Abstract
Sheep wool waste has become a problem affecting the environment, as today the wool of most species has no commercial application and is considered a waste product. Sheep’s wool is mainly composed of keratin which, due to its protein nature and multiple functional groups, has attracted great interest in applications such as support materials in tissue engineering, bioactive materials, and targeted drug delivery. Support materials can be fabricated by 3D printing by syringe extrusion. However, keratin is not suitable for this technique as it does not present proper rheological characteristics. Alginate, a biopolymer derived from brown seaweed, offers a wide range of viscosities at room temperature and offers good performance in 3D printing. Thus, keratin and alginate-based mixtures, due to their properties and ecoefficiency, are interesting candidates to prepare 3D-printed scaffolds. The aim of this work was to develop fully biobased printable inks containing keratin, alginate, salvia extracts, and cellulose nanofibers. In a first stage, keratose, an oxidized form of keratin, was obtained from sheep wool by a clean extraction methodology, and the miscibility and viscosity of keratose-alginate mixtures were assessed. In a second stage, biobased inks were prepared parting from miscible keratose-alginate mixtures. Flow analysis, spectromechanical analysis, and recovery tests were carried out to analyze the effect of the ink formulation over rheological parameters and printability. Mesh and cylinder geometries were 3D printed and their mechanical properties, as well as shape fidelity and self-standing ability, were assessed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The appearance of synthetic fibers in 1880 gave place to a progressive drop in the presence of wool fibers in the textile industry. In 2020, wool represented only a 1% of the global textile fibers market share [1]. In 2019, The European Union possessed the second largest ovine livestock population, with United Kingdom (26.8%), Spain (18.6%), Rumania (11.4%) and Greece (10.8%) heading the list [2]. The ovine livestock is mainly intended for meat industry and, to a lesser extent, for milk production. Although there are some wools that are very appreciated in the textile industry, such as Merino sheep soft and fine wool, the majority of wool has no commercial applications due to its low quality, and it is considered an agricultural solid waste, being landfilled or incinerated. For example, the wool considered in this work comes from “Latxa” sheep, a domestic sheep native to Basque Country. Its wool is comprised by thick fibers, even thicker than 100 microns, resulting in a coarse material. In this scenario, more than 200,000 tons of wool are generated each year in the European Union [3]. This results in a reduction in the profit for stockbreeders since, together with shear expenses, costs coming from residue management should also be taken into account. Moreover, the mentioned waste management practices are non-ecofriendly and generate a negative impact on the environment.
The main component of wool is keratin. Keratin comprises a sulfur-rich range of proteins, abundant in many living tissues, such as skin or hair [4]. Among others, protein based polymers are considered as the most ecoefficient bioplastics concerning their potential applications over the generated environmental impact [5]. Most of the keratin extractive processes found in literature are laborious and expensive. However, new eco-friendly methods are being developed, such as microwave assisted [6], deep euthectic solvents based [7], or microbial and enzymatic extraction processes [8]. In a previous work, we reported a clean oxidative keratin extraction methodology from “Latxa” sheep wool based on hydrogen peroxide [9], by which keratin in the oxidized form called keratose is obtained. Natural keratin presents intra and intermolecular disulfide bonds, making it chemically stable and water insoluble. In keratose, in turn, disulfide bonds are converted into cysteic acids, making it water soluble. The reported extraction methodology would generate a reduction on the environmental impact associated to this process, increasing the ecoefficiency of derived materials. Moreover, it would enable a large scale production and revalorization in sectors such as bioplastics or biomedicine [10].
Keratinous materials have the potential to be used in biomedical applications [11,12,13,14], such as scaffolds, due to their protein nature and the presence in human tissues. A recent research conducted by C.W. Lin et al. [15] highlighted that keratin scaffolds promoted mother cell adhesion, proliferation and differentiation while, also favoring the epithelialization of damaged dermal tissue. Other works revealed that cells such as fibroblasts are able to reproduce satisfactorily on keratin scaffolds, suggesting an appropriate biocompatibility [16]. It has been reported in literature that 3D scaffolds are more active than 2D scaffolds when tested in vitro with neural stem cells [17]. Thus, it can be said that morphology, dimensionality, accessibility and porosity are critical characteristics of scaffolds. Supplying an appropriate quantity of nutrients and oxygen to implanted cells is crucial for their survival and 3D scaffolds offer larger surface area for cellular adhesion and proliferation, while they allow a flow of nutrients and oxygen to the cells [18]. Nowadays there exist several methods for scaffold manufacturing, such as electrospinning, tissue decellularization, lyophilization or additive manufacturing [19, 20]. Additive manufacturing, also known as 3D printing, has attracted much attention due to the possibility of manufacturing parts on demand, with complex and personalized design. Within 3D printing, several techniques have been developed, such as fused deposition modeling (FDM), stereolithography (SLA), selective laser sintering (SLS) and syringe extrusion printing (SEP). SEP offers the advantage to work directly with the material in suspension, known as ink, overcoming the limitation of available materials range and the need of aggressive processing conditions, such as heat or the use of organic solvents, needed in other additive manufacturing techniques [21, 22]. SEP technique also offers the possibility of incorporating biologic material such as cells to the ink. This specific SEP technique takes the name of bioprinting and the ink is designed as bioink.
Printability defines a material’s suitability to be printed by SEP and it lies mainly on the rheological properties such as viscosity, shear-thinning ability, extrusion, viscoelastic shear moduli, elastic recovery and shear stress [23, 24]. An ink would be printable when it presents extrudability, defined as the ability of an ink to flow through a nozzle, and shape fidelity to reproduce a CAD design by forming a self-standing and non-spreading structure. In consequence, rheological properties of inks need to be tailored. In this scenario, alginate biopolymer is an interesting candidate due to the wide range of viscosity at room temperature at which it can be found [25, 26]. Alginate, a brown-seaweed derived biomaterial, is known for its biocompatibility [27], biodegradability [28] and mechanical integrity [29]. Keratin-alginate mixtures are an interesting approach to combine the properties of both components. For example, Silva et al. developed hybrid hydrogels based on keratin and alginate aimed at tissue engineering applications [30]. They proved that the obtained material offered a combination of chemical and mechanical stability, biocompatibility and cell viability. The rheological modulating ability of nanoentities such as cellulose nanocrystals or cellulose nanofibers has also been studied. For example, Markstedt et al. prepared alginate-nanocellulose bioinks and concluded that nanocellulose conferred the desired rheological behavior while alginate provided mechanical performance [31]. Similarly, Muller et al. converted an alginate sulfate based bioink in a printable material by incorporating nanocellulose [32].
