Abstract
Main conclusion
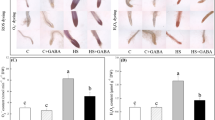
γ-Aminobutyric acid alleviates acid-aluminum toxicity to roots associated with enhanced antioxidant metabolism as well as accumulation and transportation of citric and malic acids.
Abstract
Aluminum (Al) toxicity has become the main limiting factor for crop growth and development in acidic soils and is further being aggravated worldwide due to continuous industrial pollution. The current study was designed to examine effects of GABA priming on alleviating acid-Al toxicity in terms of root growth, antioxidant defense, citrate and malate metabolisms, and extensive metabolites remodeling in roots under acidic conditions. Thirty-seven-day-old cree** bentgrass (Agrostis stolonifera) plants were used as test materials. Roots priming with or without 0.5 mM GABA for 3 days were cultivated in standard nutrient solution for 15 days as control or subjected to nutrient solution containing 5 mM AlCl3·6H2O for 15 days as acid-Al stress treatment. Roots were sampled for determinations of root characteristics, physiological and biochemical parameters, and metabolomics. GABA priming significantly alleviated acid-Al-induced root growth inhibition and oxidative damage, despite it promoted the accumulation of Al in roots. Analysis of metabolomics showed that GABA priming significantly increased accumulations of organic acids, amino acids, carbohydrates, and other metabolites in roots under acid-Al stress. In addition, GABA priming also significantly up-regulated key genes related to accumulation and transportation of malic and citric acids in roots under acid-Al stress. GABA-regulated metabolites participated in tricarboxylic acid cycle, GABA shunt, antioxidant defense system, and lipid metabolism, which played positive roles in reactive oxygen species scavenging, energy conversion, osmotic adjustment, and Al ion chelation in roots.







Similar content being viewed by others
Data availability
Not applicable.
Abbreviations
- ALMT9-like:
-
Al-activated malate transport 9-like
- APX:
-
Ascorbate peroxidase
- CAT:
-
Catalase
- cMDH-like:
-
Malate dehydrogenase, cytoplasmic-like
- CS-like:
-
Citrate synthase-like
- MATEs-like:
-
Multidrug and toxic compound extrusions-like
- MDA:
-
Malondialdehyde
- POD:
-
Peroxidase
- SOD:
-
Superoxide dismutase
- STOP1-like:
-
Sensitive to proton rhizotoxicity 1-like
References
Afzal S, Chaudhary N, Singh NK (2021) Role of soluble sugars in metabolism and sensing under abiotic stress. In: Aftab T, Hakeem KR (eds) Plant growth regulators. Springer International Publishing, Cham, 305–334. https://doi.org/10.1007/978-3-030-61153-8_14
Anoop VM, Basu U, McCammon MT, McAlister-Henn L, Taylor GJ (2003) Modulation of citrate metabolism alters aluminum tolerance in yeast and transgenic canola overexpressing a mitochondrial citrate synthase. Plant Physiol 132(4):2205–2217. https://doi.org/10.1104/pp.103.023903
Ansari MI, Jalil SU, Ansari SA, Hasanuzzaman M (2021) GABA shunt: a key-player in mitigation of ROS during stress. Plant Growth Regul 94(2):131–149. https://doi.org/10.1007/s10725-021-00710-y
Bhutto L, Osborne C, Quick W (2023) Osmotic adjustment and metabolic changes under drought stress conditions in wheat (Triticum aestivum L.) genotypes. Pak J Bot 55(3):915–923. https://doi.org/10.30848/PJB2023-3(22)
