Search
Search Results
-
Does Firm’s Alliances Increase New Drug Development Time? A Multiple Regression Analysis of Clinical Development Time
PurposeAlliances between firms are becoming more common for drug development; however, it is not clear how these alliances affect the time spent on...

-
Computers in Clinical Development
Clinical development of a pharmaceutical product plays an important role as it regulates the future of investigational new drug application approval...
-
Future Development of Automated Technique for Clinical Microbiology
At the current scenario, the human population is suffering from many types of microbial infections. However, microbial infection is diagnosed with...
-
Clinical Bridging Studies and Modeling Approach for Implementation of a Patient Centric Sampling Technique in Padsevonil Clinical Development
Volumetric absorptive microsampling (VAMS) techniques have gained popularity these last years as innovative tool for collection of blood...

-
Role of Modeling and Simulation in Preclinical and Clinical Long-Acting Injectable Drug Development
Innovations in the field of long-acting injectable drug development are increasingly being reported. More advanced in vitro and in vivo ...

-
Clinical Pharmacokinetic Studies
Drug efficacy and response are a function of drug concentration over time. In clinical pharmacokinetic studies, aspects of drug absorption,...
-
Achieving big with small: quantitative clinical pharmacology tools for drug development in pediatric rare diseases
Pediatric populations represent a major fraction of rare diseases and compound the intrinsic challenges of pediatric drug development and drug...

-
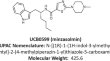
In vivo effects of the alpha-synuclein misfolding inhibitor minzasolmin supports clinical development in Parkinson’s disease
Direct targeting of alpha-synuclein (ASYN) has emerged as a disease-modifying strategy for Parkinson’s disease and other synucleinopathies which is...
-
Enabling endpoint development for interventional clinical trials in individuals with Angelman syndrome: a prospective, longitudinal, observational clinical study (FREESIAS)
BackgroundAngelman syndrome (AS) is a rare neurodevelopmental disorder characterized by the absence of a functional UBE3A gene, which causes...

-
Methylomics and cancer: the current state of methylation profiling and marker development for clinical care
Epigenetic modifications have long been recognized as an essential level in transcriptional regulation linking behavior and environmental conditions...

-
Advanced Therapy Medicinal Products: Clinical, Non-clinical, and Quality Considerations
Cell therapy, tissue engineering, and gene therapy products, together called “advanced therapy medicinal products” (ATMPs), represent a heterogeneous...
-
Sha** Clinical Reasoning
Clinical reasoningReasoning, clinical is at the core of all healthHealth-related professions, and it is long recognized as a critical skillSkill for...
-

-
A comprehensive regulatory and industry review of modeling and simulation practices in oncology clinical drug development
Exposure–response (E–R) analyses are an integral component in the development of oncology products. Characterizing the relationship between drug...

-
Development of a clinical polygenic risk score assay and reporting workflow
Implementation of polygenic risk scores (PRS) may improve disease prevention and management but poses several challenges: the construction of...

-
Clinical Development of mRNA Vaccines: Challenges and Opportunities
The emergence of safe and effective mRNA platform-based COVID-19 vaccines from the recent pandemic has changed the face of vaccine development....
-
Model-Informed Clinical Development of Once-Every-6-Month Injection of Paliperidone Palmitate in Patients with Schizophrenia: A Pharmacometric Bridging Approach (Part I)
Background and ObjectiveA model-informed drug development (MIDD) approach was implemented for paliperidone palmitate (PP) 6-month (PP6M) clinical...

-
Review of the clinical development of fexuprazan for gastroesophageal reflux–related disease
Proton pump inhibitors (PPIs) are a mainstay treatment for acid peptic disorders such as gastroesophageal reflux disease (GERD). Although PPIs are...

-
Doses Evaluated in Clinical Pharmacology Studies Investigating the Effect of Intrinsic and Extrinsic Factors on PK and Safety: Case Examples from Approved Drug Development Programs
Dose selection for investigations of intrinsic and extrinsic factors of pharmacokinetic variability as well as safety is a challenging question in...

-
Clinical Practice Guidelines for the Immunological Management of Chromosome 22q11.2 Deletion Syndrome and Other Defects in Thymic Development
Current practices vary widely regarding the immunological work-up and management of patients affected with defects in thymic development (DTD),...

