyltransferases required for cutin biosynthesis and production of cutin with suberin-like monomers. Proc Natl Acad Sci U S A 104:18339–18344. https://doi.org/10.1073/pnas.0706984104
Li B, Kamiya T, Kalmbach L, Yamagami M, Yamaguchi K, Shigenobu S, Sawa S, Danku JMC, Salt DE, Geldner N, Fujiwara T (2017) Role of LOTR1 in nutrient transport through organization of spatial distribution of root endodermal barriers. Curr Biol 27:758–765. https://doi.org/10.1016/j.cub.2017.01.030
Liu Y, Yu Y, Sun J, Cao Q, Tang Z, Liu M, Xu T, Ma D, Li Z, Sun J (2019) Root-zone-specific sensitivity of K+ and Ca2+ permeable channels to H2O2 determines ion homeostasis in salinized diploid and hexaploid Ipomoea trifida. J Exp Bot 70:1389–1405. https://doi.org/10.1093/jxb/ery461
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25:402–408. https://doi.org/10.1006/meth.2001
Ma Q, Hu J, Zhou XR, Yuan HJ, Kumar T, Luan S, Wang SM (2017) ZxAKT1 is essential for K+ uptake and K+/Na+ homeostasis in the succulent xerophyte Zygophyllum xanthoxylum. Plant J 90:48–60. https://doi.org/10.1111/tpj.13465
Martinka M, Dolan L, Pernas M, Abe J, Lux A (2012) Endodermal cell-cell contact is required for the spatial control of Casparian band development in Arabidopsis thaliana. Ann Bot 110:361–371. https://doi.org/10.1093/aob/mcs110
McLamore ES, Porterfield DM (2011) Non-invasive tools for measuring metabolism and biophysical analyte transport: self-referencing physiological sensing. Chem Soc Rev 40:5308–5320. https://doi.org/10.1039/c0cs00173b
Meyer CJ, Peterson CA, Steudle E (2011) Permeability of Iris germanica’s multiseriate exodermis to water, NaCl, and ethanol. J Exp Bot 62:1911–1926. https://doi.org/10.1093/jxb/erq380
Miyamoto N, Steudle E, Hirasawa T, Lafitte R (2001) Hydraulic conductivity of rice roots. J Exp Bot 52:1835–1846. https://doi.org/10.1093/jexbot/52.362.1835
Munns R, Tester M (2008) Mechanisms of salinity tolerance. Annu Rev Plant Biol 59:651–681. https://doi.org/10.1146/annurev.arplant.59.032607.092911
Naseer S, Lee Y, Lapierre C, Franke R, Nawrath C, Geldner N (2012) Casparian strip diffusion barrier in Arabidopsis is made of a lignin polymer without suberin. Proc Natl Acad Sci U S A 109:10101–10106. https://doi.org/10.1073/pnas.1205726109
Nassery H (1979) Salt-induced loss of potassium from plant roots. New Phytol 83:23–27. https://doi.org/10.2307/2433741
Nawrath C, Schreiber L, Franke RB, Geldner N, Reina-Pinto JJ, Kunst L (2013) Apoplastic diffusion barriers in Arabidopsis. Arabidopsis Book 11:e0167. https://doi.org/10.1199/tab.0167
Nieves-Cordones M, Alemán F, Martínez V, Rubio F (2010) The Arabidopsis thaliana HAK5 K+ transporter is required for plant growth and K+ acquisition from low K+ solutions under saline conditions. Mol Plant 3:326–333. https://doi.org/10.1093/mp/ssp102
Niu X, Bressan RA, Hasegawa PM, Pardo JM (1995) Ion homeostasis in NaCl stress environments. Plant Physiol 109:735–742. https://doi.org/10.1104/pp.109.3.735
Persson DP, Chen A, Aarts MG, Salt DE, Schjoerring JK, Husted S (2016) Multi-element bioimaging of Arabidopsis thaliana roots. Plant Physiol 172:835–847. https://doi.org/10.1104/pp.16.00770
Peterson CA, Emanuel ME, Humphreys GB (1981) Pathway of movement of apoplastic fluorescent dye tracers through the endodermis at the site of secondary root formation in corn (Zea mays) and broad bean (Vicia faba). Can J Bot 59:618–625. https://doi.org/10.1139/b81-087
Pitman MG (1984) Transport across the root and shoot/root interactions. In: Staples RC, Toennissen GH (eds) Salinity tolerance in plants: strategies for crop improvement. Wiley, New York, pp 93–123