Plant extracts have also received attention in the biomedical field since they can be natural sources of active agents, such as alkaloids, flavonoids, tannins and phenolic compounds. For example, Salvia officinalis L. is a rich source of polyphenols with promising bioactivity, antioxidant and anticancer activities [33]. Indeed, in previous works we demonstrated the antibacterial effect of Salvia officinalis L. against Staphylococcus aureus, Escherichia coli and Pseudomonas aeruginosa, when added to starch or polyurethane matrices [34, 35]. Moreover, similar to cellulose nanoentities, it has been reported that Salvia officinalis L. extracts can act as surfactants [36]. Although previously mentioned research works have demonstrated the potential of keratin and keratin-alginate blends in biomedical applications, no works concerning 3D printing of keratin-containing inks have been published to the best of authors’ knowledge. In this work, firstly keratin in form of keratose was obtained from “Latxa” sheep wool residues, by applying the clean extraction methodology aforementioned, and it was characterized in terms of Fourier transform infrared spectroscopy and thermal performance. Then, keratose-alginate mixtures were analyzed in terms of miscibility and viscosity. Finally, keratose-alginate mixtures were loaded with Salvia officinalis L., and their suitability as inks for SEP was explored performing a deep analysis on the rheological behavior by means of flow analysis, spectromechanical analysis and three interval thixotropy tests. Moreover, inks containing cellulose nanofibers were also considered and their impact on inks rheological behavior was analyzed. Finally, mesh and cylinder geometries were printed and the impact of rheological parameters on shape fidelity and self-standing ability was assessed. Moreover, the mechanical performance of printed parts was evaluated by compressive tests. The valorization of keratin rich sheep wool residues for the preparation of fully biobased inks would promote the implementation of sustainable extraction processes, while promoting the use of sustainable raw materials and contributing to Circular Economy and Ecodesign.
Materials and methods
Materials
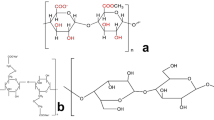
Keratose was extracted from “Latxa” sheep wool, supplied by local stockbreeders. Hydrogen peroxide (30%, Panreac) and HCl (37%, Panreac) were employed in keratose extraction process. Sodium alginate powder from brown algae with medium viscosity (A2033) and molecular weight of 2.4 × 105 g mol− 1 [33] was kindly supplied by Sigma-Aldrich. The simplified chemical structures of sodium alginate and keratose are proposed in Fig. 1a and b, respectively. Freeze dried cellulose nanofibers (CNF) with length of several micrometers and diameter between 50 and 200 nm [37], were provided by the University of Maine (lot.9004-34-6).
Obtaining of salvia extract
Salvia extract was obtained according to the method described in literature [35]. In brief, previously grinded Salvia officinalis L. plant was infused in boiling water (25 mg mL− 1) for 5 min. The obtained suspension was filtered and the obtained liquid phase was collected and lyophilized (-80 °C, 0.1 mbar) for 48 h in a Telstar LyoQuest equipment. The salvia extract was obtained as the dried product after the freeze-drying process.
Extraction of keratose
Keratose was extracted from “Latxa” sheep wool according to the aforementioned oxidative method with hydrogen peroxide [9]. In brief, previously washed and milled wool was immersed in hydrogen peroxide (H2O2, 1.5 M) for 2 h at 50 °C. Wool/H2O2 ratio was kept constant at 50 g L− 1 and pH was set at 11. The obtained liquor was filtered, and the keratose solution was acidified with HCl for keratose precipitation. Thereafter, the acid medium was substituted with water and isopropanol to wash the keratose. Finally, the keratose was lyophilized (-80 °C, 0.1 mbar, 48 h) in a Telstar LyoQuest equipment for posterior utilization.
Preparation of keratose-alginate mixtures
For the preparation of keratose (K) and alginate (A) mixtures, first keratose was dispersed in distilled water using an ultrasonic tip for 3 h at 30% amplitude. Then, alginate powder was added and the solution was subjected to mechanical agitation. The prepared mixtures were labelled as M X-Y where X denoted the K:A weight ratio over the solid content of the mixture and Y the total solid content (wt%). Table 1 shows designation, composition and solid content of the analyzed mixtures, together with viscosity at 0.2 s− 1 (considered as zero-shear rate viscosity), as well as consistency index and flow index values obtained from flow curves and power-law model fitting, respectively.
Preparation of inks
The preparation procedure of keratose-alginate-salvia extract inks was similar to keratose-alginate mixtures. First, salvia extract was dissolved in distilled water at a 10 wt% content with respect to keratose. Then, keratose was added and the mixture was dispersed using an ultrasonic tip for 3 h at 30% amplitude. Subsequently, alginate powder was added and the solution was subjected to mechanical agitation. Inks were labelled as I-X-Y where X denoted the K:A weight ratio over the total weight of the ink, and Y the solid content (wt%). For inks containing CNF, dry CNF was incorporated in previously prepared keratose-alginate-salvia extract inks, at a 2 wt% with respect to the total solid content. The resulting mixture was dispersed in a high shear mixer (Ultraturrax®) for 15 min. Inks containing CNF were labelled as I-X-Y-CNF. Designation and composition of the prepared inks are gathered in Table 2.
3D printing
To observe the impact of rheological parameters over the 3D printing performance, mesh and cylinder geometries were printed using the prepared inks in a Voladora 3D printer (Tumaker S.L., Spain) modified for layer-by-layer syringe extrusion printing. Specifically, a mechanical piston that moved the embolus of a syringe replaced the fused deposition modeling (FDM) head. This allowed the extrusion of the ink through a needle in both axes x and y. Mesh geometry of 20 mm x 20 mm (wide x length) with 2 mm height, with square-shaped pores of 5 mm x 5 mm, and cylinder geometry of 10 mm diameter and 5 mm height were CAD modeled (Fig. 5). Parts were printed at room temperature, at a speed of 6 mm s− 1 and using a 0.8 mm diameter needle. Layer height of 0.2 mm and rectilinear filling pattern were chosen. Finally, printed geometries were freeze-dried at -80 °C and 0.1 mbar for 48 h to obtain porous scaffolds.
Characterization techniques
Fourier transform infrared spectroscopy
The functional groups of keratose and alginate were analyzed by Fourier transform infrared spectroscopy (FTIR). A Nicolet Nexus spectrometer provided with a MKII Golden Gate accessory (Specac) was used, with a diamond crystal at a nominal incidence angle of 45°, and ZnSe lens. Spectra were recorded in attenuated reflection (ATR) mode in the range of 4000 − 650 cm− 1, averaging 64 scans with a resolution of 2 cm− 1.
