Abstract
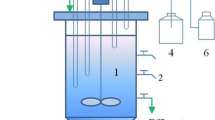
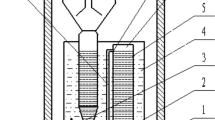
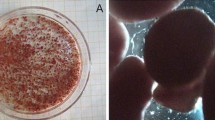
In this study, the effects of both transient and long-term inorganic carbon (IC) addition on the anaerobic ammonium oxidation (ANAMMOX) process under pseudo-steady-state and substrate inhibitions were analyzed using reactor performance and measures of sludge activity. Compared with the nitrogen removal rate (NRR) of 3.42 kg N m−3 day−1 in the control bioreactor (ICDR) without IC, the peak NRR reached 21.0 kg N m−3 day−1 in the reactor (ICAR) with sufficient IC added. It was revealed that the long-term addition of bicarbonate significantly enhanced the performance of the ANAMMOX reactor. The optimum HCO3 −/TN ratio was considered to be 1.20, which is lower than that in normal conditions. The IC concentration affected biomass activity, and the transient addition or removal of IC to differing sludge media caused a significant loss of activity. Sufficient addition of IC alleviated the inhibition of excess substrates, while the inhibition was aggravated by the IC limitation. The half-maximal (50 %) inhibitory concentrations of substrate for the sludge were 295 mg L−1 NO2 −–N and 361 mg L−1 NH4 +–N with 120 mg L−1 of fixed HCO3 − and 346 mg L−1 NO2 −–N and 456 mg L−1 NH4 +–N with unlimited IC, respectively. Changing the HCO3 −/TN (in milligrams per milligram) ratio resulted in the variation of ANAMMOX stoichiometric ratios. Sludge characterization parameters in the ICDR, including biomass, extracellular polymeric substances, heme C, and so on, were lower than those in ICAR. Filamentous bacteria and spherical bacteria were also observed in the reactor with limited IC.







Similar content being viewed by others
References
Anthonisen AC, Loehr RC, Prakasam TBS, Srinath EG (1976) Inhibition of nitrification by ammonia and nitrous acid. J Water Pollut Control Fed 48:835–852
APHA, AWWA, WEF (1998) Standard methods for water and wastewater examination. American Public Health Association, Washington, DC
Casey TG, Wentzel MC, Loewenthal RE, Ekama GA, Marais GVR (1992) A hypothesis for the cause of low F/M filament bulking in nutrient removal activated sludge systems. Water Res 26:867–869
Casey TG, Wentzel MC, Ekama GA, Loewenthal RE, Marais GVR (1994) A hypothesis for the causes and control of anoxic–aerobic (AA) filament bulking in nutrient removal activated sludge systems. Water Sci Technol 29(7):203–212
Chamchoi N, Nitisoravut S (2007) Anammox enrichment from different conventional sludges. Chemosphere 66:2225–2232
Chen TT, Zheng P, Shen LD (2012) Growth and metabolism characteristics of anaerobic ammonium-oxidizing bacteria aggregates. Appl Microbiol Biotechnol. doi:10.1007/s00253-012-4346-z
Dolby JM, Stephens S, Honour P (1977) Bacteriostasis of Escherichia coli by milk: II. Effect of bicarbonate and transferrin on the activity of infant feeds. J Hyg Camb 78:235–242
Ferguson SJ (1994) Denitrification and its control. Antonie van Leeuwenhoek 66:89–110
Fernández I, Dosta J, Fajardo C, Campos JL, Mosquera-Corral A, Méndez R (2012) Short- and long-term effects of ammonium and nitrite on the Anammox process. J Environ Manage 95(Suppl):S170–S174
Isaka K, Date Y, Sumino T, Tsuneda S (2007) Ammonium removal performance of anaerobic ammonium-oxidizing bacteria immobilized in polyethylene glycol gel carrier. Appl Microbiol Biotechnol 76:1457–1495
