Abstract
Semi-hydrogenation of acetylene in excess ethylene is a key industrial process for ethylene purification. Supported Pd catalysts have attracted most attention due to their superior intrinsic activity but often suffer from low selectivity. Pd single-atom catalysts (SACs) are promising to significantly improve the selectivity, but the activity needs to be improved and the feasible preparation of Pd SACs remains a grand challenge. Here, we report a simple strategy to construct Pd1/TiO2 SACs by selectively encapsulating the co-existed small amount of Pd nanoclusters/nanoparticles based on their different strong metal-support interaction (SMSI) occurrence conditions. In addition, photo-thermo catalysis has been applied to this process where a much-improved catalytic activity was obtained. Detailed characterization combined with DFT calculation suggests that photo-induced electrons transferred from TiO2 to the adjacent Pd atoms facilitate the activation of acetylene. This work offers an opportunity to develop highly stable Pd SACs for efficient catalytic semi-hydrogenation process.
Similar content being viewed by others
Introduction
Ethylene, one of the basic building blocks to produce plastic and key chemicals, is predominantly manufactured from steam cracking of hydrocarbons thus usually concomitant with small amount of co-produced acetylene. The co-existed acetylene can severely poison the downstream catalyst for ethylene transformation, thereby has to be diminished to an acceptable level (often <5 ppm)1,2. Diverse methods have been developed to eliminate the acetylene impurity among which the electrocatalysis has been proven a green chemistry approach3,4. For example, Shi et al. reported a room-temperature electrochemical reduction strategy of acetylene over a layered double hydroxide (LDH)-derived Cu catalyst, which manifested high catalytic performance but suffers from unaddressed issues for large-scale applications, such as the low cell energy efficiency3. On the other hand, thermocatalytic semi-hydrogenation of acetylene into ethylene seems more efficient, and has been extensively applied in industry for decades. Among various catalysts explored, supported Pd catalysts have attracted most attention on account of their superior intrinsic activity. Unfortunately, the low selectivity, especially at the full conversion of acetylene, has long been a serious concern. Several strategies based on the “active site isolation” concept, such as selective poisoning/covering special Pd sites (Lindlar catalysts5) or forming Pd-M alloy/intermetallic compounds (industrially used Ag-Pd/Al2O3 catalysts)6,7,8,9,10,11 to weaken the adsorption of ethylene have been frequently used to improve the selectivity12,13,14,15,16,17,18,19,20,21, which are, nevertheless, often at the cost of activity loss due to the presence of substantial inaccessible Pd sites.
Single-atom catalysts (SACs) have attracted rapid growing interests as a new frontier in heterogeneous catalysis field22,23. In SACs, isolated metal atoms are spatially separated and uniformly distributed on the surface of the support, perfectly meeting the “active-site isolation” concept while simultaneously maximizing the metal utilization efficiency. Hence, SACs have been regarded as an ideal candidate for semi-hydrogenation of alkyne and have shown promising catalytic performance24,25,26,27,28,29. Unfortunately, SACs are generally less effective for H2 activation, giving rise to a depressed hydrogenation activity30. Moreover, to maintain the isolated dispersion and good stability of SACs, a very low metal loading is often used. This is particularly true for Pd-based SACs31. All these render the semi-hydrogenation of acetylene on Pd SACs working currently at elevated temperatures. Therefore, to meet industrial application, it is necessary to develop stable and efficient Pd SACs meanwhile lowering the working temperature.