Thermogravimetric analysis
Thermogravimetric analysis (TGA) of keratose was performed on a TGA/SDTA 851 Metler Toledo equipment in order to evaluate thermal stability. Pure keratose sample was heated from room temperature to 700 °C at a heating rate of 10 °C min− 1, under nitrogen atmosphere.
Rheological measurements
Viscosity of keratose-alginate mixtures was analyzed by flow test. Rheological behavior of the prepared ink formulations was analyzed by flow test, oscillatory shear stress sweep and three-interval-thixotropy tests, using a Haake Viscotester iQ (Thermo Scientific). Tests were carried out by plate-plate geometry (P35/Al adapter), employing plates of 35 mm diameter. Working gap and temperature were set at 1 mm and 25 °C, respectively.
For flow tests, shear rate sweeps from 0.2 to 1000 s− 1 were performed. Prior to each measurement, samples were equilibrated for 40 s at 0.2 s− 1. Flow index and consistency index of each ink were calculated from Power Law (Eq. 1).
where η is the viscosity (Pa·s), γ̇ is the shear rate (s−1), K is the consistency index (Pa·sn) and n is the flow index (dimensionless).
Spectromechanical analysis was carried out by dynamic oscillatory shear stress sweep tests, working in a shear stress range of 10 to 3000 Pa. The evolution of storage modulus (G’) and loss modulus (G’’) with shear stress was determined. G’ and G’’ values were determined at 10 Pa shear stress. Yield point was set at the point of deviation of G’ from linearity, as proposed by Cyriac et al. [38]. Flow point, on the other hand, is defined as the crossover point for G’ and G’’.
Last, structure recovery was analyzed by three-interval-thixotropy tests. Viscosity values were measured at a first shear rate interval of 0.2 s− 1 during 100 s, followed by a second shear rate interval of 50 s− 1 for 100 s, finishing with a third shear rate interval at the same conditions as in the first stage. For the determination of the structural recovery capacity of the inks, a relation of the ink’s viscosity on the first and third shear rate intervals was calculated as expressed in Eq. 2.
where η1I is the viscosity of the material on the first shear rate interval and η3I is the viscosity of the material on the third shear rate interval, both measured after 60 s of the beginning of the interval.
Mechanical properties
Compressive properties of the printed specimens were tested at room temperature, in a Universal testing machine (Instron 5967) equipped with 500 N load cell at a crosshead speed of 2 mm min− 1. 3D printed cylinder shaped specimens were compressed until 60% deformation. According to ASTM D 1621-16 standard, compressive strength (σc) was taken as the stress at the yield point and compressive modulus (Ec) was considered as the slope at the elastic region of stress-strain curve. Densification strain (εd) was calculated as the strain value at the intersection point between the stress plateau and the densification slope. Results were averaged from at least four measurements.
Results and discussion
Keratose and alginate characterization
FTIR results of keratose (Fig. 2a) showed typical spectra of proteins. Bands related with amide I, II and III regions were identified in 1700 − 1200 cm− 1 range. Amide I band, due to C = O stretching, was observed at 1657 cm− 1. Amide II band was identified at 1547 cm− 1, mainly related to out of phase combination of N-H in plane bending and C-N stretching vibration. Amide III band due to C-N stretching was identified at 1240 cm− 1 [39, 40]. According to literature, the identified wavenumbers for amide bands would be indicative of a predominant α-helix conformation of keratose [41, 42]. This also agrees with the mammalian origin of keratin used in this work [40]. Apart from amide regions, bands corresponding to N-H stretching (3296 cm− 1) and C-H stretching (2925 cm− 1) were also identified. Also, a band located at 1042 cm− 1 and a shoulder located at 1174 cm− 1 were identified, and were related to the presence of sulfinate groups [10]. Concerning the spectrum corresponding to alginate, a broad band related to O-H stretching vibration was observed at around 3500 − 3100 cm− 1. The band at 2925 cm− 1 was assigned to C-H stretching and the band at 1591 cm− 1 was related to carboxylate - (C = O)-O- asymmetric stretching. The band at 1406 cm− 1 belonged to C-OH deformation vibration and carboxylate -(C = O)-O- symmetric stretching. Finally, the band at 1026 cm− 1 was assigned to C-O stretching vibration of pyranose rings [43].
Thermal stability of keratose and alginate was analyzed by thermogravimetric analysis. Obtained thermogram (TG) and the derivative (DTG) are shown in Fig. 2b. For keratose, three thermal degradation stages were observed. The first one, at 75 °C, was assigned to the release of moisture and volatile compounds. The second one, beginning around 200 °C, was related to the degradation of side chains, in good accordance to Aluigi et al. who analyzed the thermal degradation of keratin extracted from merino wool [44]. The shoulder observed at around 220 °C in this endotherm was related to the removal of sulfur containing gases. The last stage, starting at 444 °C, could be related to pyrolytic decomposition [45]. The maximum degradation rate, defined as the minimum of DTG curve was determined at 325 °C. A char amount of 20 wt% was measured. In the case of alginate, two-stage thermal degradation was observed. The first stage, beginning at 100 °C, could be related to absorbed water and volatile compounds. The second one, in the range of 195–270 °C was related with disentanglement of polymeric network [46]. A final char amount of 24 wt% was determined for alginate, in good agreement with literature [47]. The maximum degradation rate was measured at 212 °C.
Keratose-alginate mixtures characterization
Prior to the preparation of inks, the miscibility of keratose-alginate aqueous mixtures was considered. In a qualitative approach, both the effect of K:A weight ratio and the effect of solid content were analyzed. Photographs of the mixtures are shown in Fig. 3a. Concerning K:A weight ratio, it was observed that for a constant solid content of 10 wt%, the system was miscible until a K:A weight ratio of 3:1. With higher keratose contents, the system split in two phases. Concerning solid content, it was varied from 6 to 14 wt% kee** K:A weight ratio constant at the miscibility limit of 3:1. It was observed that the system was miscible until a solid content of 10 wt%. At lower K:A ratios the system was miscible up to a solid content of 15 wt%, such as M-2:1–15. Thus, it was concluded that an increase in K:A weight ratio or in solid content hindered miscibility. For further characterization, miscible mixtures listed in Table 1 were considered.
Results concerning flow test of miscible systems are shown in Fig. 3c-e; Table 1. All samples showed pseudoplastic behavior, evidenced by flow index values in 0–1 range. Analyzing the effect of K:A ratio, it was observed that viscosity values increased as K:A ratio decreased in mixtures with both 6 wt% and 10 wt% solid content, Fig. 3c and d respectively, probably related to the higher content of alginate. The same effect was qualitatively observed in Fig. 3b, where mixtures with 6 wt% solid content at different K:A ratios are shown. Analyzing these results it can be concluded that the increase of alginate content resulted in inks with high viscosity, which may be related with the high molecular weight of alginate (2.4 × 105 g mol− 1) [48, 49]. Concerning solid content, Fig. 3e, it was verified that it played a vital role concerning viscosity of the mixtures [50], since M-2:1–15 showed the highest zero viscosity value (2383 Pa.s) among all analyzed samples.