Blum A, Ebercon A (1981) Cell membrane stability as a measure of drought and heat tolerance in wheat. Crop Sci 21(1):43–47. https://doi.org/10.2135/cropsci1981.0011183x002100010013x
Bouche N, Fromm H (2004) GABA in plants: just a metabolite? Trends Plant Sci 9(3):110–115. https://doi.org/10.1016/j.tplants.2004.01.006
Çekiç F (2018) Exogenous GABA stimulates endogenous GABA and phenolic acid contents in tomato plants under salt stress. Celal Bayar Univ J Sci 14(1):61–64. https://doi.org/10.18466/cbayarfbe.348935
Chance B, Maehly A (1955) Assay of catalases and peroxidases. Methods Enzymol 2:764–775
Chauhan DK, Yadav V, Vaculik M, Gassmann W, Pike S, Arif N, Singh VP, Deshmukh R, Sahi S, Tripathi DK (2021) Aluminum toxicity and aluminum stress-induced physiological tolerance responses in higher plants. Crit Rev Biotechnol 41(5):715–730. https://doi.org/10.1080/07388551.2021.1874282
Chen W, Tang L, Wang J, Zhu H, ** J, Yang J, Fan W (2022) Research advances in the mutual mechanisms regulating response of plant roots to phosphate deficiency and aluminum toxicity. Int J Mol Sci 23(3):1137. https://doi.org/10.3390/ijms23031137
Chinopoulos C (2020) Acute sources of mitochondrial NAD+ during respiratory chain dysfunction. Exp Neurol 327:113218. https://doi.org/10.1016/j.expneurol.2020.113218
Choudhury S, Sharma P (2014) Aluminum stress inhibits root growth and alters physiological and metabolic responses in chickpea (Cicer arietinum L.). Plant Physiol Biochem 85:63–70. https://doi.org/10.1016/j.plaphy.2014.10.012
Dhindsa RS, Plumb-Dhindsa P, Thorpe TA (1981) Leaf senescence: correlated with increased levels of membrane permeability and lipid peroxidation, and decreased levels of superoxide dismutase and catalase. J Exp Bot 32(1):93–101
Dumanović J, Nepovimova E, Natić M, Kuča K, Jaćević V (2021) The significance of reactive oxygen species and antioxidant defense system in plants: a concise overview. Front Plant Sci 11:552969. https://doi.org/10.3389/fpls.2020.552969
Elstner EF, Heupel A (1976) Inhibition of nitrite formation from hydroxylammoniumchloride: a simple assay for superoxide dismutase. Anal Biochem 70(2):616–620
Fan J, Chen K, Xu J, Khaldun A, Chen Y, Chen L, Yan X (2022) Physiological effects induced by aluminium and fluoride stress in tall fescue (Festuca arundinacea Schreb). Ecotoxicol Environ Saf 231:113192. https://doi.org/10.1016/j.ecoenv.2022.113192
Feng D, Gao Q, Sun X, Ning S, Qi N, Hua Z, Tang J (2023) Effects of foliage-applied exogenous γ-aminobutyric acid on seedling growth of two rice varieties under salt stress. PLoS ONE 18(2):e0281846. https://doi.org/10.1371/journal.pone.0281846
Fontana JE, Wang G, Sun R, Xue H, Li Q, Liu J, Davis KE, Thornburg TE, Zhang B, Zhang Z, Pan X (2020) Impact of potassium deficiency on cotton growth, development and potential microRNA-mediated mechanism. Plant Physiol Biochem 153:72–80. https://doi.org/10.1016/j.plaphy.2020.05.006
Giannakoula A, Moustakas M, Mylona P, Papadakis I, Yupsanis T (2008) Aluminum tolerance in maize is correlated with increased levels of mineral nutrients, carbohydrates and proline, and decreased levels of lipid peroxidation and Al accumulation. J Plant Physiol 165(4):385–396. https://doi.org/10.1016/j.jplph.2007.01.014