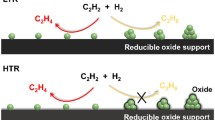
Strong metal–support interaction (SMSI), a topic being extensively studied for more than 40 years in heterogeneous catalysis area32,33, has sparked renewed interests due to their potential in modifying catalyst performance, and especially in stabilizing catalysts34,35,36,37,38,39,40. Recently, we found that isolated Pt atoms supported on TiO2 can manifest classical SMSI34 but at a much higher reduction temperature. A feature of this finding is that the co-existed nanoparticles (NPs) can be selectively encapsulated while single atoms keep exposed through reduction at suitable temperatures. This finding might be extended to TiO2 supported other metal catalysts thus providing a new strategy to construct stable SACs. On the other hand, Photo-thermo catalysis is an emerging sub-discipline that involves the integration of thermo- and photocatalytic processes, which is distinct from the traditional thermo-catalysis because photogenerated carriers can directly transfer into the orbitals of adsorbed molecules to promote their desorption, dissociation, or activation thus trigger the chemical reaction, giving rise to a totally different reaction pathway41,42,43,44. Recent pioneering studies have demonstrated that the coupling of thermo- and photocatalytic processes overcomes the low activity in photocatalysis and high reaction barrier in thermocatalysis, thus offering a promising strategy to promote the activity and/or selectivity for various meaningful reactions, such as hydrogenation, oxidation, CO2 reduction, Fischer-Tropsch synthesis, water–gas shift reaction45,46,47,48,49,50,51,52. Despite of these great progress, whether photo-thermocatalysis is possible to boost semi-hydrogenation of acetylene still remains inconclusive. Few studies related to photocatalysis of selective hydrogenation for nitrobenzene53, benzaldehyde54, and alkynyl group55,56,57,58,59 were reported, but the related works for photo-thermocatalytic acetylene semi-hydrogenation are limited. Swearer et al. firstly used Pd NPs and aluminum nanocrystals (AlNC) to construct a heterometallic antenna-reactor complexes photocatalyst for semi-hydrogenation of acetylene but with a low product yield60. The other one is working at relatively high temperature by converting photo into heat rather than an integration of photo-thermo catalysis process at lower temperature28.
Herein, we report a simple yet general strategy to improve the selectivity of TiO2 supported Pd catalysts prepared by a variety of methods via selectively encapsulating the co-existed small amount of Pd nanoclusters/nanoparticles (NPs) due to their different SMSI occurrence temperatures. In addition, on account of the superior photocatalysis of TiO2 support, a much-improved catalytic activity was obtained by integrating photo-thermo catalysis and a dramatically decreased working temperature of as low as 70 °C was realized. Detailed studies reveal that photo-induced electrons transferred from TiO2 to the adjacent Pd atoms facilitate the activation of acetylene and thus benefit the photo-thermo catalytic semi-hydrogenation reaction.
Results
Synthesis and structural characterization of Pd/TiO2
The Pd/TiO2 catalyst was firstly synthesized by ball milling based on the so called “precursor-dilution” strategy61,62,63,64 to obtain better dispersion but we will propose later that much more practical methods such as strong electrostatic adsorption (SEA) and even impregnation methods also work well. The obtained Pd/TiO2 was reduced at 200 °C and 600 °C, denoted as Pd/TiO2-200H, and Pd/TiO2-600H, respectively. For comparison, the Pd/TiO2-600H catalyst was re-oxidized by 10 vol% O2 at 300 °C, denoted as Pd/TiO2-600H-O300. In addition, pure rutile supported catalysts were also prepared and tested in similar procedures.
The BET specific surface area was measured to be about 70 m2 g−1 by N2 physical adsorption–desorption process, and the incorporation of Pd did not change the surface area much, Supplementary Fig. 1. As shown in Supplementary Fig. 2, the X-ray diffraction (XRD) spectrum of the synthesized TiO2 support displays typical patterns of both anatase and rutile, suggesting a mixture structure. After loading of Pd, and even after reduction at different temperatures and re-oxidation, there is no obvious structure change of the TiO2 support as evidenced by the similar diffraction patterns of various catalysts to that of TiO2 support. In addition, no any diffraction pattern associated with Pd species is observed, suggesting either the Pd is highly dispersed or the Pd loading is too low to be detected. The high dispersion of Pd was further examined by aberration-corrected scanning transmission electron microscopy (AC-STEM). High-magnification high-angle annular dark-field (HAADF) STEM images reveal the presence of relatively high density of Pd single atoms on all catalysts, Fig. 1a–d. Meanwhile some other HAADF-STEM images indicate the presence of small portion of Pd NPs, Supplementary Fig. 3. It stressed the great difficulty in fabricating “absolute” Pd SACs (presence of only isolated single atoms without any clusters/NPs) even with such an effective ball-milling method61,62,63,64. To our knowledge, so far the Pd SACs with relatively high metal loading on non-carbon supports have been rarely reported65,66,67.
a–d AC-HAADF-STEM images of a Pd/TiO2, b Pd/TiO2-200H, c Pd/TiO2-600H, and d Pd/TiO2-600H-O300; Pd single atoms are highlighted in yellow circles. e, f DRIFT spectra of CO adsorption on Pd/TiO2-200H, Pd/TiO2-600H, and Pd/TiO2-600H-O300 e at CO saturation adsorption and f upon He purging for 2.5 min at room temperature. g XANES spectra of different Pt/TiO2 catalysts at Pd K-edge absorption edge.
