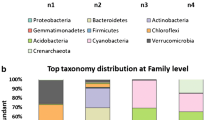
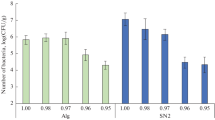
Abstract
The current study deals with the diversity and distribution of culturable cold-adapted bacteria in Himalayan Thajwas glacial soil. The total bacterial count of the seven soil samples collected near glacier snout at different altitudes was investigated through a culture-dependent method. The highest cfu/g (6.8±2 × 107) was recorded at site I, and the lowest cfu/g (4.6±2 × 103) was recorded at site VII on nutrient agar (NA) media. The 31 isolated strains were identified using 16S rRNA gene sequencing. Based on their optimum growth at different temperatures, these bacterial isolates were categorized into two overlap** groups: psychrophiles (optimum growth ≥15°C) and psychrotrophs (optimum growth ≥20°C). Besides, these bacterial strains were screened for their cold-active enzyme activity. Further, the microbial diversity was affected by higher incubation temperature, which reduced the richness due to the selection of psychrotrophic microorganisms. Variation in microbial diversity may be related to soil physicochemical properties since a high positive correlation was observed with water content, total nitrogen and organic carbon. This is the first research to look at microbial communities from the Thajwas Glacier, and it adds to our knowledge of the microbial diversity found in Himalayan cold ecosystems.






Similar content being viewed by others
Data availability
Data about the 16S rRNA gene sequences have been deposited at NCBI GenBank under the accession numbers: MN102377-91, MT478140, MT478142-50, MT477765, MT478875, and MT495407-10.
Code availability
Not applicable.
Abbreviations
- BLAST:
-
Basic Local Alignment Search Tool
- CFU:
-
Colony forming units
- CMC:
-
Carboxymethyl cellulose sodium salt
- DNA:
-
Deoxyribonucleic acid
- MEGA:
-
Molecular Evolutionary Genetic Analysis
- NA:
-
Nutrient agar
- NCBI:
-
National Center for Biotechnology Information
- OGT:
-
Optimum growth temperature
- PAST:
-
PAleontological Statistics
- PCR:
-
Polymerase chain reaction
- R2A:
-
Reasoner’s 2 agar
- rRNA:
-
Ribosomal Ribonucleic acid
- SPSS:
-
Statistical Package for the Social Sciences
- USA:
-
United States of America
- WCA:
-
Whole colony appearance
References
Al-Ghanayem AA, Joseph B (2020) Current prospective in using cold-active enzymes as eco-friendly detergent additive. Appl Microbiol Biotechnol 104:2871–2882. https://doi.org/10.1007/s00253-020-10429-x
Alam SI, Singh L (2002) Proteolytic heterotrophic bacteria of cyanobacterial assemblage from Schirmacher oasis, Antarctica, capable of growing under extreme conditions. Curr Sci 83:1000–1004
Allen MJ, Edberg SC, Reasoner DJ (2004) Heterotrophic plate count bacteria–what is their significance in drinking water? Int J Food Microbiol 92:265–274. https://doi.org/10.1016/j.ijfoodmicro.2003.08.017
Anesio AM, Laybourn-Parry J (2012) Glaciers and ice sheets as a biome. Trends Ecol Evol 27:219–225. https://doi.org/10.1016/j.tree.2011.09.012
Bhatia RK, Ullah S, Hoque MZ et al (2020) Psychrophiles: a source of cold-adapted enzymes for energy efficient biotechnological industrial processes. J Environ Chem Eng 9(1):104607. https://doi.org/10.1016/j.jece.2020.104607
Brankatschk R, Towe S, Kleineidam K et al (2011) Abundances and potential activities of nitrogen cycling microbial communities along a chronosequence of a glacier forefield. ISME J 5(6):1025–37. https://doi.org/10.1038/ismej.2010.184
Bremner JM (1965) Total nitrogen. In: Methods of soil analysis, part 2: chemical and microbiological properties. Wiley, pp 1149–1178
Donhauser J, Frey B (2018) Alpine soil microbial ecology in a changing world. FEMS Microbiol Ecol 94(9):9. https://doi.org/10.1093/femsec/fiy099