Giannopolitis CN, Ries SK (1977) Superoxide dismutases: I. Occurrence in higher plants. Plant Physiol 59(2):309–314. https://doi.org/10.1104/pp.59.2.309
Gurmessa B (2021) Soil acidity challenges and the significance of liming and organic amendments in tropical agricultural lands with reference to Ethiopia. Environ Dev Sustain 23(1):77–99. https://doi.org/10.1007/s10668-020-00615-2
Hajiboland R, Panda CK, Lastochkina O, Gavassi MA, Habermann G, Pereira JF (2023) Aluminum toxicity in plants: present and future. J Plant Growth Regul 42(7):3967–3999. https://doi.org/10.1007/s00344-022-10866-0
Hao J, Peng A, Li Y, Zuo H, Li P, Wang J, Yu K, Liu C, Zhao S, Wan X, Pittman JK, Zhao J (2022) Tea plant roots respond to aluminum-induced mineral nutrient imbalances by transcriptional regulation of multiple cation and anion transporters. BMC Plant Biol 22(1):203. https://doi.org/10.1186/s12870-022-03570-4
Heinemann B, Hildebrandt TM (2021) The role of amino acid metabolism in signaling and metabolic adaptation to stress-induced energy deficiency in plants. J Exp Bot 72(13):4634–4645. https://doi.org/10.1093/jxb/erab182
Hidayah AN, Yahya S, Sopandie D (2020) The tolerance of oil palm (Elaeis guineensis) seedlings to Al stress is enhanced by citric acid and natural peat water. Biodiversitas 21(10):4850–4858. https://doi.org/10.13057/BIODIV/D211051
Hijaz F, Killiny N (2019) Exogenous GABA is quickly metabolized to succinic acid and fed into the plant TCA cycle. Plant Signal Behav 14(3):e1573096. https://doi.org/10.1080/15592324.2019.1573096
Hoagland DR, Arnon DI (1950) The water-culture method for growing plants without soil. Calif Agric Exp Stat Circ. https://doi.org/10.1097/00010694-193910000-00022. (2nd edn)
Hu L, Zhou K, Ren G, Yang S, Liu Y, Zhang Z, Li Y, Gong X, Ma F (2020) Myo-inositol mediates reactive oxygen species-induced programmed cell death via salicylic acid-dependent and ethylene-dependent pathways in apple. Hortic Res 7:138–150. https://doi.org/10.1038/s41438-020-00357-2
Huang B (2021) Grass research for a productive, healthy and sustainable society. Grass Res 1(1):1–2. https://doi.org/10.48130/GR-2021-0001
Hui NY, Liu P, Wang ZY, Chen WR, Xu GD (2011) The effect of aluminum treatments on the root growth and cell ultrastructure of two soybean genotypes. Crop Prot 30(3):323–328. https://doi.org/10.1016/j.cropro.2010.11.024
Igamberdiev AU, Eprintsev AT (2016) Organic acids: the pools of fixed carbon involved in redox regulation and energy balance in higher plants. Front Plant Sci 7:205704. https://doi.org/10.3389/fpls.2016.01042
Ito H, Kobayashi Y, Yamamoto YY, Koyama H (2019) Characterization of NtSTOP1-regulating genes in tobacco under aluminum stress. Soil Sci Plant Nutr 65(3):251–258. https://doi.org/10.1080/00380768.2019.1603064
Iuchi S, Koyama H, Iuchi A, Kobayashi Y, Kitabayashi S, Kobayashi Y, Ikka T, Hirayama T, Shinozaki K, Kobayashi M (2007) Zinc finger protein STOP1 is critical for proton tolerance in Arabidopsis and coregulates a key gene in aluminum tolerance. Proc Natl Acad Sci USA 104(23):9900–9905. https://doi.org/10.1073/pnas.0700117104
Jiang F, Wang T, Wang Y, Kochian LV, Chen F, Liu J (2017) Identification and characterization of suppressor mutants of stop1. BMC Plant Biol 17(1):128. https://doi.org/10.1186/s12870-017-1079-2