(a) Photographs of keratose-alginate mixtures, (b) qualitative evolution of viscosity in keratose-alginate mixtures with 6 wt% solid content and different K:A ratios, and the evolution of viscosity as a function of shear rate analyzing (c) the effect of K:A ratio in keratose-alginate mixtures with 6 wt% solid content, (d) the effect of K:A ratio in keratose-alginate mixtures with 10 wt% solid content, and (e) the effect of variation of solid content in keratose-alginate mixtures with K:A ratio of 2:1
Consistency index values (Table 1) showed a direct relationship with the alginate content of the mixtures, in concordance with zero-shear viscosity values. The highest values were measured for M-1:1–10 (244 Pa.sn) and M-2:1–15 (756 Pa.sn) samples. Concerning the effect of keratose in the mixtures, a notorious decrease of consistency index values was observed as keratose content increased, when comparing mixtures with the same solid content. With respect to flow index values (Table 1), values did not show a direct relationship with solid content. As a general trend, values decreased with keratose incorporation, improving the pseudoplastic behavior of the mixtures. M-3:1–10 sample did not follow this tendency, probably because this mixture was on the limit of miscibility and it would have influenced the rheological behavior.
From this preliminary study, it was concluded that samples M-1:1–10 and M-2:1–15 showed the highest potential to be used as inks, since high zero-shear viscosity and consistency index values would result in good shape fidelity [51]. On the other hand, results suggested that increasing the solid content would be an effective strategy to maximize the keratose incorporation on the inks.
Rheological characterization of inks
Rheological values determined for the inks are gathered in Table 3. Flow curves of prepared inks are shown in Fig. 4a.
Similar to their mixture counterparts, inks also showed pseudoplastic behavior with flow index values in the range of 0–1, vital for a proper 3D printing process. The incorporation of Salvia caused an increase of viscosity and consistency index values when comparing both M-1:1–10 vs. I 1:1–10 and M-2:1–15 vs. I-2:1–15 (Tables 1 and 3). This behavior was also observed by A. Silva-Weiss et al. on chitosan and chitosan-starch blends with murta leaf extract [52]. On the other hand, the change in flow index was less pronounced and did not follow a direct trend, in accordance with literature reports [53]. Concerning the effect of solid content in rheology, I-2:1–15 showed higher viscosity than I-1:1–10 in the studied shear rate range, presumably due to the higher solid content of I-2:1–15, as previously commented. Unexpectedly, the incorporation of CNF resulted in a reduction of zero-shear viscosity and consistency index values when comparing I-1:1–10 and I 1:1-10-CNF. This would suggest that alginate-CNF interactions can compete with alginate-water interactions, reducing the viscosity of the inks. A similar effect was observed by Liu S. et al. [54] when analyzing graphene oxide-carrageenan hydrogels, reporting changes in gelation ability of carrageenan in aqueous solution as a consequence of graphene-oxide incorporation. Moreover, it is also reported in the literature that CNF incorporation can cause a disruption on the alginate chain ordering [55]. Thus, it could be said that despite the hydrogen bonding interactions occurring due to the presence of CNF [56, 57], they were not sufficient to counteract the disrupted interactions and hence the viscosity of the system decreased. On the contrary, on the inks with higher solid content, the incorporation of CNF resulted in a notorious increase on viscosity and consistency index values when comparing I-2:1–15 and I-2:1-15-CNF. This could be indicative that more hydrogen bonding interactions occurred, sufficient to counteract the effect of the disrupted interactions. These results might suggest that I-2:1-15-CNF would offer the best shape fidelity. In both I-1:1–10 and I-2:1–15, the incorporation of CNF caused a decrease in flow index values, improving the pseudoplasticity and thus extrudability of the inks. Low flow index values reflect the incapability of the system to reestablish interactions occurring at rest, suggesting that CNF interfere on inks structure.
Apart from pseudoplasticity, G’ and G’’ values and the ratio between them are important parameters when analyzing printability of inks [58]. To that end, spectromechanical analysis was carried out by a shear stress sweep test (Fig. 4b). All inks showed G’ > G’’ values, indicating a solid-like behavior. According to literature, a minimum G’ value of 103 Pa is required to obtain non spreading and self-standing 3D structures [59]. Similarly, inks with G’ values of 105 Pa might be too stiff to be extruded [60]. I-1:1–10 showed a G’ value of 840 Pa, below the theoretical minimum. Comparing I-1:1–10 and I-2:1–15, it was observed that G’ value increased, suggesting that I-2:1–15 would offer better shape fidelity and better ability to support the weight of deposited layers. Thus, higher solid content in I-2:1–15 would have contributed to the increase of G’ value. Furthermore, the addition of CNF both in I-1:1-10-CNF and I-2:1-15-CNF resulted in an appropriate strategy to increase G’ values of the inks.
The ratio of G’’ to G’ is defined as the loss tangent (tanδ). To obtain good printability, a balance should exist between the two modulus and a printability range can be defined. For example, T. Gao et al. [61] determined a tanδ range of 0.25–0.45 for satisfactory printing of alginate-gelatin bioinks. In the case of alginate reinforced with polysaccharides such as CNF, a tanδ range of 0.2–0.5 was proposed [37]. High tanδ values, due to disproportionally high loss modulus, would cause ink spreading upon deposition. On the contrary, low tanδ values due to very high storage modulus would result in non-uniformity of the filament and problems of cohesion between the printed layers [61,62,63]. Considering this, 0.86 value determined for I-1:1–10 suggested that material spreading problems could occur due to the predominance of viscous behavior rather than elastic. The increase of solid content as well as the incorporation of CNF gave place to a reduction of tanδ, suggesting that the formation of an elastic network was promoted rather than viscous, and better shape fidelity would be achieved.