Duc L, Noll M, Meier BE et al (2009) High diversity of diazotrophs in the forefield of a receding alpine glacier. Microbial Ecol 57:179–90. https://doi.org/10.1007/s00248-008-9408-5
Farooq S, Nazir R, Ganai SA, Ganai BA (2021) Isolation and characterization of a new cold-active protease from psychrotrophic bacteria of Western Himalayan glacial soil. Sci Reports 11:1–14. https://doi.org/10.1038/s41598-021-92197-w
Fierer N, Allen AS, Schimel JP, Holden PA (2003) Controls on microbial CO2 production: A comparison of surface and subsurface soil horizons. Glob Chang Biol 9:1322–1332. https://doi.org/10.1046/j.1365-2486.2003.00663.x
Fierer N, Bradford MA, Jackson RB (2007) Toward an ecological classification of soil bacteria. Ecology 88:1354–1364. https://doi.org/10.1890/05-1839
Frey B, Rieder SR, Brunner I et al (2010) Weathering-associated bacteria from the Damma glacier forefield: physiological capabilities and impact on granite dissolution. Appl Environ Microbiol 76(14):88–96. https://doi.org/10.1128/AEM.00657-10
Frey B, Buhler L, Schmutz S et al (2013) Molecular characterization of phototrophic microorganisms in the forefield of a receding glacier in the Swiss Alps. Environ Res Lett 8:1–11. https://doi.org/10.1088/1748-9326/8/1/015033
Gangwar P, Alam SI, Bansod S, Singh L (2009) Bacterial diversity of soil samples from the western Himalayas, India. Can J Microbiol 55:564–577. https://doi.org/10.1139/W09-011
Gangwar P, Alam SI, Singh L (2011) Metabolic Characterization of cold active Pseudomonas, Arthrobacter, Bacillus, and Flavobacterium spp. from Western Himalayas. Indian J Microbiol 51:70–75. https://doi.org/10.1007/s12088-011-0092-7
Gardner WH (1986) Water content. pp 493–594. In: A Klute (ed) Methods of soil analysis. Part 1. Physical and mineralogical methods. Agron. Monogr., 9. 2nd edn. ASA, CSSA, and SSSA, Madison
Gensberger ET, Gössl E-M, Antonielli L et al (2015) Effect of different heterotrophic plate count methods on the estimation of the composition of the culturable microbial community. PeerJ 3:e862. https://doi.org/10.7717/PEERJ.862
Gilichinsky D, Vishnivetskaya T, Petrova M et al (2008) Bacteria in permafrost Psychrophiles: from Biodiversity to Biotechnology Margesin R, Schinner F, Marx J-C, Gerday C (eds). Springer-Verlag: Berlin Heidelberg, pp 83–102
Hammer DAT, Ryan PD, Hammer Ø, Harper DAT (2001) Past: Paleontological Statistics Software Package for Education and Data Analysis. Palaeontol. Electron. 4(1):9
Hassan N, Anesio AM, Rafiq M et al (2020) Temperature driven membrane lipid adaptation in glacial psychrophilic bacteria. Front Microbiol 11:824. https://doi.org/10.3389/FMICB.2020.00824
Johnson SS, Hebsgaard MB, Christensen TR et al (2007) Ancient bacteria show evidence of DNA repair. Proc Natl Acad Sci USA 104:14401–14405. https://doi.org/10.1073/pnas.0706787104
Kuddus M, Ramteke PW (2012) Recent developments in production and biotechnological applications of cold-active microbial proteases. Crit Rev Microbiol 38:330–338. https://doi.org/10.3109/1040841X.2012.678477
Kuddus M, Roohi Saima, Ahmad IZ (2012) Cold-active extracellular α-amylase production from novel bacteria Microbacterium foliorum GA2 and Bacillus cereus GA6 isolated from Gangotri glacier, Western Himalaya. J Genet Eng Biotechnol 10:151–159. https://doi.org/10.1016/j.jgeb.2012.03.002
Kumar S, Stecher G, Tamura K et al (2016) MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol 33:1870–1874. https://doi.org/10.1093/molbev/msw054
Kumar S, Suyal DC, Yadav A et al (2019) Microbial diversity and soil physiochemical characteristic of higher altitude. PLoS One 14:1–15. https://doi.org/10.1371/journal.pone.0213844
Lauro FM, Allen MA, Wilkins D et al (2011) Psychrophiles: Genetics, genomics, evolution. In: Extremophiles Handbook. Japan: Springer, pp 865–890.