Jisha K, Vijayakumari K, Puthur JT (2013) Seed priming for abiotic stress tolerance: an overview. Acta Physiol Plant 35(5):1381–1396. https://doi.org/10.1007/s11738-012-1186-5
Jouve L, Hoffmann L, Hausman J-F (2004) Polyamine, carbohydrate, and proline content changes during salt stress exposure of aspen (Populus tremula L.): involvement of oxidation and osmoregulation metabolism. Plant Biol 6(1):74–80. https://doi.org/10.1055/s-2003-44687
Khan A, McNeilly T, Collins J (2000) Accumulation of amino acids, proline, and carbohydrates in response to aluminum and manganese stress in maize. J Plant Nutr Soil 23(9):1303–1314. https://doi.org/10.1080/01904160009382101
Khan N, Ali S, Zandi P, Mehmood A, Ullah S, Ikram M, Ismail I, Shahid M, Babar M (2020) Role of sugars, amino acids and organic acids in improving plant abiotic stress tolerance. Pak J Bot 52(2):355–363. https://doi.org/10.30848/PJB2020-2(24)
Kovermann P, Meyer S, Hörtensteiner S, Picco C, Scholz-Starke J, Ravera S, Lee Y, Martinoia E (2007) The Arabidopsis vacuolar malate channel is a member of the ALMT family. Plant J 52(6):1169–1180. https://doi.org/10.1111/j.1365-313x.2007.03367.x
Li Z, Yu J, Peng Y, Huang B (2016) Metabolic pathways regulated by γ-aminobutyric acid (GABA) contributing to heat tolerance in cree** bentgrass (Agrostis stolonifera). Sci Rep 6(1):30338. https://doi.org/10.1038/srep30338
Li Y, Fan Y, Ma Y, Zhang Z, Yue H, Wang L, Li J, Jiao Y (2017a) Effects of exogenous γ-aminobutyric acid (GABA) on photosynthesis and antioxidant system in pepper (Capsicum annuum L.) seedlings under low light stress. J Plant Growth Regul 36(2):436–449. https://doi.org/10.1007/s00344-016-9652-8
Li Z, Yu J, Peng Y, Huang B (2017b) Metabolic pathways regulated by abscisic acid, salicylic acid and γ-aminobutyric acid in association with improved drought tolerance in cree** bentgrass (Agrostis stolonifera). Physiol Plant 159(1):42–58. https://doi.org/10.1111/ppl.12483
Li Y, Liu B, Peng Y, Liu C, Zhang X, Zhang Z, Liang W, Ma F, Li C (2020a) Exogenous GABA alleviates alkaline stress in Malus hupehensis by regulating the accumulation of organic acids. Sci Hortic 261:108982. https://doi.org/10.1016/j.scienta.2019.108982
Li Z, Cheng B, Peng Y, Zhang Y (2020b) Adaptability to abiotic stress regulated by γ-aminobutyric acid in relation to alterations of endogenous polyamines and organic metabolites in cree** bentgrass. Plant Physiol Biochem 157:185–194. https://doi.org/10.1016/j.plaphy.2020.10.025
Li Z, Fu J, Shi D, Peng Y (2020c) Myo-inositol enhances drought tolerance in cree** bentgrass through alteration of osmotic adjustment, photosynthesis, and antioxidant defense. Crop Sci 60(4):2149–2158. https://doi.org/10.1002/csc2.20186
Li Z, Zhou M, Zeng W, Zhang Y, Liu L, Liu W, Peng Y (2023) Root metabolites remodeling regulated by γ-aminobutyric acid (GABA) improves adaptability to high temperature in cree** bentgrass. Plant Soil. https://doi.org/10.1007/s11104-023-05905-y
Liang Y, Bai T, Liu B, Yu W, Teng W (2022) Different antioxidant regulation mechanisms in response to aluminum-induced oxidative stress in Eucalyptus species. Ecotoxicol Environ Saf 241:113748. https://doi.org/10.1016/j.ecoenv.2022.113748