Very recently, we discovered that Pt single atoms on TiO2 can manifest classical SMSI upon reduction but at a much higher reduction temperature compared with Pt NPs. The most meaningful feature of this discovery is that the NP active sites can be selectively encapsulated upon reduction at certain temperatures, therefore the catalytic performance can be finely tuned34,35,36,37,38,39,40. We believe this scenario is general and may be extended to TiO2 supported Pd catalysts to distinctly refine their catalytic performance and we will prove this in the following.
Diffuse reflectance infrared Fourier transform (DRIFT) spectra of CO adsorption were first employed to study the SMSI state of our Pd/TiO2 sample and the results are presented in Fig. 1e, f and Supplementary Fig. 4. For CO saturation adsorption on Pd/TiO2-200H, Fig. 1e, two peaks centered at 2106 and 2091 cm−1 and a broad band existed in the range of 1840–1990 cm−1 were observed in addition to the two gas phase CO bands. The former two are ascribed to linear CO adsorption on Pd single atoms and Pd NPs1 and Eq. 2:
Data availability
The data that support the findings of this study are available within the paper and its Supplementary information, and all data are available from the authors on reasonable request.
References
Borodziński, A. & Bond, G. C. Selective hydrogenation of ethyne in ethene-rich streams on palladium catalysts. Part 1. Effect of changes to the catalyst during reaction. Cat. Rev. Sci. Eng. 48, 91–144 (2006).
Borodziński, A. & Bond, G. C. Selective hydrogenation of ethyne in ethene-rich streams on palladium catalysts, part 2: steady-state kinetics and effects of palladium particle size, carbon monoxide, and promoters. Cat. Rev. Sci. Eng. 50, 379–469 (2008).
Shi, R. et al. Room-temperature electrochemical acetylene reduction to ethylene with high conversion and selectivity. Nat. Catal. 4, 565–574 (2021).
Wang, S. et al. Highly efficient ethylene production via electrocatalytic hydrogenation of acetylene under mild conditions. Nat. Commun. 12, 7072 (2021).
García-Mota, M. et al. A density functional theory study of the ‘mythic’ Lindlar hydrogenation catalyst. Theor. Chem. Acc. 128, 663–673 (2010).
Pei, G. X. et al. Promotional effect of Pd single atoms on Au nanoparticles supported on silica for the selective hydrogenation of acetylene in excess ethylene. New J. Chem. 38, 2043 (2014).
Pei, G. X. et al. Ag alloyed Pd single-atom catalysts for efficient selective hydrogenation of acetylene to ethylene in excess ethylene. ACS Catal. 5, 3717–3725 (2015).
Pei, G. X. et al. Performance of Cu-Alloyed Pd single-atom catalyst for semihydrogenation of acetylene under simulated front-end conditions. ACS Catal. 7, 1491–1500 (2017).
Osswald, J. et al. Palladium-gallium intermetallic compounds for the selective hydrogenation of acetylenePart II: Surface characterization and catalytic performance. J. Catal. 258, 219–227 (2008).
Zhou, H. et al. PdZn intermetallic nanostructure with Pd-Zn-Pd ensembles for highly active and chemoselective semi-hydrogenation of acetylene. ACS Catal. 6, 1054–1061 (2016).
Feng, Q. et al. Isolated single-atom Pd sites in intermetallic nanostructures: high catalytic selectivity for semihydrogenation of alkynes. J. Am. Chem. Soc. 139, 7294–7301 (2017).