Yield point, defined as the stress where a drop in G’ value occurs, is another parameter affecting the printability of inks. I-1:1–10 and I-2:1–15 showed similar yield stress values, 114 and 137 Pa, respectively. High yield stress values might favor shape fidelity [64]. However, it should be taken into account that high yield stress values would result in high forces needed to extrude the material [37], compromising the extrudability. It was observed that the incorporation of CNF caused a decrease in yield point, thus lowering the pressure needed for printing and improving extrudability. Moreover, as previously commented, the increase of G’ values as a consequence of CNF incorporation would also promote shape fidelity. Flow point values, determined at G’ = G’’, showed the same tendency as viscosity values did. The low flow point value determined for I-1:1-10-CNF and the relative proximity of yield and flow points may compromise shape fidelity due to filament spreading. The results until now indicate that the rheological behavior of I 2:1–15 and specially I-2:1-15-CNF would be the most suitable for printing, with a good combination of viscosity, extrudability and storage modulus.
The elastic recovery, that is, the ability of the inks to recover its original structure after being subjected to high shear rates, was assessed by three-interval thixotropy test and results are shown in Fig. 5. When shear was applied, an instantaneous decrease of viscosity was observed for all inks. This was indicative of flow ability, suggesting good suitability of the inks to be processed by 3D printing. Subsequent to high shear interval, when shear ceased, inks showed high viscosity values. The recovery percentage of each ink after 60 s was calculated and results are shown in Table 3. Although recovery percentages were not very high, the high viscosity of inks and adequate tanδ values would contribute to shape fidelity and avoid ink spreading. It was also observed that recovery percentages improved with CNF incorporation, suggesting a faster recovery of CNF-based interactions. It is also worth noting that thanks to the significantly higher initial viscosity of inks with 15 wt% solid content, they were able to maintain higher viscosities than inks with 10 wt% after the high shear rate stage, despite their lower recovery values, signaling to a better shape fidelity in 3D printing.
Characterization of printed samples
Photographs of 3D printed mesh and cylinder specimens are shown in Fig. 6. When comparing mesh specimens printed with I-1:1–10 and I-2:1–15 inks, it could be said that I 2:1–15 gave place to a more detailed reproduction of CAD design, showing sharper edges. In the same fashion, a better ability to reproduce cylinder geometries was denoted. Thus, it could be said that I-2:1–15 provided better shape fidelity and self-standing ability. Phenomena like filament spreading and poor self-standing ability were observed when printing with ink I-1:1–10, probably due to the lower viscosity, consistency index and G’ values, together with a higher tanδ value, as commented previously. The structural recovery did not seem to be such a critical factor, since a value of 35% was measured for I-2:1–15 while for I-1:1–10 it was 51% (Table 3). With relation to the previously commented, the higher final viscosity values may be responsible for better shape fidelity and self-standing ability rather than structural recovery values.
When observing the effect of CNF incorporation in I-1:1–10, a more pronounced filament spreading effect was observed in the mesh specimen printed with I-1:1-10-CNF than its counterpart I-1:1–10. This effect was attributed to the lowering of viscosity and consistency index because of CNF incorporation together with a decrease in flow point, as reported previously. In contrast, CNF incorporation resulted in an improvement when printing cylinder specimens. This may suggest that the increase of G’ and structural recovery, together with a decrease of tanδ were responsible for a better self-standing ability shown by I-1:1-10-CNF. In the case of CNF incorporation in I-2:1–15, it resulted in a general improvement of rheological properties, observing a satisfactory performance of the ink. Both mesh and cylinder specimens were printed with high shape fidelity and self-standing ability was also observed. Again, the notorious improvement of shape fidelity and self-standing ability observed for specimens printed with 1–2:1-15-CNF when compared to specimens printed with I-1:1-10-CNF supported the idea that final viscosity values should be taken into account more than structural recovery values. This was in good agreement with the effect observed when comparing I-1:1–10 vs. I-1-1:1-10-CNF printed specimens.
To analyze the impact of ink formulation over the mechanical performance of the printed parts, compressive mechanical analysis was carried out in cylinder specimens. Compressive properties were determined for cylinder specimens printed with I-1:1-10CNF and I-2:1-15-CNF (Table 4) since no satisfactory parts, able to be tested, were printed with I-1:1–10 and I-2:1–15. It should be taken into account that inks are in viscous liquid state while parts comprise the solid component of the ink. In this way, mechanical performance of the parts could not be directly related with composition and rheological behavior of inks.
Concerning the results, it was observed that parts printed with I-2:1-15-CNF showed remarkably higher compressive Young modulus values than parts printed with I-1:1-10CNF. Specific Young modulus of cylinders printed with 1–2:1-15-CNF was also notably higher, although the densities measured for specimens printed with both inks were very similar. These results evidenced the strong reinforcing effect of keratose. In good agreement with this, higher compressive strength was also determined for I-2:1-15CNF derived cylinders. The densification strain marks the total collapse of porous structure and the beginning of a notorious increase in stress values, known as densification regime [65]. In this case, densification strain values were very similar for all samples, probably due to very similar density values [66].
Conclusions
Miscible keratose alginate systems were prepared by greenly extracted keratose and renewably sourced alginate. It was observed that the increase of both total solid content and alginate content resulted in an increase of viscosity. Fully biobased printable inks were satisfactorily prepared containing keratose, alginate, salvia extracts and cellulose nanofibers. Concerning rheological behavior, salvia incorporation caused an increase of viscosity. Furthermore, increasing solid content resulted in an appropriate strategy to improve the printability of inks: viscosity and consistency index increased and so did the storage modulus. Moreover, tanδ value shifted to a more balanced value between loss modulus and storage modulus. On the contrary, extrudability was not improved. The incorporation of CNF did not cause the same effect in both inks viscosity, so, the performance of CNF as an effective rheological modulator seems to be influenced by solid content. However, it is to mention that the incorporation of CNF resulted in a general improvement of storage modulus. The analysis of printed parts allowed assessing the impact of inks rheological parameters over the shape fixity and self-standing ability. In general, it was concluded that viscosity and tanδ values were directly related with shape fixity. Moreover, storage modulus showed to be a crucial factor when self-standing ability of inks was implied. In fact, only inks containing CNF were able to print cylinder geometries, which was related to their high storage modulus value. Keratose noticeably improved mechanical properties of the printed parts, obtaining samples with higher Young modulus and compressive strength.
References
textileexchange.org, Preferred-Fiber-and-Materials-Market-Report_2021. Available online: https://textileexchange.org/wp-content/uploads/2021/08/Textile-Exchange_Preferred-Fiber-and-Materials-Market-Report_2021.pdf (accessed on 05 Sept 2022)
ec.europa.eu/Eurostat, Sheep population-annual data. Available online: https://ec.europa.eu/eurostat/databrowser/view/apro_mt_lssheep/default/table?lang=en (accessed on 05 Sept. 2022)
Petek, B.; Marinšek Logar, R. Management of waste sheep wool as valuable organic substrate in European Union countries. J Mater Cycles Waste Manag 2020, 23, 44–54, DOI https://doi.org/10.1007/s10163-020-01121-3.