Liu J, Magalhaes JV, Shaff J, Kochian LV (2009) Aluminum-activated citrate and malate transporters from the MATE and ALMT families function independently to confer Arabidopsis aluminum tolerance. Plant J 57(3):389–399. https://doi.org/10.1111/j.1365-313X.2008.03696.x
Liu J, Li Y, Wang W, Gai J, Li Y (2016) Genome-wide analysis of MATE transporters and expression patterns of a subgroup of MATE genes in response to aluminum toxicity in soybean. BMC Genom 17(1):223. https://doi.org/10.1186/s12864-016-2559-8
Liu W, Xu F, Lv T, Zhou W, Chen Y, ** C, Lu L, Lin X (2018) Spatial responses of antioxidative system to aluminum stress in roots of wheat (Triticum aestivum L.) plants. Sci Total Environ 627:462–469. https://doi.org/10.1016/j.scitotenv.2018.01.021
Liu C, Liu Y, Wang S, Ke Q, Yin L, Deng X, Feng B (2020) Arabidopsis mgd mutants with reduced monogalactosyldiacylglycerol contents are hypersensitive to aluminium stress. Ecotoxicol Environ Saf 203:110999. https://doi.org/10.1016/j.ecoenv.2020.110999
Liu L, Wu X, Sun W, Yu X, Demura T, Li D, Zhuge Q (2021) Galactinol synthase confers salt-stress tolerance by regulating the synthesis of galactinol and raffinose family oligosaccharides in poplar. Ind Crop Prod 165:113432. https://doi.org/10.1016/j.indcrop.2021.113432
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25(4):402–408. https://doi.org/10.1006/meth.2001.1262
Magalhaes JV, Piñeros MA, Maciel LS, Kochian LV (2018) Emerging pleiotropic mechanisms underlying aluminum resistance and phosphorus acquisition on acidic soils. Front Plant Sci 9:1420. https://doi.org/10.3389/fpls.2018.01420
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol 22(5):867–880
Nehela Y, Killiny N (2019) ‘Candidatus Liberibacter asiaticus’ and its vector, Diaphorina citri, augment the tricarboxylic acid cycle of their host via the γ-aminobutyric acid shunt and polyamines pathway. Mol Plant Microbe Interact 32(4):413–427. https://doi.org/10.1094/mpmi-09-18-0238-r
Ofoe R, Thomas RH, Asiedu SK, Wang-Pruski G, Fofana B, Abbey L (2023) Aluminum in plant: benefits, toxicity and tolerance mechanisms. Front Plant Sci 13:1085998. https://doi.org/10.3389/fpls.2022.1085998
Ohyama Y, Ito H, Kobayashi Y, Ikka T, Morita A, Kobayashi M, Imaizumi R, Aoki T, Komatsu K, Sakata Y, Iuchi S, Koyama H (2013) Characterization of AtSTOP1 orthologous genes in tobacco and other plant species. Plant Physiol 162(4):1937–1946. https://doi.org/10.1104/pp.113.218958
Palmer AJ, Baker A, Muench SP (2016) The varied functions of aluminium-activated malate transporters-much more than aluminium resistance. Biochem Soc Trans 44(3):856–862. https://doi.org/10.1042/bst20160027
Prasad M (2003) Phytoremediation of metal-polluted ecosystems: hype for commercialization. Russ J Plant Physiol 50(5):686–701. https://doi.org/10.1023/A:1025604627496
Qiu Y, Su M, Liu Y, Chen M, Gu J, Zhang J, Jia W (2007) Application of ethyl chloroformate derivatization for gas chromatography-mass spectrometry based metabonomic profiling. Anal Chim Acta 583(2):277–283. https://doi.org/10.1016/j.aca.2006.10.025
Qu X, Zhou J, Masabni J, Yuan J (2020) Phosphorus relieves aluminum toxicity in oil tea seedlings by regulating the metabolic profiling in the roots. Plant Physiol Biochem 152:12–22. https://doi.org/10.1016/j.plaphy.2020.04.030