Pollard M, Beisson F, Li Y, Ohlrogge JB (2008) Building lipid barriers: biosynthesis of cutin and suberin. Trends Plant Sci 13:236–246. https://doi.org/10.1016/j.tplants.2008.03.003
Ranathunge K, Schreiber L (2011) Water and solute permeabilities of Arabidopsis roots in relation to the amount and composition of aliphatic suberin. J Exp Bot 62:1961–1974. https://doi.org/10.1093/jxb/erq389
Ranathunge K, Steudle E, Lafitte R (2005) A new precipitation technique provides evidence for the permeability of Casparian bands to ions in young roots of corn (Zea mays L.) and rice (Oryza sativa L.). Plant Cell Environ 28:1450–1462. https://doi.org/10.1111/j.1365-3040.2005.01391.x
Ranathunge K, Schreiber L, Franke R (2011) Suberin research in the genomics era - new interest for an old polymer. Plant Sci 180:399–413. https://doi.org/10.1016/j.plantsci.2010.11.003
Ranathunge K, Kim YX, Wassmann F, Kreszies T, Zeisler V, Schreiber L (2017) The composite water and solute transport of barley (Hordeum vulgare) roots: effect of suberized barriers. Ann Bot 119:629–643. https://doi.org/10.1093/aob/mcw252
Robbins NE, Trontin C, Duan L, Dinneny JR (2014) Beyond the barrier: communication in the root through the endodermis. Plant Physiol 166:551–559. https://doi.org/10.1104/pp.114.244871
Rubio F, Nieves-Cordones M, Alemán F, Martínez V (2008) Relative contribution of AtHAK5 and AtAKT1 to K+ uptake in the high-affinity range of concentrations. Physiol Plant 134:598–608. https://doi.org/10.1111/j.1399-3054.2008.01168.x
Schreiber L (2010) Transport barriers made of cutin, suberin and associated waxes. Trends Plant Sci 15:546–553. https://doi.org/10.1016/j.tplants.2010.06.004
Schreiber L, Hartmann K, Skrabs M, Zeier J (1999) Apoplastic barriers in roots: chemical composition of endodermal and hypodermal cell walls. J Exp Bot 50:1267–1280. https://doi.org/10.1093/jexbot/50.337.1267
Shabala S, Demidchik V, Shabala L, Cuin TA, Smith SJ, Miller AJ, Davies JM, Newman IA (2006) Extracellular Ca2+ ameliorates NaCl-induced K+ loss from Arabidopsis root and leaf cells by controlling plasma membrane K+-permeable channels. Plant Physiol 141:1653–1665. https://doi.org/10.2307/20205886
Shabala S, Bose J, Fuglsang AT, Pottosin I (2016) On a quest for stress tolerance genes: membrane transporters in sensing and adapting to hostile soils. J Exp Bot 67:1015–1031. https://doi.org/10.1093/jxb/erv465
Steudle E, Peterson CA (1998) How does water get through roots? J Exp Bot 49:775–788. https://doi.org/10.1093/jxb/49.322.775
Sun J, Dai S, Wang R, Chen S, Li N, Zhou X, Lu C, Shen X, Zheng X, Hu Z, Zhang Z, Song J, Xu Y (2009a) Calcium mediates root K+/Na+ homeostasis in poplar species differing in salt tolerance. Tree Physiol 29:1175–1186. https://doi.org/10.1093/treephys/tpp048
Sun J, Chen S, Dai S, Wang R, Li N, Shen X, Zhou X, Lu C, Zheng X, Hu Z, Zhang Z, Song J, Xu Y (2009b) NaCl-induced alternations of cellular and tissue ion fluxes in roots of salt-resistant and salt-sensitive poplar species. Plant Physiol 149:1141–1153. https://doi.org/10.1104/pp.108.129494
Tester M, Davenport R (2003) Na+ tolerance and Na+ transport in higher plants. Ann Bot 91:503–527. https://doi.org/10.1093/aob/mcg058
Vishwanath SJ, Delude C, Domergue F, Rowland O (2015) Suberin: biosynthesis, regulation, and polymer assembly of a protective extracellular barrier. Plant Cell Rep 34:573–586. https://doi.org/10.1007/s00299-014-1727-z
Wang SM, Zhao GQ, Gao YS, Tang ZC, Zhang CL (2005) Puccinellia tenuiflora exhibits stronger selective for K+ over Na+ than wheat. J Plant Nutr 27:1841–1857. https://doi.org/10.1081/PLN-200026435
Wang SM, Zhang JL, Flowers TJ (2007) Low-affinity Na+ uptake in the halophyte Suaeda maritima. Plant Physiol 145:559–571. https://doi.org/10.1104/pp.107.104315