Wang, B.; Yang, W.; McKittrick, J.; Meyers, M.A. Keratin: Structure, mechanical properties, occurrence in biological organisms, and efforts at bioinspiration. Progress in materials science 2016, 76, 229–318, DOI https://doi.org/10.1016/j.pmatsci.2015.06.001.
Álvarez-Castillo, E.; Felix, M.; Bengoechea, C.; Guerrero, A. Proteins from Agri-Food Industrial Biowastes or Co-Products and their applications as green materials. Foods 2021, 10, 981, DOI https://doi.org/10.3390/foods10050981.
Pulidori, E.; Micalizzi, S.; Bramanti, E.; Bernazzani, L.; Duce, C.; De Maria, C.; Montemurro, F.; Pelosi, C.; De Acutis, A.; Vozzi, G.; Tinè, M.R. One-Pot process: microwave-assisted keratin extraction and direct electrospinning to obtain keratin-based bioplastic. International Journal of Molecular Sciences. 2021, 22, 9597, DOI https://doi.org/10.3390/ijms22179597.
Okoro, O.V.; Jafari, H.; Hobbi, P.; Lei, N.; Houman, A.; Amin, S. Enhanced keratin extraction from wool waste using a deep eutectic solvent. Chemical Papers 2022, 76, 2637–2648, DOI https://doi.org/10.1007/s11696-021-02029-4.
Feroz, S.; Muhammad, N.; Ratnayake, J.; Dias, G. Keratin - based materials for biomedical applications. Bioactive Materials 2020, 5, 3, 496–509, DOI https://doi.org/10.1016/j.bioactmat.2020.04.007
Fernández-d’Arlas, B. Improved aqueous solubility and stability of wool and feather proteins by reactive-extraction with H2O2 as bisulfide (SS) splitting agent. European Polymer Journal 2018, 103, 187–197, DOI https://doi.org/10.1016/j.eurpolymj.2018.04.010.
Fernández-d’Arlas, B. Tough and functional cross-linked bioplastics from Sheep wool keratin. Scientific reports 2019, 9, 14810–12, DOI https://doi.org/10.1038/s41598-019-51393-5.
Zhu, H.; Li, R.; Wu, X.; Chen, K.; Che, J. Controllable fabrication and characterization of hydrophilic PCL/wool keratin nanonets by electronetting. European polymer journal 2017, 86, 154–161, DOI https://doi.org/10.1016/j.eurpolymj.2016.11.023.
Täuber, A.; Müller-Goymann, C.C. In vitro permeation and penetration of ciclopirox olamine from poloxamer 407-based formulations – comparison of isolated human stratum corneum, bovine hoof plates and keratin films. International journal of pharmaceutics 2015, 489, 73–82, DOI https://doi.org/10.1016/j.ijpharm.2015.04.043.
Yang, G.; Yao, Y.; Wang, X. Comparative study of kerateine and keratose based composite nanofibers for biomedical applications. Materials Science & Engineering C 2018, 83, 1–8, DOI 10.1016/j.msec.2017.07.057.
de Guzman, R.C.; Saul, J.M.; Ellenburg, M.D.; Merrill, M.R.; Coan, H.B.; Smith, T.L.; Van Dyke, M.E. Bone regeneration with BMP-2 delivered from keratose scaffolds. Biomaterials 2012, 34, 1644–1656, DOI https://doi.org/10.1016/j.biomaterials.2012.11.002.
Lin, C.; Chen, Y.; Tang, K.; Yang, K.; Cheng, N.; Yu, J. Keratin scaffolds with human adipose stem cells: physical and biological effects toward wound healing. J Tissue Eng Regen Med 2019, DOI https://doi.org/10.1002/term.2855.
Kakkar, P.; Verma, S.; Manjubala, I.; Madhan, B. Development of keratin–chitosan–gelatin composite scaffold for soft tissue engineering. Materials Science Engineering C 2014, 45, 343–347, DOI https://doi.org/10.1016/j.msec.2014.09.021.
Jiang, Z.; Song, Q.; Tang, M.; Yang, L.; Cheng, Y.; Zhang, M.; Xu, D.; Cheng, G. Enhanced Migration of neural stem cells by Microglia grown on a Three-Dimensional Graphene Scaffold. ACS applied materials & interfaces 2016, 8, 25069–25077, DOI https://doi.org/10.1021/acsami.6b06780.
Hosseinkhani, M.; Mehrabani, D.; Karimfar, M.H.; Bakhtiyari, S.; Manafi, A.; Shirazi, R. Tissue engineered scaffolds in regenerative medicine. World J Plast Surg. 2014, 3, 3–7.
Zhao, P.; Gu, H.; Mi, H.; Rao, C.; Fu, J.; Turng, L. Fabrication of scaffolds in tissue engineering: a review. Front Mech Eng 2017, 13, 107–119, DOI https://doi.org/10.1007/s11465-018-0496-8.
Gupta Sweta, K.; Gupta, S.K. Decellularization methods for scaffold fabrication. Methods in Molecular Biology (New York, NY, United States) 2018, 1577, 1–10.
Hsieh, F.; Lin, H.; Hsu, S. 3D bioprinting of neural stem cell-laden thermoresponsive biodegradable polyurethane hydrogel and potential in central nervous system repair. Biomaterials 2015, 71, 48–57, DOI https://doi.org/10.1016/j.biomaterials.2015.08.028.
Hung, K.; Tseng, C.; Dai, L.; Hsu, S. Water-based polyurethane 3D printed scaffolds with controlled release function for customized cartilage tissue engineering. Biomaterials 2016, 83, 156–168, DOI https://doi.org/10.1016/j.biomaterials.2016.01.019.
Schwab, A.; Levato, R.; D’Este, M.; Piluso, S.; Eglin, D.; Malda, J. Printability and shape fidelity of Bioinks in 3D bioprinting. Chem Rev 2020, 120, 11028–11055.
Bedell Matthew, L.M. Polymeric Systems for Bioprinting. Chem Rev 2020, 120, 10744–10792.
Jeon, O; Bouhadir, K. H.; Mansour, J. M.; Alsberg, E. O. Photocrosslinked alginate hydrogels with tunable biodegradation rates and mechanical properties. Biomaterials 2009, 30, 2724–2734.
Zehnder T.; Sarker, B.; Boccaccini, A. R.; Detsch, R. Evaluation of an alginate-gelatine crosslinked hydrogel for bioplotting. Biofabrication 2015, 7, 1–025001.
Lee, K.Y.; Mooney, D.J. Alginate: Properties and biomedical applications. Progress in polymer science 2012, 37, 106–126, DOI https://doi.org/10.1016/j.progpolymsci.2011.06.003.