Ramesh SA, Tyerman SD, Xu B, Bose J, Kaur S, Conn V, Domingos P, Ullah S, Wege S, Shabala S, Feijó JA, Ryan PR, Gilliham M (2015) GABA signalling modulates plant growth by directly regulating the activity of plant-specific anion transporters. Nat Commun 6(1):7879. https://doi.org/10.1038/ncomms8879
Reddy AS, Shad Ali G (2011) Plant serine/arginine-rich proteins: roles in precursor messenger RNA splicing, plant development, and stress responses. Wires RNA 2(6):875–889. https://doi.org/10.1002/wrna.98
Rizhsky L, Liang H, Shuman J, Shulaev V, Davletova S, Mittler R (2004) When defense pathways collide. The response of Arabidopsis to a combination of drought and heat stress. Plant Physiol 134(4):1683–1696. https://doi.org/10.1104/pp.103.033431
Roessner U, Wagner C, Kopka J, Trethewey RN, Willmitzer L (2000) Simultaneous analysis of metabolites in potato tuber by gas chromatography–mass spectrometry. Plant J 23(1):131–142. https://doi.org/10.1046/j.1365-313x.2000.00774.x
Sheteiwy MS, Shao H, Qi W, Hamoud YA, Shaghaleh H, Khan NU, Yang R, Tang B (2019) GABA-alleviated oxidative injury induced by salinity, osmotic stress and their combination by regulating cellular and molecular signals in rice. Int J Mol Sci 20(22):5709. https://doi.org/10.3390/ijms20225709
Shi SQ, Shi Z, Jiang ZP, Qi LW, Sun XM, Li CX, Liu JF, **ao WF, Gong S (2010) Effects of exogenous GABA on gene expression of Caragana intermedia roots under NaCl stress: regulatory roles for H2O2 and ethylene production. Plant Cell Environ 33(2):149–162. https://doi.org/10.1111/j.1365-3040.2009.02065.x
Silva CO, Brito DS, da Silva AA, do Rosário Rosa V, Santos MFS, de Souza GA, Azevedo AA, Dal-Bianco M, Oliveira JA, Ribeiro C (2020) Differential accumulation of aluminum in root tips of soybean seedlings. Braz J Bot 43:99–107. https://doi.org/10.1007/s40415-020-00593-9
Silva-Navas J, Salvador N, Pozo J, Benito C, Gallego FJ (2021) The rye transcription factor ScSTOP1 regulates the tolerance to aluminum by activating the ALMT1 transporter. Plant Sci 310(3):110951. https://doi.org/10.1016/j.plantsci.2021.110951
Sun C, Liu L, Yu Y, Liu W, Lu L, ** C, Lin X (2015) Nitric oxide alleviates aluminum-induced oxidative damage through regulating the ascorbate-glutathione cycle in roots of wheat. J Integr Plant Biol 57(6):550–561. https://doi.org/10.1111/jipb.12298
Taji T, Ohsumi C, Iuchi S, Seki M, Kasuga M, Kobayashi M, Yamaguchi-Shinozaki K, Shinozaki K (2002) Important roles of drought- and cold-inducible genes for galactinol synthase in stress tolerance in Arabidopsis thaliana. Plant J 29(4):417–426. https://doi.org/10.1046/j.0960-7412.2001.01227.x
Tan M, Hassan MJ, Peng Y, Feng G, Huang L, Liu L, Liu W, Han L, Li Z (2022) Polyamines metabolism interacts with γ-aminobutyric acid, proline and nitrogen metabolisms to affect drought tolerance of cree** bentgrass. Int J Mol Sci 23(5):2779. https://doi.org/10.3390/ijms23052779
Tesfaye M, Temple SJ, Allan DL, Vance CP, Samac DA (2001) Overexpression of malate dehydrogenase in transgenic alfalfa enhances organic acid synthesis and confers tolerance to aluminum. Plant Physiol 127(4):1836–1844. https://doi.org/10.1104/pp.010376
