Abstract
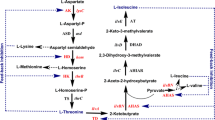
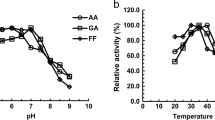
Escherichia coli isocitrate dehydrogenase (IDH) is regulated by reversible phosphorylation on Ser113. Latest phosphoproteomic studies revealed that eukaryotic IDHs can also be phosphorylated on the analogous Ser site. So as to understand the possible phosphorylation mechanism, the equivalent Ser of NADP-IDHs from yeast Saccharomyces cerevisiae (ScIDH) and Yarrowia lipolytica(YlIDH) were investigated by site-directed mutagenesis. ScIDH Ser110 and YlIDH Ser103 were replaced by Asp or Glu to mimic a continuous phosphorylation state. Meanwhile, the effects of another four amino acids (Thr, Tyr, Gly, Ala) with various side chain on IDH activity were determined as well. Enzymatic analysis showed that replacement of Ser with Asp or Glu nearly inactivated ScIDH and YlIDH. Four other mutant enzymes of ScIDH, S110T, S110G, S110A, and S110Y, retained 38.07%, 3.24%, 2.65%, and 0.01% of its original activity, and four other mutant enzymes of YlIDH, S103T, S103G, S103A, and S103Y retained 44.26%, 27.99%, 16.29%, and 0.01% of its original activity, respectively. These results suggested that phosphorylation on eukaryotic IDHs has identical consequence to that on the bacterial IDHs. We thus presume that phosphorylation on the substrate-binding Ser shall be a common regulatory mechanism among IDHs.




Similar content being viewed by others
References
Zhu, G., Golding, G. B., & Dean, A. M. (2005). The selective cause of an ancient adaptation. Science, 307(5713), 1279–1282.
Ma, T., Peng, Y., Huang, W., & Ding, J. (2017). Molecular mechanism of the allosteric regulation of the αγ heterodimer of human NAD-dependent isocitrate dehydrogenase. Scientific Reports, 7(1), 40921.
Ren, Z., **ong, Y., Deng, C., & Jiang, S. (2012). Cloning, differential expression, and association analysis with fat traits of porcine IDH3γ gene. Applied Biochemistry and Biotechnology, 166(4), 1112–1120.
Zhao, X. Y., Wang, P., Zhu, G. Y., Wang, B. J., & Zhu, G. P. (2014). Enzymatic characterization of a type II isocitrate dehydrogenase from pathogenic leptospira interrogans serovar Lai strain 56601. Applied Biochemistry and Biotechnology, 172(1), 487–496.
Kim, H., Kim, S. H., Cha, H., Kim, S. R., Lee, J. H., & Park, J. W. (2016). IDH2 deficiency promotes mitochondrial dysfunction and dopaminergic neurotoxicity: implications for Parkinson’s disease. Free Radical Research, 50(8), 853–860.
Thorsness, P. E., & Koshland, D. E., Jr. (1987). Inactivation of isocitrate dehydrogenase by phosphorylation is mediated by the negative charge of the phosphate. Journal of Biological Chemistry, 262(22), 10422–10425.
Dean, A. M., Lee, M. H., & Koshland, D. E., Jr. (1989). Phosphorylation inactivates Escherichia coli isocitrate dehydrogenase by preventing isocitrate binding. Journal of Biological Chemistry, 264(34), 20482–20486.
Hurley, J. H., Dean, A. M., Sohl, J. L., Koshland, D. E., Jr., & Stroud, R. M. (1990). Regulation of an enzyme by phosphorylation at the active site. Science, 249(4972), 1012–1016.
Dean, A. M., & Koshland, D. E., Jr. (1990). Electrostatic and steric contributions to regulation at the active site of isocitrate dehydrogenase. Science, 249(4972), 1044–1046.
LaPorte, D. C. (1993). The isocitrate dehydrogenase phosphorylation cycle: regulation and enzymology. Journal of Cellular Biochemistry, 51(1), 14–18.
Singh, S. K., Matsuno, K., LaPorte, D. C., & Banaszak, L. J. (2001). Crystal structure ofbacillus subtilisisocitrate dehydrogenase at 1.55 Å. Journal of Biological Chemistry, 276(28), 26154–26163.