Martău, G.A.; Mihai, M.; Vodnar, D.C. The Use of Chitosan, Alginate, and pectin in the Biomedical and Food Sector—Biocompatibility, Bioadhesiveness, and Biodegradability. Polymers 2019, 11, DOI https://doi.org/10.3390/polym11111837.
Drury, J.L.; Dennis, R.G.; Mooney, D.J. The tensile properties of alginate hydrogels. Biomaterials 2004, 25, 3187–3199, DOI https://doi.org/10.1016/j.biomaterials.2003.10.002.
Silva, R; Singh, R.; Sarker, B.; Papageorgiou, D.G.; Juhasz, J.A.; Roether, J.A.; Cicha, I.; et al. Hybrid hydrogels based on keratin and alginate for tissue engineering. Journal of Materials Chemistry B 2014, 2, 5441–5451, DOI https://doi.org/10.1039/C4TB00776J.
Markstedt, K.; Mantas, A.; Tournier, I.; Martínez Ávila, H.; Hägg, D.; Gatenhom, P. 3D Bioprinting Human Chondrocytes with Nanocellulose-Alginate Bioink for Cartilage tissue Engineering Applications. Biomacromolecules 2015, 16, 1489–1496, DOI https://doi.org/10.1021/acs.biomac.5b00188
Müller, M.; Öztürk, E.; Oystein, A.; Gatenholm, P.; Zenobi-Wong, M. Alginate Sulfate-Nanocellulose Bioinks for Cartilage Bioprinting Applications. Ann Biomed Eng 2017, 45, 210–223, DOI: https://doi.org/10.1007/s10439-016-1704-5.
Dziadek, M.; Dziadek, K.; Zagrajczuk, B.; Menaszek, E.; Cholewa-Kowalska, K. Poly(ε-caprolactone)/bioactive glass composites enriched with polyphenols extracted from sage (Salvia officinalis L.). Mat. Lett. 2016, 183, 386–390, DOI https://doi.org/10.1016/j.matlet.2016.07.077.
Gonzalez-Munduate, K.; García-Astrain, C.; Santamaria-Echart, A.; Ugarte, L.; Averous, L.; Eceiza, A.; Gabilondo, N. Starch/graphene hydrogels via click chemistry with relevant electrical and antibacterial properties. Carbohydrate Polymers 2018, 202, 372–381, DOI https://doi.org/10.1016/j.carbpol.2018.09.007.
Santamaria-Echart, A.; Fernandes, I.; Barreiro, F.; Retegi, A.; Arbelaiz, A.; Corcuera, M.A.; Eceiza, A. Development of waterborne polyurethane-ureas added with plant extracts: study of different incorporation routes and their influence on particle size, thermal, mechanical and antibacterial properties. Progress in organic coatings 2018, 117, 76–90, DOI https://doi.org/10.1016/j.porgcoat.2018.01.006.
Santamaria-Echart, A.; Fernandes, I.; Ugarte, L.; Barreiro, F.; Corcuera, M.A.; Eceiza, A. Green nanocomposites from Salvia-based waterborne polyurethane-urea dispersions reinforced with nanocellulose 2021, 150, 105989, DOI https://doi.org/10.1016/j.porgcoat.2020.105989.
Olmos-Juste, R.; Alonso-Lerma, B.; Pérez-Jiménez, R.; Gabilondo, N.; Eceiza, A. 3D printed alginate-cellulose nanofibers based patches for local curcumin administration. Carbohydrate polymers 2021, 264, 118026, DOI https://doi.org/10.1016/j.carbpol.2021.118026.
Cyriac, F.; Lugt, P. M.; Bosman, R. On a New Method to determine the yield stress in lubricating grease. Tribology Transactions 2015, 58, 1021–1030, DOI: https://doi.org/10.1080/10402004.2015.1035414.
Barth, A. Infrared spectroscopy of proteins. Biochimica et biophysica acta. Bioenergetics 2007, 1767, 1073–1101, DOI https://doi.org/10.1016/j.bbabio.2007.06.004.
Zoccola, M.; Aluigi, A.; Tonin, C. Characterisation of keratin biomass from butchery and wool industry wastes. Journal of molecular structure 2009, 938, 35–40, DOI https://doi.org/10.1016/j.molstruc.2009.08.036.
Lym An, D.J.; Urray-Wijelath, J.M.; Feughelm An, M. Effect of temperature on the conformation of extended a -Keratin. APPLIED SPECTROSCOPY 2001, 55, DOI https://doi.org/10.1366/0003702011952343
Wojciechowska, E.; Włochowicz, A.; Wesełucha-Birczyńska, A. Application of Fourier-transform infrared and Raman spectroscopy to the study of the influence of orthosilicic acid on the structure of wool fibre. Journal of Molecular Structure 2000, 555, 397, DOI https://doi.org/10.1016/s0022-2860(00)00626-8.
Fenoradosoa, T.A.; Ali, G.; Delattre, C.; Laroche, C.; Petit, E.; Wadouachi, A.; Michaud, P. Extraction and characterization of an alginate from the brown seaweed Sargassum turbinarioides Grunow. J Appl Phycol 2009, 22, 131–137, DOI https://doi.org/10.1007/s10811-009-9432-y.
Aluigi, A.; Zoccola, M.; Vineis, C.; Tonin, C.; Ferrero, F.; Canetti, M. Study on the structure and properties of wool keratin regenerated from formic acid. International journal of biological macromolecules 2007, 41, 266–273, DOI https://doi.org/10.1016/j.ijbiomac.2007.03.002.
Pakkaner, E.; Yalçin, D.; Uysal, B.; Top, A. Self-assembly behavior of the keratose proteins extracted from oxidized Ovis aries wool fibers. International Journal of Biological Macromolecules 2019, 125, 1008–1015, DOI https://doi.org/10.1016/j.ijbiomac.2018.12.129.
Ross, A. B.; Hall, C.; Anastasakis, K.; Westwood, A.; Jones, J.M.; Crewe, R.J. Influence of cation on the pyrolysis and oxidation of alginates. Journal of Analytical and Applied Pyrolysis 2011, 91, 344–351, DOI https://doi.org/10.1016/j.jaap.2011.03.012.
Siddaramaiah,, Swamy, T.M.M.; Ramaraj, B.; Lee, J.H. Sodium alginate and its blends with starch: thermal and morphological properties. J. Appl. Polym. Sci. 2008, 109, 4075–4081, DOI https://doi.org/10.1002/app.28625.