Jetten MS, Strous M, van de Pas–Schoonen KT, Schalk J, van Dongen U, van de Graaf AA, Logemann S, Muyzer G, van Loosdrecht MCM, Kuenen JG (1998) The anaerobic oxidation of ammonium. FEMS Microbiol Rev 22:421–437
** RC, Yang GF, Ma C, Yu JJ, Zhang QQ, **ng BX (2012) Influence of effluent recirculation on the performance of Anammox process. Chem Eng J 200–202:176–185
Kartal B, Kuypers MMM, Lavik G, Schalk J, Op den Camp HJM, Jetten MSM, Strous M (2007) Anammox bacteria disguised as denitrifiers: nitrate reduction to dinitrogen gas via nitrite and ammonium. Environ Microbiol 9:635–642
Kartal B, Keltjens JT, Jetten MSM (2008a) The metabolism of anammox. Encyclopedia of life science (ELS). Wiley, Chichester, pp 1–9. doi:10.1002/9780470015902.a0021315
Kartal B, van Niftrik L, Rattray J, van de Vossenberg JLCM, Schmid MC, Damste JSS, Jetten MSM, Strous M (2008b) Candidatus ‘Brocadia fulgida’: an autofluorescent anaerobic ammonium oxidizing bacterium. FEMS Microbiol Ecol 63:46–55
Kartal B, Maalcke WJ, de Almeida NM, Cirpus I, Gloerich J, Geerts W, Op den Camp HJM, Harhangi HR, Janssen-Megens EM, Francoijs KJ, Stunnenberg HG, Keltjens JT, Jetten MSM, Strous M (2011) Molecular mechanism of anaerobic ammonium oxidation. Nature 479:127–130
Kartal B, van Niftrik L, Keltjens JT, Op den Camp HJM, Jetten MSM (2012) Anammox—growth physiology, cell biology and metabolism. Adv Microb Physiol 60:211–262
Kimua Y, Isaka K, Kazama F (2011) Effects of inorganic carbon limitation on anaerobic ammonium oxidation (anammox) activity. Bioresour Technol 102:4390–4394
Liao DX, Li XM, Yang Q, Zeng GM, Liang G, Yue X (2008) Effect of inorganic carbon on anaerobic ammonium oxidation enriched in sequencing batch reactor. J Environ Sci 20:940–944
López H, Puig S, Ganigué R, Ruscalleda M, Balaguer MD, Colprim J (2008) Start-up and enrichment of granular anammox SBR to treat high nitrogen load wastewaters. J Chem Technol Biotechnol 83:233–241
Lotti T, van der Star WR, Kleerebezem R, Lubello C, van Loosdrecht MCM (2012) The effect of nitrite inhibition on the anammox process. Water Res 46:2559–2569
Mulder A, van de Graaf AA, Robertson LA, Kuenen JG (1995) Anaerobic ammonium oxidation discovered in a denitrifying fluidized bed reactor. FEMS Microbiol Ecol 16:177–184
Oshiki M, Shimokawa M, Fujii N, Satoh H, Okabe S (2011) Physiological characteristics of the anaerobic ammonium-oxidizing bacterium ‘Candidatus Brocadia sinica’. Microbiology (UK) 157:1706–1713
Ragsdale SW, Wood HG (1991) Enzymology of the acetyl-CoA pathway of CO2 fixation. Crit Rev Biochem Mol Biol 26(3–4):261–300
Schouten S, Strous M, Kuypers MMM, Rijpstra WIC, Baas M, Schubert CJ, Jetten MSM, Damsté SS (2004) Stable carbon isotopic fractionations associated with inorganic carbon fixation by anaerobic ammonium oxidizing bacteria. Appl Environ Microbiol 40:3785–3788
Sheng GP, Zhang ML, Yu HQ (2006) Characterization of adsorption properties of extracellular polymeric substances (EPS) extracted from sludge. Colloids Surf B 62:83–90
Sinclair PR, Gorman N, Jacobs JM (2001) Measurement of heme concentration. Curr Protoc Toxicol Chapter 8:Unit 8.3. doi:10.1002/0471140856.tx0803s00
Skiadas IV, Gavala HN, Schmidt JE, Ahring BK (2003) Anaerobic granular sludge and biofilm reactors. Adv Biochem Eng Biotechnol 82:35–67
Sprott GD, Patel GB (1986) Ammonia toxicity in pure cultures of methanogenic bacteria. Syst Appl Microbiol 7:358–363
Strous M, Heijnen JJ, Kuenen JG, Jetten MSM (1998) The sequencing batch reactor as a powerful tool to study very slowly growing micro-organisms. Appl Microbiol Biotechnol 50:589–596