Singh, S. K., Miller, S. P., Dean, A., Banaszak, L. J., & LaPorte, D. C. (2002). Bacillus subtilisisocitrate dehydrogenase. Journal of Biological Chemistry, 277(9), 7567–7573.
Wang, P., Song, P., **, M., & Zhu, G. (2013). Isocitrate dehydrogenase from Streptococcus mutans: biochemical properties and evaluation of a putative phosphorylation Site at Ser102. PLoS One, 8(3), e58918.
Ceccarelli, C., Grodsky, N. B., Ariyaratne, N., Colman, R. F., & Bahnson, B. J. (2002). Crystal structure of porcine mitochondrial NADP+-dependent isocitrate dehydrogenase complexed with Mn2+and isocitrate. Journal of Biological Chemistry, 277(45), 43454–43462.
Xu, X., Zhao, J., Xu, Z., Peng, B., Huang, Q., Arnold, E., & Ding, J. (2004). Structures of human cytosolic NADP-dependent isocitrate dehydrogenase reveal a novel self-regulatory mechanism of activity. Journal of Biological Chemistry, 279(32), 33946–33957.
Peng, Y., Zhong, C., Huang, W., & Ding, J. (2008). Structural studies ofSaccharomyces cerevesiaemitochondrial NADP-dependent isocitrate dehydrogenase in different enzymatic states reveal substantial conformational changes during the catalytic reaction. Protein Science, 17(9), 1542–1554.
Cozzone, A. J. (1998). Regulation of acetate metabolism by protein phosphorylation in enteric bacteria. Annual Review of Microbiology, 52(1), 127–164.
Prasad, U. V., Vasu, D., Kumar, Y. N., Kumar, P. S., Yeswanth, S., Swarupa, V., Phaneendra, B. V., Chaudhary, A., & Sarma, P. V. G. K. (2013). Cloning, expression and characterization of NADP-dependent isocitrate dehydrogenase from Staphylococcus aureus. Applied Biochemistry and Biotechnology, 169(3), 862–869.
Prasad, U. V., Vasu, D., Yeswanth, S., Swarupa, V., Sunitha, M. M., Choudhary, A., et al. (2015). Phosphorylation controls the functioning ofStaphylococcus aureusisocitrate dehydrogenase – favours biofilm formation. Journal of Enzyme Inhibition Medical Chemistry, 30(4), 655–661.
Balganesh, T., Datta, S.&Ghosh, I. (2004). WIPO Patent Application WO/2004/ 087943 A1. 1–29.
Singh, V. K., & Ghosh, I. (2006). Kinetic modeling of tricarboxylic acid cycle and glyoxylate bypass in Mycobacterium tuberculosis, and its application to assessment of drug targets. Theoretical Biology and Medical Modelling, 3(1), 27.
Mertins, P., Mani, D. R., Ruggles, K. V., Gillette, M. A., Clauser, K. R., Wang, P., et al. (2016). Proteogenomics connects somatic mutations to signalling in breast cancer. Nature, 534(7605), 55–62.
Mertins, P., Yang, F., Liu, T., Mani, D. R., Petyuk, V. A., Gillette, M. A., Clauser, K. R., Qiao, J. W., Gritsenko, M. A., Moore, R. J., Levine, D. A., Townsend, R., Erdmann-Gilmore, P., Snider, J. E., Davies, S. R., Ruggles, K. V., Fenyo, D., Kitchens, R. T., Li, S., Olvera, N., Dao, F., Rodriguez, H., Chan, D. W., Liebler, D., White, F., Rodland, K. D., Mills, G. B., Smith, R. D., Paulovich, A. G., Ellis, M., & Carr, S. A. (2014). Ischemia in tumors induces early and sustained phosphorylation changes in stress kinase pathways but does not affect global protein levels. Molecular and Cellular Proteomics, 13(7), 1690–1704.
Klammer, M., Kaminski, M., Zedler, A., Oppermann, F., Blencke, S., Marx, S., Müller, S., Tebbe, A., Godl, K., & Schaab, C. (2012). Phosphosignature predicts dasatinib response in non-small cell lung cancer. Molecular and Cellular Proteomics, 11(9), 651–668.
Mertins, P., Qiao, J. W., Patel, J., Udeshi, N. D., Clauser, K. R., Mani, D. R., Burgess, M. W., Gillette, M. A., Jaffe, J. D., & Carr, S. A. (2013). Integrated proteomic analysis of post-translational modifications by serial enrichment. Nature Methods, 10(7), 634–637.