Lee, J. H., Kim, S. K., Ahn, I. Y., Kim, W.-J. & Moon, S. H. Performance of Pd-Ag/Al2O3 catalysts prepared by the selective deposition of Ag onto Pd in acetylene hydrogenation. Catal. Commun. 12, 1251–1254 (2011).
Kim, S. K., Lee, J. H., Ahn, I. Y., Kim, W.-J. & Moon, S. H. Performance of Cu-promoted Pd catalysts prepared by adding Cu using a surface redox method in acetylene hydrogenation. Appl. Catal., A 401, 12–19 (2011).
Kim, E. et al. Pd catalyst promoted by two metal oxides with different reducibilities: properties and performance in the selective hydrogenation of acetylene. Appl. Catal., A 471, 80–83 (2014).
Kang, J. H., Shin, E. W., Kim, W. J., Park, J. D. & Moon, S. H. Selective hydrogenation of acetylene on TiO2-added Pd catalysts. J. Catal. 208, 310–320 (2002).
Ahn, I. Y., Kim, W. J. & Moon, S. H. Performance of La2O3- or Nb2O5-added Pd/SiO2 catalysts in acetylene hydrogenation. Appl. Catal., A 308, 75–81 (2006).
McKenna, F.-M. & Anderson, J. A. Selectivity enhancement in acetylene hydrogenation over diphenyl sulphide-modified Pd/TiO2 catalysts. J. Catal. 281, 231–240 (2011).
McCue, A. J. & Anderson, J. A. Sulfur as a catalyst promoter or selectivity modifier in heterogeneous catalysis. Catal. Sci. Technol. 4, 272–294 (2014).
McKenna, F. M., Wells, R. P. & Anderson, J. A. Enhanced selectivity in acetylene hydrogenation by ligand modified Pd/TiO2 catalysts. Chem. Commun. 47, 2351–2353 (2011).
McKenna, F. M., Mantarosie, L., Wells, R. P. K., Hardacre, C. & Anderson, J. A. Selective hydrogenation of acetylene in ethylene rich feed streams at high pressure over ligand modified Pd/TiO2. Catal. Sci. Technol. 2, 632 (2012).
McCue, A. J., McKenna, F.-M. & Anderson, J. A. Triphenylphosphine: a ligand for heterogeneous catalysis too? Selectivity enhancement in acetylene hydrogenation over modified Pd/TiO2 catalyst. Catal. Sci. Technol. 5, 2449–2459 (2015).
Liu, Z. The simplest construction of single-site catalysts for efficient energy conversion. Acta Phys. Chim. Sin. 35, 1047–1048 (2019).
Qiao, B. et al. Single-atom catalysis of CO oxidation using Pt1/FeOx. Nat. Chem. 3, 634–641 (2011).
Huang, X. et al. Enhancing both selectivity and coking-resistance of a single-atom Pd1/C3N4 catalyst for acetylene hydrogenation. Nano Res. 10, 1302–1312 (2017).
Huang, F. et al. Anchoring Cu1 species over nanodiamond-graphene for semi-hydrogenation of acetylene. Nat. Commun. 10, 4431 (2019).
Dai, X. et al. Single Ni sites distributed on N-doped carbon for selective hydrogenation of acetylene. Chem. Commun. 53, 11568–11571 (2017).
Huang, F. et al. Atomically dispersed Pd on nanodiamond/graphene hybrid for selective hydrogenation of acetylene. J. Am. Chem. Soc. 140, 13142–13146 (2018).
Zhou, S. et al. Pd single-atom catalysts on nitrogen-doped graphene for the highly selective photothermal hydrogenation of acetylene to ethylene. Adv. Mater. 31, e1900509 (2019).
Zhang, L., Zhou, M., Wang, A. & Zhang, T. Selective hydrogenation over supported metal catalysts: from nanoparticles to single atoms. Chem. Rev. 120, 683–733 (2020).
Guan, E. et al. Supported metal pair-site catalysts. ACS Catal. 10, 9065–9085 (2020).
**, J. et al. Synthesis strategies, catalytic applications, and performance regulation of single-atom catalysts. Adv. Funct. Mater. 31, 2008318 (2021).