Gombotz, W.R.; Wee, S.F. Protein release from alginate matrices. Advanced drug delivery reviews 2012, 64, 194–205, DOI https://doi.org/10.1016/j.addr.2012.09.007.
Hernández-González, A.C.; Téllez-Jurado, L.; Rodríguez-Lorenzo, L.M. Alginate hydrogels for bone tissue engineering, from injectables to bioprinting: a review. Carbohydrate polymers 2020, 229, 115514, DOI https://doi.org/10.1016/j.carbpol.2019.115514.
Vadillo, J.; Larraza, I.; Calvo-Correas, T.; Gabilondo, N.; Derail, C.; Eceiza, A. Design of a Waterborne polyurethane–urea ink for direct ink writing 3D Printing. Materials 2021, 14, 3287, DOI https://doi.org/10.3390/ma14123287.
Williams, D.; Thayer, P.; Martinez, H.; Gatenholm, E.; Khademhosseini, A. A perspective on the physical, mechanical and biological specifications of bioinks and the development of functional tissues in 3D bioprinting. Bioprinting 2018, 9, 19–36, DOI https://doi.org/10.1016/j.bprint.2018.02.003.
Silva-Weiss, A.; Bifani, V.; Ihl, M.; Sobral, P.J.A.; Gómez-Guillén, M.C. Structural properties of films and rheology of film-forming solutions based on chitosan and chitosan-starch blend enriched with murta leaf extract. Food Hydrocolloids 2013, 31, 458–466, DOI https://doi.org/10.1016/j.foodhyd.2012.11.028.
Yang, J.; Pal, R. Investigation of surfactant-polymer interactions using rheology and surface tension measurements. Polymers 2020, 12, 2302. DOI https://doi.org/10.3390/polym12102302.
Liu, S.; Bao, H.; Li, L. Thermoreversible gelation and scaling laws for graphene oxide-filled κ-carrageenan hydrogels. European polymer journal 2016, 79, 150–162, DOI https://doi.org/10.1016/j.eurpolymj.2016.04.027.
Deepa, B.; Abraham, E.; Pothan, L.A.; Cordeiro, N.; Faria, M.; Thomas, S. Biodegradable nanocomposite Films based on Sodium Alginate and Cellulose Nanofibrils. Materials 2016, 9, DOI https://doi.org/10.3390/ma9010050.
Abdollahi, M.; Alboofetileh, M.; Behrooz, R.; Rezaei, M.; Miraki, R. Reducing water sensitivity of alginate bio-nanocomposite film using cellulose nanoparticles. International journal of biological macromolecules 2013, 54, 166–173, DOI https://doi.org/10.1016/j.ijbiomac.2012.12.016.
Alashwal, B.Y.; Saad Bala, M.; Gupta, A.; Sharma, S.; Mishra, P. Improved properties of keratin-based bioplastic film blended with microcrystalline cellulose: a comparative analysis. Journal of King Saud University. Science 2020, 32, 853–857, DOI https://doi.org/10.1016/j.jksus.2019.03.006.
Chimene, D.; Lennox, K.K.; Kaunas, R.R.; Gaharwar, A.K. Advanced Bioinks for 3D Printing: a materials Science Perspective. Ann Biomed Eng 2016, 44, 2090–2102, DOI https://doi.org/10.1007/s10439-016-1638-y.
Li, L.; Lin, Q.; Tang, M.; Duncan, A.J.E.; Ke, C. Advanced polymer designs for direct-ink‐write 3D Printing. Chemistry: a European journal 2019, 25, 10768–10781, DOI https://doi.org/10.1002/chem.201900975.
Álvarez-Castillo, E.; Oliveira, S.; Bengoechea, C.; Sousa, I.; Raymundo, A.; Guerrero, A. A rheological approach to 3D printing of plasma protein based doughs. Journal of food engineering 2021, 288, 110255, DOI https://doi.org/10.1016/j.jfoodeng.2020.110255.
Gao, T.; Gillispie, G.J.; Copus, J.S.; Pr, A.K.; Seol, Y.; Atala, A.; Yoo, J.J.; Lee, S.J. Optimization of gelatin–alginate composite bioink printability using rheological parameters: a systematic approach. Biofabrication 2018, 10, DOI https://doi.org/10.1088/1758-5090/aacdc7.
Duan, B.; Hockaday, L.A.; Kang, K.H.; Butcher, J.T. 3D bioprinting of heterogeneous aortic valve conduits with alginate/gelatin hydrogels. Journal of Biomedical Materials Research Part A 2013, 101A, 1255–1264, DOI https://doi.org/10.1002/jbm.a.34420.
Vadillo, J.; Larraza, I.; Calvo-Correas, T.; Gabilondo, N.; Derail, C.; Eceiza, A. Role of in situ added cellulose nanocrystals as rheological modulator of novel waterborne polyurethane urea for 3D-printing technology. Cellulose 2021, 28, 4729–4744, DOI https://doi.org/10.1007/s10570-021-03826-6.
Sweeney, M.; Campbell, L.L.; Hanson, J.; Pantoya, M.L.; Christopher, G.F. Characterizing the feasibility of processing wet granular materials to improve rheology for 3D printing. J Mater Sci 2017, 52, 13040–13053, DOI https://doi.org/10.1007/s10853-017-1404-z.
Brydon, A.D.; Bardenhagen, S.G.; Miller, E.A.; Seidler, G.T. Simulation of the densification of real open-celled foam microstructures. Journal of the mechanics and physics of solids 2005, 53, 2638–2660, DOI https://doi.org/10.1016/j.jmps.2005.07.007.
Mondal, D.P.; Barnwal, A.K.; Diwakar, V. Effect of strain rate and relative density on the Compressive deformation of Open Cell Ti6Al Alloy Foam through P/M Route. Journal of applied mechanical engineering 2017, 6, DOI https://doi.org/10.4172/2168-9873.1000291.
Acknowledgements
Authors deeply thank the finantial support provided by the BASQUE GOVERNMENT in the frame of Grupos Consolidados (IT1690-22), the UNIVERSITY OF THE BASQUE COUNTRY (UPV/EHU) (GIU18/216 Research Group) and the PROVINCIAL COUNCIL OF GIPUZKOA (DG19/28 and DG20/06). Technical and human support provided by SGIker (UPV/EHU /FEDER, EU) is gratefully acknowledged.
Funding
Open Access funding provided thanks to the CRUE-CSIC agreement with Springer Nature.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ugarte, L., Fernández-d’Arlas, B., Larraza, I. et al. Revalorization of sheep-wool keratin for the preparation of fully biobased printable inks. J Polym Environ 31, 4302–4313 (2023). https://doi.org/10.1007/s10924-023-02881-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10924-023-02881-3