Rigbolt, K. T., Prokhorova, T. A., Akimov, V., Henningsen, J., Johansen, P. T., Kratchmarova, I., et al. (2011). Science Signaling, 4, rs3.
Lundby, A., Andersen, M. N., Steffensen, A. B., Horn, H., Kelstrup, C. D., Francavilla, C., et al. (2013). Science Signaling, 6, rs11.
Li, X., Wang, P., Ge, Y., Wang, W., Abbas, A., & Zhu, G. P. (2013). NADP+-specific isocitrate dehydrogenase from oleaginous yeast yarrowia lipolytica CLIB122: biochemical characterization and coenzyme sites evaluation. Applied Biochemistry and Biotechnology, 171(2), 403–416.
Pereira, J. M., Chevalier, C., Chaze, T., Gianetto, Q., Impens, F., Matondo, M., Cossart, P., & Hamon, M. A. (2018). Infection reveals a modification of SIRT2 critical for chromatin association. Cell Reports, 23(4), 1124–1137.
Prezel, E., Elie, A., Delaroche, J., Stoppin-Mellet, V., Bosc, C., Serre, L., et al. (2017). Molecular Biology of the Cell, 29, 154–165.
Boese, C. J., Nye, J., Buster, D. W., McLamarrah, T. A., Byrnes, A. E., Slep, K. C., Rusan, N. M., & Rogers, G. C. (2018). Asterless is a Polo-like kinase 4 substrate that both activates and inhibits kinase activity depending on its phosphorylation state. Molecular Biology of the Cell, 29(23), 2874–2886.
Larkin, M. A., Blackshields, G., Brown, N. P., Chenna, R., McGettigan, P. A., McWilliam, H., Valentin, F., Wallace, I. M., Wilm, A., Lopez, R., Thompson, J. D., Gibson, T. J., & Higgins, D. G. (2007). Clustal W and Clustal X version 2.0. Bioinformatics, 23(21), 2947–2948.
Gouet, P., Courcelle, E., Stuart, D. I., & Metoz, F. (1999). ESPript: analysis of multiple sequence alignments in PostScript. Bioinformatics, 15(4), 305–308.
Wang, P., Lv, C., & Zhu, G. (2015). Novel type II and monomeric NAD+ specific isocitrate dehydrogenases: phylogenetic affinity, enzymatic characterization and evolutionary implication. Scientific Reports, 5(1), 9150.
Zheng, J., & Jia, Z. (2010). Structure of the bifunctional isocitrate dehydrogenase kinase/phosphatase. Nature, 465(7300), 961–965.
Karlstrom, M., Steen, I. H., Madern, D., Fedoy, A. E., Birkeland, N. K., & Ladenstein, R. (2006). The crystal structure of a hyperthermostable subfamily II isocitrate dehydrogenase from Thermotoga maritima. FEBS Journal, 273(13), 2851–2868.
Leiros, H. K., Fedoy, A. E., Leiros, I., & Steen, I. H. (2012). The complex structures of isocitrate dehydrogenase fromClostridium thermocellumandDesulfotalea psychrophilasuggest a new active site locking mechanism. FEBS Open Bio, 2(1), 159–172.
Matsuno, K., Blais, T., Serio, A. W., Conway, T., Henkin, T. M., & Sonenshein, A. L. (1999). Metabolic imbalance and sporulation in an isocitrate dehydrogenase mutant of Bacillus subtilis. Journal of Bacteriology, 181(11), 3382–3391.
Funding
This work was supported by the National Natural Science Foundation of China (31570010), the Provincial Project of Natural Science Research for Colleges and Universities of Anhui Province of China (KJ2018A0319) the Provincial Innovation Project for Oversea Talents in Anhui Province and Innovation Team of Scientific Research Platform in Anhui Universities.
Author information
Authors and Affiliations
Ethics declarations
Conflicts of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, P., Liu, T., Zhou, X. et al. Evaluation of the Potential Phosphorylation Effect on Isocitrate Dehydrogenases from Saccharomyces cerevisiae and Yarrowia lipolytica. Appl Biochem Biotechnol 187, 1131–1142 (2019). https://doi.org/10.1007/s12010-019-02974-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-019-02974-7