Tauster, S. J. & Fung, S. C. Strong metal-support interactions-occurrence among binary oxides of groups IIA-VB. J. Catal. 55, 29–35 (1978).
Tauster, S. J. Strong metal-support interactions. Acc. Chem. Res. 20, 389–394 (1987).
Han, B. et al. Strong metal-support interactions between Pt single atoms and TiO2. Angew. Chem. Int. Ed. 59, 11824–11829 (2020).
Du, X. et al. Size-dependent strong metal-support interaction in TiO2 supported Au nanocatalysts. Nat. Commun. 11, 5811 (2020).
Ma, D. Size-dependent strong metal-support interaction. Acta Phys. Chim. Sin. 38, 2101039 (2022).
Wang, H. et al. Strong metal-support interactions on gold nanoparticle catalysts achieved through Le Chatelier’s principle. Nat. Catal. 4, 418–424 (2021).
Zhang, Y. et al. Structure sensitivity of Au-TiO2 strong metal-support interactions. Angew. Chem. Int. Ed. 60, 12074–12081 (2021).
Tang, H. et al. Ultrastable hydroxyapatite/titanium-dioxide-supported gold nanocatalyst with strong metal-support interaction for carbon monoxide oxidation. Angew. Chem. Int. Ed. 55, 10606–10611 (2016).
Dong, J. et al. Reaction-induced strong metal-support interactions between metals and inert boron nitride nanosheets. J. Am. Chem. Soc. 142, 17167–17174 (2020).
Zhou, L. et al. Light-driven methane dry reforming with single atomic site antenna-reactor plasmonic photocatalysts. Nat. Energy 5, 61–70 (2020).
Zhang, C. et al. Al-Pd nanodisk heterodimers as antenna-reactor photocatalysts. Nano Lett. 16, 6677–6682 (2016).
Zhou, L. et al. Quantifying hot carrier and thermal contributions in plasmonic photocatalysis. Science 362, 69–72 (2018).
O’Brien, P. G. et al. Photomethanation of gaseous CO2 over Ru/silicon nanowire catalysts with visible and near-infrared photons. Adv. Sci. 1, 1400001 (2014).
Wang, Z. J., Song, H., Liu, H. & Ye, J. Coupling of solar energy and thermal energy for carbon dioxide reduction: status and prospects. Angew. Chem. Int. Ed. 59, 8016–8035 (2020).
Ghoussoub, M., **a, M., Duchesne, P. N., Segal, D. & Ozin, G. Principles of photothermal gas-phase heterogeneous CO2 catalysis. Energy Environ. Sci. 12, 1122–1142 (2019).
Mateo, D., Cerrillo, J. L., Durini, S. & Gascon, J. Fundamentals and applications of photo-thermal catalysis. Chem. Soc. Rev. 50, 2173–2210 (2021).
Zhao, L. et al. Solar-driven water-gas shift reaction over CuOx/Al2O3 with 1.1% of light-to-energy storage. Angew. Chem. Int. Ed. 58, 7708–7712 (2019).
Gao, W. et al. Photo-driven syngas conversion to lower olefins over oxygen-decorated Fe5C2 catalyst. Chem 4, 2917–2928 (2018).
Zhang, X. et al. Product selectivity in plasmonic photocatalysis for carbon dioxide hydrogenation. Nat. Commun. 8, 14542 (2017).
Marimuthu, A., Zhang, J. & Linic, S. Tuning selectivity in propylene epoxidation by plasmon mediated photo-switching of Cu oxidation state. Science 339, 1590–1593 (2013).
Song, C., Wang, Z., Yin, Z., **ao, D. & Ma, D. Principles and applications of photothermal catalysis. Chem. Catal. 2, 52–83 (2022).
Imamura, K., Nakanishi, K., Hashimoto, K. & Kominami, H. Chemoselective reduction of nitrobenzenes having other reducible groups over titanium(IV) oxide photocatalyst under protection-, gas-, and metal-free conditions. Tetrahedron 70, 6134–6139 (2014).
Fukui, M., Kouda, H., Tanaka, A., Hashimoto, K. & Kominami, H. Heterogeneous Meerwein-Ponndorf-Verley-type reduction of aromatic aldehydes having other reducible functional groups over a TiO2 photocatalyst. ChemistrySelect 2, 2293–2299 (2017).