LaMontagne MG, Schimel JP, Holden PA (2003) Comparison of subsurface and surface soil bacterial communities in California grassland as assessed by terminal restriction fragment length polymorphisms of PCR-amplified 16S rRNA genes. Microb Ecol 46:216–227. https://doi.org/10.1007/s00248-003-1006-y
Liu Y, Yao T, Jiao N et al (2009) Bacterial diversity in the snow over Tibetan Plateau Glaciers. Extremophiles 13:411–423. https://doi.org/10.1007/s00792-009-0227-5
Maharana A, Ray P (2015) A novel cold-active lipase from psychrotolerant Pseudomonas sp. AKM-L5 showed organic solvent resistant and suitable for detergent formulation. J Mol Catal B Enzym 120:173–178. https://doi.org/10.1016/J.MOLCATB.2015.07.005
Margesin R, Collins T (2019) Microbial ecology of the cryosphere (glacial and permafrost habitats): current knowledge. Appl. Microbiol. Biotechnol. 103:2537–2549. https://doi.org/10.1007/s00253-019-09631-3
Miteva VI, Brenchley JE (2005) Detection and isolation of ultrasmall microorganisms from a 120,000-year-old Greenland glacier ice core. Appl Environ Microbiol 71:7806–7818. https://doi.org/10.1128/AEM.71.12.7806-7818.2005
Möller C, Dreyfuss MM (1996) Microfungi from Antarctic lichens, mosses and vascular plants. Mycologia 88:922–933. https://doi.org/10.1080/00275514.1996.12026732
Morita RY (1975) Psychrophilic bacteria. Bacteriological Reviews 39:144–167
Nemergut DR, Anderson SP, Cleveland CC et al (2007) Microbial community succession in an unvegetated, recently deglaciated soil. Microbiol Ecol 53:110–122. https://doi.org/10.1007/s00248-006-9144-7
Nicholson WL, Munakata N, Horneck G et al (2000) Resistance of bacillus endospores to extreme terrestrial and extraterrestrial environments. Microbiol Mol Biol Rev 64:548–572. https://doi.org/10.1128/mmbr.64.3.548-572.2000
Pessi IS, Elias SdeO, Simões FL et al (2012) Functional diversity of microbial communities in soils in the vicinity of Wanda Glacier, Antarctic Peninsula. Microbes Environ 27:200–203. https://doi.org/10.1264/jsme2.ME11311
Pradhan S, Srinivas TNR, Pindi PK et al (2010) Bacterial biodiversity from Roopkund Glacier, Himalayan mountain ranges, India. Extremophiles 14:377–395. https://doi.org/10.1007/s00792-010-0318-3
Priscu JC, Christner BC (2004) Earth’s icy biosphere. In: Bull AT (ed) Microbial diversity and bioprospecting. ASM Press, Washington, DC, pp 130–145
Priscu JC, Christner BC, Foreman CM et al (2007) Biological material in ice cores. In: Encyclopedia of quaternary sciences. Elsevier
Rafiq M, Hayat M, Anesio AM et al (2017) Recovery of metallo-tolerant and antibiotic resistant psychrophilic bacteria from Siachen glacier, Pakistan. PLoS One 12(7):e0178180. https://doi.org/10.1371/journal.pone.0178180
Reasoner DJ (2004) Heterotrophic plate count methodology in the United States. Int J Food Microbiol 92:307–315. https://doi.org/10.1016/j.ijfoodmicro.2003.08.008
Reasoner DJ, Geldreich EE (1985) A new medium for the enumeration and subculture of bacteria from potable water. Applied Environ Microbiol 49(1):1–7
Rime T, Hartmann M, Frey B (2016) Potential sources of microbial colonizers in an initial soil ecosystem after retreat of an alpine glacier. ISME J 10:25–41. https://doi.org/10.1038/ismej.2015.238
Salwoom L, Rahman RNZRA, Salleh AB et al (2019) Isolation, characterisation, and lipase production of a cold-adapted bacterial strain Pseudomonas sp. LSK25 isolated from Signy Island, Antarctica. Molecules 24:1–14. https://doi.org/10.3390/molecules24040715
Schmidt SK, Reed SC, Nemergut DR et al (2008) The earliest stages of ecosystem succession in high-elevation (5000 metres above sea level), recently deglaciated soils. Proc R Soc B 275:2793–2802. https://doi.org/10.1098/rspb.2008.0808
Sherpa MT, Najar IN, Das S, Thakur N (2018) Bacterial diversity in an alpine debris-free and debris-cover accumulation zone Glacier Ice, North Sikkim, India. Indian J Microbiol 58:470–478. https://doi.org/10.1007/s12088-018-0747-8
Shivaji S, Reddy GSN, Aduri RP et al (2004) Bacterial diversity of a soil sample from Schirmacher Oasis, Antarctica. Cell Mol Biol 50:525–536.