Strous M, Fuerst JA, Kramer EHM, Logemann S, Muyzer G, van de Pas-Schoonen KT, Webb R, Kuenen JG, Jetten MSM (1999a) Missing lithotroph identified as new planctomycete. Nature 400:446–449
Strous M, Kuenen JG, Jetten MSM (1999b) Key physiology of anaerobic ammonium oxidation. Appl Environ Microbiol 65:3248–3250
Strous M, Pelletier E, Mangenot S, Rattei T, Lehner A, Taylor MW, Horn M, Daims H, Bartol-Mavel D, Wincker P, Barbe V, Fonknechten N, Vallenet D, Segurens B, Schenowitz-Truong C, Medigue C, Collingro A, Snel B, Dutilh BE, Op den Camp HJ, van der Drift C, Cirpus I, van de Pas-Schoonen KT, Harhangi HR, van Niftrik L, Schmid M, Keltjens J, van de Vossenberg J, Kartal B, Meier H, Frishman D, Huynen MA, Mewes HW, Weissenbach J, Jetten MSM, Wagner M, Le Paslier D (2006) Deciphering the evolution and metabolism of an anammox bacterium from a community genome. Nature 440:790–794
Tang CJ, Zheng P, Wang CH, Mahmood Q, Zhang JQ, Chen XG, Zhang L, Chen JW (2011) Performance of high-loaded ANAMMOX UASB reactors containing granular sludge. Water Res 45:135–144
van de Graaf AA, de Bruijn P, Robertson LA, Jetten MSM, Kuenen JG (1996) Autotrophic growth of anaerobic ammonium-oxidizing microorganisms in a fluidized bed reactor. Microbiology (UK) 142:2187–2196
van der Star WRL, Miclea AI, van Dongen UGJM, Muyzer G, Picioreanu C, van Loosdrecht MM (2008) The membrane bioreactor: a novel tool to grow Anammox bacteria as free cells. Biotechnol Bioeng 101:286–294
Waki M, Tokutomi T, Yokoyama H, Tanaka Y (2007) Nitrogen removal from animal waste treatment water by anammox enrichment. Bioresour Technol 14:2775–2780
Wu J, Zhou HM, Li HZ, Zhang PC, Jiang J (2009) Impacts of hydrodynamic shear force on nucleation of flocculent sludge in anaerobic reactor. Water Res 43:3029–3036
Yang GF, ** RC (2012) The joint inhibitory effects of phenol, copper (II), oxytetracycline (OTC) and sulfide on Anammox activity. Bioresour Technol 126:187–192
Yang HW, Jiang ZP, Shi SQ, Tang WZ (2002) INT-dehydrogenase activity test for assessing anaerobic biodegradability of organic compounds. Ecotox Environ Safe 53:416–421
Yang J, Zhang L, Fukuzaki Y, Hira D, Furukawa K (2010) High-rate nitrogen removal by the anammox process with a sufficient inorganic carbon source. Bioresour Technol 101:9471–9478
Yao HJ, Tian SP, Wang YS (2004) Sodium bicarbonate enhances biocontrol efficacy of yeasts on fungal spoilage of pears. Int J Food Microbiol 93:297–304
Yu JJ, ** RC (2012) The ANAMMOX reactor under transient-state conditions: process stability with fluctuations of the nitrogen concentration, inflow rate, pH and sodium chloride addition. Bioresour Technol 119:166–173
Zekker I, Rikmann E, Tenno T, Vabamäe P, Kroon K, Loorits L, Saluste A, Tenno T (2012) Effect of HCO3 − concentration on anammox nitrogen removal rate in a moving bed biofilm reactor. Environ Technol 33(19–21):2263–2271
Acknowledgments
This work was supported by the National Natural Science Foundation of China (nos. 51078121 and 51278162), the **nmiao Talent Program of Zhejiang Province (no. 2012R421059), and the Program for Excellent Young Teachers in Hangzhou Normal University (HNUEYT) (JTAS 2011-01-020).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 121 kb)
Rights and permissions
About this article
Cite this article
**, RC., Yu, JJ., Ma, C. et al. Transient and long-term effects of bicarbonate on the ANAMMOX process. Appl Microbiol Biotechnol 98, 1377–1388 (2014). https://doi.org/10.1007/s00253-013-5004-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-013-5004-9