Wang CM, Zhang JL, Liu XS, Li Z, Wu GQ, Cai JY, Flowers TJ, Wang SM (2009) Puccinellia tenuiflora maintains a low Na+ level under salinity by limiting unidirectional Na+ influx resulting in a high selectivity for K+ over Na+. Plant Cell Environ 32:486–496. https://doi.org/10.1111/j.1365-3040.2009.01942.x
Wang Q, Guan C, Wang P, Lv ML, Ma Q, Wu GQ, Zhang JL, Bao AK, Wang SM (2015) Coordination between AtHKT1;1 and AtHAK5 in low-affinity Na+ uptake in Arabidopsis thaliana under mild salt stress. Plant Growth Regul 75:615–623. https://doi.org/10.1007/s10725-014-9964-2
Wang CM, **a ZR, Wu GQ, Yuan HJ, Wang XR, Li JH, Tian FP, Zhang Q, Zhu XQ, He JJ, Kumar T, Wang XL, Zhang JL (2016) The coordinated regulation of Na+ and K+ in Hordeum brevisubulatum responding to time of salt stress. Plant Sci 252:358–366. https://doi.org/10.1016/j.plantsci.2016.08.009
Werck-Reichhart D, Feyereisen R (2000) Cytochromes P450: a success story. Genome Biol 1:1–9. https://doi.org/10.1186/gb-2000-1-6-reviews3003
Yan SP, Zhang QY, Tang ZC, Su WA, Sun WN (2006) Comparative proteomic analysis provides new insights into chilling stress responses in rice. Mol Cell Proteomics 5:484–496. https://doi.org/10.1074/mcp.M500251-MCP200
Yeo AR, Yeo ME, Flowers TJ (1987) The contribution of an apoplastic pathway to sodium uptake by rice roots in saline conditions. J Exp Bot 38:1141–1153. https://doi.org/10.1093/jxb/38.7.1141
Zhang JL, Flowers TJ, Wang SM (2010) Mechanisms of sodium uptake by roots of higher plants. Plant Soil 326:45–60. https://doi.org/10.1007/s11104-009-0076-0
Zimmermann HM, Hartmann K, Schreiber L, Steudle E (2000) Chemical composition of apoplastic transport barriers in relation to radial hydraulic conductivity of corn roots (Zea mays L.). Planta 210:302–311. https://doi.org/10.1007/pl00008138
Acknowledgements
We thank Dr. **aolong Chen (Lanzhou University) for technical assistance and Prof. Fengmin Li (Lanzhou University) for providing WinRHIZO software. This research was supported by grants from the National Natural Science Foundation of China (31730093 and 31802122) and the Fundamental Research Funds for the Central Universities (lzujbky-2018-k01 and 2019HQZZ17).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Responsible Editor: Janusz J. Zwiazek.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Supplemental Fig. S1
The growth status of wild-type and cyp86a1 mutant plants grown under different NaCl concentrations in soil. Seeds were germinated and grown on 1/2 MS plates, 7-d-old seedlings were transferred to soil and grown for 21 d, then treated with different salt treatments (0–30 mM NaCl) for 21 d prior to the images being taken (PDF 170 kb)
Supplemental Fig. S2
Chemical composition of suberin monomers in roots of wild-type and cyp86a1 mutant plants under different concentrations of NaCl. (a) All aliphatic suberin monomers and total aromatic monomers; (b) All ω-hydroxyacid monomers identified; (c) All α,ω-diacid monomers identified. The growth conditions and treatments are the same as for Fig. 2c. Mean values are shown in μg·mg−1 delipidated dry residue (DR) ± SD of three biological replicates, each replicate is made up by 0.4–0.5 g of fresh roots (~10 pooled plants). Columns with different letters indicate significant difference at p < 0.05 (Duncan’s test) (PDF 1186 kb)
Rights and permissions
About this article
Cite this article
Wang, P., Wang, CM., Gao, L. et al. Aliphatic suberin confers salt tolerance to Arabidopsis by limiting Na+ influx, K+ efflux and water backflow. Plant Soil 448, 603–620 (2020). https://doi.org/10.1007/s11104-020-04464-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-020-04464-w