Shivaji S, Pratibha MS, Sailaja B et al (2011) Bacterial diversity of soil in the vicinity of Pindari glacier, Himalayan mountain ranges, India, using culturable bacteria and soil 16S rRNA gene clones. Extremophiles 15:1–22. https://doi.org/10.1007/s00792-010-0333-4
Singh P, Singh SM, Dhakephalkar P (2014) Diversity, cold active enzymes and adaptation strategies of bacteria inhabiting glacier cryoconite holes of High Arctic. Extremophiles 18:229–242. https://doi.org/10.1007/s00792-013-0609-6
Srinivas TNR, Singh SM, Pradhan S et al (2011) Comparison of bacterial diversity in proglacial soil from Kafni Glacier, Himalayan Mountain ranges, India, with the bacterial diversity of other glaciers in the world. Extremophiles 15:673–690. https://doi.org/10.1007/s00792-011-0398-8
Steven B, Briggs G, McKay CP et al (2007) Characterization of the microbial diversity in a permafrost sample from the Canadian high Arctic using culture-dependent and culture-independent methods. FEMS Microbiol Ecol 59:513–523. https://doi.org/10.1111/j.1574-6941.2006.00247.x
Sułowicz S, Bondarczuk K, Ignatiuk D et al (2020) Microbial communities from subglacial water of naled ice bodies in the forefield of Werenskioldbreen, Svalbard. Sci Total Environ 723:138025. https://doi.org/10.1016/j.scitotenv.2020.138025
Varin T, Lovejoy C, Jungblut AD et al (2012) Metagenomic analysis of stress genes in microbial mat communities from Antarctica and the High Arctic. Appl Environ Microbiol 78(2):549–559. https://doi.org/10.1128/AEM.06354-11
Walkley A, Black TA (1934) An examination of the Degtjareff method for determining soil organic matter, and a proposed modification of the chromic acid titration method. Soil Sci 37:29–38
Watson ME, Brown JR (1998) pH and Lime Requirement. In: Brown JR (ed) Recommended Chemical Soil Test Procedures for the North Central Region, NCR Publication No. 221, Missouri Agricultural Experiment Station, Columbia, MO, 13-16
Wernicke F, Kampfer P, Dott W (1990) Influence of media and conditions on the isolation of bacterial species from drinking water. Zentralblatt fur Hygiene 190(1–2):26–38
Willerslev E, Hansen AJ, Ronn R et al (2004) Long-term persistence of bacterial DNA. Curr Biol 14(1):9–10. https://doi.org/10.1016/j.cub.2003.12.012
Yergeau E, Kowalchuk GA (2008) Responses of Antarctic soil microbial communities and associated functions to temperature and freeze-thaw cycle frequency. Environ Microbiol 10:2223–2235. https://doi.org/10.1111/j.1462-2920.2008.01644.x
Zhang LM, Wang M, Prosser JI et al (2009) Altitude ammonia-oxidizing bacteria and archaea in soils of Mount Everest. FEMS Microbiol Ecol 70:208–217. https://doi.org/10.1111/j.1574-6941.2009.00775.x
Zhang DC, Brouchkov A, Griva G et al (2013) Isolation and characterization of bacteria from Ancient Siberian Permafrost Sediment. Biology (Basel) 2:85–106. https://doi.org/10.3390/biology2010085
Zumsteg A, Luster J, Göransson H et al (2012) Bacterial, archaeal and fungal succession in the forefield of a receding glacier. Microb Ecol 63:552–64. https://doi.org/10.1007/s00248-011-9991-8
Author information
Authors and Affiliations
Contributions
SF framed the work design, collected, compiled and interpreted the data. Further, conducted experiments, wrote the manuscript and designed the figures. RN designed the research work, evaluated the data and did the revision of the manuscript. BAG and HM did the critical revision of the manuscript. GJD helped in statistical analysis. Both RN and BAG helped in designing the experiments and supervised the overall work. All the authors helped in writing the manuscript and provided critical feedback.
Corresponding author
Ethics declarations
Ethical statement
The authors in the current study carried out no animal or human studies. Authors further confirm that this work is original and has not been published elsewhere, nor is it currently under consideration for publication elsewhere.
Consent to participate
Consent was obtained from all the authors for the submission of the manuscript to the journal.
Consent for publication
The publication of this manuscript has been approved by all the authors.
Conflict of interest
The authors report no conflict of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Farooq, S., Nazir, R., Ganai, B.A. et al. Psychrophilic and psychrotrophic bacterial diversity of Himalayan Thajwas glacial soil, India. Biologia 77, 203–213 (2022). https://doi.org/10.1007/s11756-021-00915-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11756-021-00915-6