Abstract
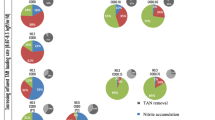
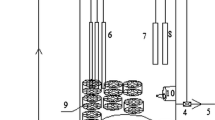
Anaerobic ammonium-oxidizing (anammox) bacteria were immobilized in polyethylene glycol gel carriers. A small amount of seed sludge [0.24% (w/v)] was entrapped in the carriers, and continuous feeding tests were performed. Nitrogen removal activity increased gradually, reaching 3.7 kg N/m3 reactor per day on day 67. The average of nitrogen conversion rate was calculated as 3.4 kg N/m3 reactor per day. Microscopic examination clearly showed that small red clusters formed in the gel carrier. Moreover, fluorescence in situ hybridization analysis revealed that these clusters consisted of anammox bacteria. From real-time polymerase chain reaction analysis, the growth of anammox bacteria in the gel carriers was clearly shown by increased concentration of 16S rRNA gene of planctomycete from 4.3 × 108 to 4.2 × 109 copies/ml between days 41 and 55. To determine the effects of inoculation on the start-up of the reactor, the amount of seed sludge in the gel carrier was varied and it was found that the start-up period could be reduced to as little as 25 days when a sludge concentration of 1.4% (w/v) was used. This is the first report of successful immobilization and cultivation of anammox bacteria in a gel carrier.








Similar content being viewed by others
References
Amann RI (1995) In situ identification of micro-organism by whole cell hybridization with rRNA-targeted nucleic probes. In: Akkerman ADC, van Elsas JD, de Bruijn FJ (eds) Molecular microbial ecology manual. Kluwer, Dordrecht, pp 1–15
Asano H, Myoga H, Asano M, Toyao M (1992) A study of nitrification utilizing whole microorganisms immobilized by the PVA-freezing method. Water Sci Technol 26:1037–1046
Furukawa K, Rouse JD, Bhatti ZI, Imajo U, Nakamura K, Ishida H (2002) Anaerobic oxidation of ammonium confirmed in continuous flow treatment using a nonwoven biomass carrier. Jpn J Water Treat Biol 38:87–94
Imajo U, Tokutomi T, Furukawa K (2005) Evaluation of various reactor types for the anammox process. J Jpn Soc Water Environ 28:185–190
Isaka K, Date Y, Sumino T, Yoshie S, Tsuneda S (2006) Growth characteristic of anaerobic ammonium-oxidizing (anammox) bacteria in an anaerobic biological filtrated (ABF) reactor. Appl Microbiol Biotechnol 70:47–52
Mulder A, Van de Graaf AA, Robertson LA, Kuenen JG (1995) Anaerobic ammonium oxidation discovered in a denitrifying fluidized bed reactor. FEMS Microbiol Ecol 16:177–184
Neef A, Amann R, Schlesner H, Schleifer KH (1998) Monitoring a widespread bacterial group: in situ detection of planctomycetes with 16S rRNA-targeted probes. Microbiology 144:3257–3266
Noda N, Ikuta H, Ebie Y, Hirata A, Tsuneda S, Matsumura M, Sumino T, Inamori Y (2000) Rapid quantification and in situ detection of nitrifying bacteria in biofilms by monoclonal antibody method. Water Sci Technol 65:3182–3191
Okabe S, Satoh H, Watanabe Y (1999) In situ analysis of nitrifying biofilms as determined by in situ hybridization and the use of microelectrodes. Appl Environ Microbiol 65:3182–3191
Saiki Y, Iwabuchi C, Katami A, Kitagawa Y (2002) Microbial analyses by fluorescence in situ hybridization of well-settled granular sludge in brewery wastewater treatment plants. J Biosci Bioeng 93:601–606
Sliekers AO, Third KA, Abma W, Kuenen JG, Jetten MSM (2003) CANON and anammox in a gas-lift reactor. FEMS Microbiol Lett 218:339–344
Strous M, Van Gerven E, Zheng P, Kuenen JG, Jetten MSM (1997) Ammonium removal from concentrated waste streams with the anaerobic ammonium oxidation (ANAMMOX) process in different reactor configurations. Water Res 31:1955–1962
Strous M, Heijnen JJ, Kuenen JG, Jetten MSM (1998) The sequencing batch reactor as a powerful tool for the study of slowly growing anaerobic ammonium-oxidizing microorganisms. Appl Microbiol Biotechnol 50:589–596
Strous M, Fuerst JA, Kramer EHM, Logemann S, Muyzer G, Van de Pas-Schoonen KT, Webb R, Kuenen JG, Jetten MSM (1999) Missing lithotroph identified as new planctomycete. Nature 400:446–449
Sumino T, Nakamura H, Mori N (1992) Immobilization of activated sludge by polyethylene glycol prepolymer. J Ferment Bioeng 73:37–42
Van de Graaf AA, de Bruijn P, Robertson LA, Jetten MSM, Kuenen JG (1996) Autotrophic growth of anaerobic ammonium-oxidizing microorganisms in a fluidized bed reactor. Microbiol 142:2187–2196
Van de Graaf AA, Peter de Bruijn, Robertson LA, Jetten MSM, Kuenen JG (1997) Metabolic pathway of anaerobic ammonium oxidation on the basis of 15N studies in a fluidized bed reactor. Microbiology 143:2415–2421
Vogelsang C, Husby A, Osgaard K (1997) Functional stability of temperature-compensated nitrification in domestic wastewater treatment obtained with PVA-SBQ/alginate gel entrapment. Water Res 31:1659–1664
Weatherburn MW (1967) Phenol-hypochlorite reaction for determination of ammonia. J Anal Chem 39:971–974
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Isaka, K., Date, Y., Sumino, T. et al. Ammonium removal performance of anaerobic ammonium-oxidizing bacteria immobilized in polyethylene glycol gel carrier. Appl Microbiol Biotechnol 76, 1457–1465 (2007). https://doi.org/10.1007/s00253-007-1106-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-007-1106-6