Kojima, Y., Fukui, M., Tanaka, A., Hashimoto, K. & Kominami, H. Additive-free semihydrogenation of an alkynyl group to an alkenyl group over Pd-TiO2 photocatalyst utilizing temporary in-situ deactivation. ChemCatChem 10, 3605–3611 (2018).
Lian, J. et al. Unexpectedly selective hydrogenation of phenylacetylene to styrene on titania supported platinum photocatalyst under 385 nm monochromatic light irradiation. Chin. J. Catal. 41, 598–603 (2020).
Fukui, M. et al. Visible light-induced diastereoselective semihydrogenation of alkynes to cis-alkenes over an organically modified titanium(IV) oxide photocatalyst having a metal co-catalyst. J. Catal. 374, 36–42 (2019).
Wang, B., Duke, K., Scaiano, J. C. & Lanterna, A. E. Cobalt-molybdenum co-catalyst for heterogeneous photocatalytic H-mediated transformations. J. Catal. 379, 33–38 (2019).
Kominami, H. et al. Copper-modified titanium dioxide: a simple photocatalyst for the chemoselective and diastereoselective hydrogenation of alkynes to alkenes under additive-free conditions. ChemCatChem 8, 2019–2022 (2016).
Swearer, D. F. et al. Heterometallic antenna-reactor complexes for photocatalysis. PNAS 113, 8916–8920 (2016).
Schreyer, H. et al. Milling down to nanometers: a general process for the direct dry synthesis of supported metal catalysts. Angew. Chem. Int. Ed. 58, 11262–11265 (2019).
Liu, Y. et al. Catalytic oxidation of 5-hydroxymethylfurfural to 2,5-diformylfuran over atomically dispersed ruthenium catalysts. Ind. Eng. Chem. Res. 59, 4333–4337 (2020).
Gan, T. et al. Unveiling the kilogram-scale gold single-atom catalysts via ball milling for preferential oxidation of CO in excess hydrogen. Chem. Eng. J. 389, 124490 (2020).
He, X. et al. Mechanochemical kilogram-scale synthesis of noble metal single-atom catalysts. Cell Rep. Phys. Sci. 1, 100004 (2020).
Xu, H. et al. Entropy-stabilized single-atom Pd catalysts via high-entropy fluorite oxide supports. Nat. Commun. 11, 3908 (2020).
Liu, P. X. et al. Photochemical route for synthesizing atomically dispersed palladium catalysts. Science 352, 797–801 (2016).
Wei, S. et al. Direct observation of noble metal nanoparticles transforming to thermally stable single atoms. Nat. Nanotechnol. 13, 856 (2018).
**n, P. et al. Revealing the active species for aerobic alcohol oxidation by using uniform supported palladium catalysts. Angew. Chem. Int. Ed. 57, 4642–4646 (2018).
Kast, P. et al. Strong metal-support interaction and alloying in Pd/ZnO catalysts for CO oxidation. Catal. Today 260, 21–31 (2016).
DeRita, L. et al. Catalyst architecture for stable single atom dispersion enables site-specific spectroscopic and reactivity measurements of CO adsorbed to Pt atoms, oxidized Pt clusters, and metallic Pt clusters on TiO2. J. Am. Chem. Soc. 139, 14150–14165 (2017).
Tang, H. et al. Classical strong metal-support interactions between gold nanoparticles and titanium dioxide. Sci. Adv. 3, e1700231 (2017).
Panpranot, J., Kontapakdee, K. & Praserthdam, P. Effect of TiO2 crystalline phase composition on the physicochemical and catalytic properties of Pd/TiO2 in selective acetylene hydrogenation. J. Phys. Chem. B 110, 8019–8024 (2006).
Yang, J., Lv, C. Q., Guo, Y. & Wang, G. C. A DFT+U study of acetylene selective hydrogenation on oxygen defective anatase (101) and rutile (110) TiO2 supported Pd4 cluster. J. Chem. Phys. 136, 104107 (2012).
Chen, H. et al. Photoinduced strong metal-support interaction for enhanced catalysis. J. Am. Chem. Soc. 143, 8521–8526 (2021).