Tomaz T, Bagard M, Pracharoenwattana I, Lindén P, Lee CP, Carroll AJ, Ströher E, Smith SM, Gardeström P, Millar AH (2010) Mitochondrial malate dehydrogenase lowers leaf respiration and alters photorespiration and plant growth in Arabidopsis. Plant Physiol 154(3):1143–1157. https://doi.org/10.1104/pp.110.161612
Uchida A, Jagendorf AT, Hibino T, Takabe T, Takabe T (2002) Effects of hydrogen peroxide and nitric oxide on both salt and heat stress tolerance in rice. Plant Sci 163(3):515–523. https://doi.org/10.1016/s0168-9452(02)00159-0
Valluru R, Van den Ende W (2011) Myo-inositol and beyond-emerging networks under stress. Plant Sci 181(4):387–400. https://doi.org/10.1016/j.plantsci.2011.07.009
Vijayakumari K, Jisha K, Puthur JT (2016) GABA/BABA priming: a means for enhancing abiotic stress tolerance potential of plants with less energy investments on defence cache. Acta Physiol Plant 38:230. https://doi.org/10.1007/s11738-016-2254-z
Wang Y, Xu H, Kou J, Shi L, Zhang C, Xu F (2013) Dual effects of transgenic Brassica napus overexpressing CS gene on tolerances to aluminum toxicity and phosphorus deficiency. Plant Soil 362(1):231–246. https://doi.org/10.1007/s11104-012-1289-1
Wang Q, Sun H, Dong Q, Sun T, ** Z, Hao Y, Yao Y (2016) The enhancement of tolerance to salt and cold stresses by modifying the redox state and salicylic acid content via the cytosolic malate dehydrogenase gene in transgenic apple plants. Plant Biotechnol J 14(10):1986–1997. https://doi.org/10.1111/pbi.12556
Wang Y, Gu W, Meng Y, **e T, Li L, Li J, Wei S (2017) γ-Aminobutyric acid imparts partial protection from salt stress injury to maize seedlings by improving photosynthesis and upregulating osmoprotectants and antioxidants. Sci Rep 7:43609. https://doi.org/10.1038/srep43609
Wang P, Dong Y, Zhu L, Hao Z, Hu L, Hu X, Wang G, Cheng T, Shi J, Chen J (2021) The role of γ-aminobutyric acid in aluminum stress tolerance in a woody plant, Liriodendron chinense× tulipifera. Hortic Res 8:80. https://doi.org/10.1038/s41438-021-00517-y
Wang P, Zhou S, Li A, **e L (2022) Influence of aluminum at low pH on the rhizosphere processes of Masson pine (Pinus massoniana Lamb). Plant Growth Regul 97:499–510. https://doi.org/10.1007/s10725-022-00816-x
Wang C, Bian C, Li J, Han L, Guo D, Wang T, Sun Z, Ma C, Liu X, Tian Y, Zheng X (2023) Melatonin promotes Al3+ compartmentalization via H+ transport and ion gradients in Malus hupehensis. Plant Physiol 193(1):821–839. https://doi.org/10.1093/plphys/kiad339
Yamamoto Y, Kobayashi Y, Devi SR, Rikiishi S, Matsumoto H (2003) Oxidative stress triggered by aluminum in plant roots. Plant Soil 101:39–243. https://doi.org/10.1023/a:1026127803156
Yao YX, Dong QL, Zhai H, You CX, Hao YJ (2011) The functions of an apple cytosolic malate dehydrogenase gene in growth and tolerance to cold and salt stresses. Plant Physiol Biochem 49(3):257–264. https://doi.org/10.1016/j.plaphy.2010.12.009
Yao H, Zhang S, Zhou W, Liu Y, Liu Y, Wu Y (2020) The effects of exogenous malic acid in relieving aluminum toxicity in Pinus massoniana. Int J Phytoremed 22(6):669–678. https://doi.org/10.1080/15226514.2019.1707162
Ye J, Wang X, Hu T, Zhang F, Wang B, Li C, Yang T, Li H, Lu Y, Giovannoni JJ, Zhang Y, Ye Z (2017) An indel in the promoter of Al-activated malate transporter 9 selected during tomato domestication determines fruit malate contents and aluminum tolerance. Plant Cell 29(9):2249–2268. https://doi.org/10.1105/tpc.17.00211