Pallassana, V. & Neurock, M. Electronic factors governing ethylene hydrogenation and dehydrogenation activity of pseudomorphic PdML/Re(0001), PdML/Ru(0001), Pd(111), and PdML/Au(111) surfaces. J. Catal. 191, 301–317 (2000).
Qin, R. et al. Carbon monoxide promotes the catalytic hydrogenation on metal cluster catalysts. Research 2020, 4172794 (2020).
Chen, Y. et al. Discovering partially charged single-atom Pt for enhanced anti-Markovnikov alkene hydrosilylation. J. Am. Chem. Soc. 140, 7407–7410 (2018).
Wan, Q., Hu, S., Dai, J., Chen, C. & Li, W. X. First-principles kinetic study for Ostwald ripening of late transition metals on TiO2 (110). J. Phys. Chem. C. 123, 1160–1169 (2018).
Wan, Q. X., Hu, S. L., Dai, J. N., Chen, C. Q. & Li, W. X. Influence of crystal facet and phase of titanium dioxide on Ostwald ripening of supported Pt nanoparticles from first-principles kinetics. J. Phys. Chem. C. 123, 11020–11031 (2019).
Yeganeh, S. & Voorhis, T. V. Triplet excitation energy transfer with constrained density functional theory. J. Phys. Chem. C. 114, 20756–20763 (2010).
Kubas, A. M., Gajdos, F., Heck, A., Oberhofer, H. & Blumberger, J. Electronic couplings for molecular charge transfer: benchmarking CDFT, FODFT and FODFTB against high-level ab initio calculations. II. Phys. Chem. Chem. Phys. 17, 14342–14354 (2015).
Lu, T. & Chen, F. Multiwfn: a multifunctional wavefunction analyzer. J. Comput. Chem. 33, 580–592 (2012).
Acknowledgements
The authors thank Prof. Can Li at Dalian Institute of Chemical Physics for his valuable discussions and suggestions. This work was financially supported by National Key Research and Development Program of China (2021YFA1500503), National Natural Science Foundation of China (21972135, 21961142006, 22090033 and 51701201), and CAS Project for Young Scientists in Basic Research (YSBR-022). The synchrotron radiation experiment for XAFS was performed at the BL08B2 of SPring-8 with the approval of Japan Synchrotron Radiation Research Institute (Proposal Nos. 2019B3415, 2020A3415).
Author information
Authors and Affiliations
Contributions
B.Q. and T.Z. conceived and supervised the project. Y.G. carried out the catalyst synthesis, catalytic performance test, stability evaluation, and conducted some characterizations. Y.H. conducted the simulation and calculation. B.H., M.A., Q.L., and Li.Li. helped to analyze the data. Y.S. and Q.J. carried out the STEM and EELS characterizations. Y.T.C. and Le.Li. carried out the XAS measurements and analysis. B.Z., M.S., and Y.Z. performed some photocatalytic experiments and helped on the discussion of mechanism. R.L. guided and supervised the experiments of photo-thermo catalysis and wrote the corresponding section. Y.G. and B.Q. wrote the paper. All authors contributed to project discussions and modified the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Guo, Y., Huang, Y., Zeng, B. et al. Photo-thermo semi-hydrogenation of acetylene on Pd1/TiO2 single-atom catalyst. Nat Commun 13, 2648 (2022). https://doi.org/10.1038/s41467-022-30291-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-022-30291-x
- Springer Nature Limited
This article is cited by
-
A MOF-supported Pd1–Au1 dimer catalyses the semihydrogenation reaction of acetylene in ethylene with a nearly barrierless activation energy
Nature Catalysis (2024)
-
Pt-doped Ru nanoparticles loaded on ‘black gold’ plasmonic nanoreactors as air stable reduction catalysts
Nature Communications (2024)
-
Ambient hydrogenation of solid aromatics enabled by a high entropy alloy nanocatalyst
Nature Communications (2024)
-
The stability of single-atom catalysts in thermocatalysis
Surface Science and Technology (2024)
-
Acetylene hydrogenation to ethylene by water at low temperature on a Au/α-MoC catalyst
Nature Catalysis (2023)