Yu L, Sun J, Guo S, Yan J, Zhu W (2012) Antioxidant enzyme activities in root tips of Vigna unguiculata L. seedlings under aluminum stress. Acta Botan Boreali-Occiden Sin 32(11):2299–2304. https://doi.org/10.3969/j.issn.1000-4025.2012.11.022
Zhang Q, Wang M, Hu J, Wang W, Fu X, Liu J (2015) PtrABF of Poncirus trifoliata functions in dehydration tolerance by reducing stomatal density and maintaining reactive oxygen species homeostasis. J Exp Bot 66(19):5911–5927. https://doi.org/10.1093/jxb/erv301
Zheng SJ, Ma JF, Matsumoto H (1998) Continuous secretion of organic acids is related to aluminium resistance during relatively long-term exposure to aluminium stress. Physiol Plant 103(2):209–214. https://doi.org/10.1034/j.1399-3054.1998.1030208.x
Zheng Y, Cabassa-Hourton C, Planchais S, Lebreton S, Savouré A (2021) The proline cycle as an eukaryotic redox valve. J Exp Bot 72(20):6856–6866. https://doi.org/10.1093/jxb/erab361
Zhou Y, Yang Z, Gong L, Liu R, Sun H, You J (2018) Molecular characterization of GmSTOP1 homologs in soybean under Al and proton stress. Plant Soil 427:213–230. https://doi.org/10.1007/s11104-018-3645-2
Zhou M, Yuan Y, Lin J, Lin L, Zhou J, Li Z (2023) γ-Aminobutyric acid priming alleviates acid-aluminum toxicity to cree** bentgrass by regulating metabolic homeostasis. Int J Mol Sci 24(18):14309. https://doi.org/10.3390/ijms241814309
Zhu CQ, Cao XC, Bai ZG, Zhu LF, Hu WJ, Hu AY, Abliz B, Zhong C, Liang QD, Huang J, Zhang JH, ** QY (2019) Putrescine alleviates aluminum toxicity in rice (Oryza sativa) by reducing cell wall Al contents in an ethylene-dependent manner. Physiol Plant 167(4):471–487. https://doi.org/10.1111/ppl.12961
Acknowledgements
This research was supported by the National Natural Science Foundation of China (32171684).
Author information
Authors and Affiliations
Contributions
Min Zhou: data curation, investigation, formal analysis, and writing—original draft. Cheng Huang: methodology and investigation. Junnan Lin, Yan Yuan, Long Lin, and Jianzhen Zhou: investigation and material cultivation. Zhou Li: conceptualization, technical guidance, writing—review and editing, methodology, formal analysis.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Additional information
Communicated by Dorothea Bartels.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
425_2024_4461_MOESM1_ESM.docx
Supplementary Information Table S1 Details of primer sequences of tested genes. Table S2 Relative retention time and mass to charge ratios of 70 identified metabolites in cree** bentgrass. Figure S1-4. Contents of different amino acids, organic acids, sugars, and other metabolites in cree** bentgrass roots under control condition and acid-aluminum stress (DOCX 500 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhou, M., Huang, C., Lin, J. et al. γ-Aminobutyric acid (GABA) priming alleviates acid-aluminum toxicity to roots of cree** bentgrass via enhancements in antioxidant defense and organic metabolites remodeling. Planta 260, 33 (2024). https://doi.org/10.1007/s00425-024-04461-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00425-024-04461-8