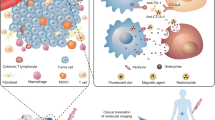
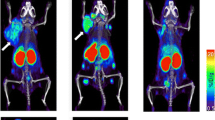
Abstract
Cancer immunotherapy treatments have shown promising results in several clinical trials and have demonstrated beneficial therapeutic outcomes even in very advanced stages of cancer. Recently, immune checkpoint inhibitor (ICIs)-based cancer immunotherapy treatments, such as: anti-CTLA-4, anti-PD-L1, and anti-PD-1 therapies, have been found to be highly effective as monotherapies and as a combination of cancer immunotherapies in many clinical trials. ICIs are antibodies or small molecules that specifically target and block the binding site of specific ligands on immune cells, thus strengthening the immune system’s response to cancer cells in the tumor microenvironment (TME). With the rapid rise of both number and popularity of the immune checkpoint blockade (ICB) cancer therapeutics, there has been an increasing need for reliable, robust, and noninvasive imaging approaches to measure in vivo immune checkpoint markers. Several immunoPET tracers that can be administered noninvasively to measure ICs in the TME have been developed to assist in the selection of appropriate patients for ICB treatments. In this chapter, we discuss the various immunotherapy strategies, including ICB therapy, and the corresponding immunoPET tracers used to evaluate and measure the IC markers to stratify the patients for suitable ICB therapy.
Similar content being viewed by others
References
Aluicio-Sarduy E, Ellison PA, Barnhart TE, Cai W, Nickles RJ, Engle JW (2018) PET radiometals for antibody labeling. J Labelled Comp Radiopharm 61(9):636–651. https://doi.org/10.1002/jlcr.3607
Arndt C et al (2019) A theranostic PSMA ligand for PET imaging and retargeting of T cells expressing the universal chimeric antigen receptor UniCAR. Onco Targets Ther 8(11):1659095. https://doi.org/10.1080/2162402X.2019.1659095
Baggio L, Laureano AM, Silla L, Lee DA (2017) Natural killer cell adoptive immunotherapy: coming of age. Clin Immunol 177:3–11. https://doi.org/10.1016/j.clim.2016.02.003
Bahce I et al (2014) Pilot study of (89)Zr-bevacizumab positron emission tomography in patients with advanced non-small cell lung cancer. EJNMMI Res 4(1):35. https://doi.org/10.1186/s13550-014-0035-5
Barbari C et al (2020) Immunotherapies and combination strategies for immuno-oncology. Int J Mol Sci 21(14):5009. https://doi.org/10.3390/ijms21145009
Baum RP et al (2010) Molecular imaging of HER2-expressing malignant tumors in breast cancer patients using synthetic 111In- or 68Ga-labeled affibody molecules. J Nucl Med 51(6):892–897. https://doi.org/10.2967/jnumed.109.073239
Belderbos S et al (2020) Simultaneous in vivo PET/MRI using fluorine-18 labeled Fe3O4@Al(OH)3 nanoparticles: comparison of nanoparticle and nanoparticle-labeled stem cell distribution. EJNMMI Res 10(1):73. https://doi.org/10.1186/s13550-020-00655-9
Bensch F et al (2018a) (89)Zr-atezolizumab imaging as a non-invasive approach to assess clinical response to PD-L1 blockade in cancer. Nat Med 24(12):1852–1858. https://doi.org/10.1038/s41591-018-0255-8
Bensch F et al (2018b) Comparative biodistribution analysis across four different (89)Zr-monoclonal antibody tracers-the first step towards an imaging warehouse. Theranostics 8(16):4295–4304. https://doi.org/10.7150/thno.26370
Bergstrom M (2017) The use of microdosing in the development of small organic and protein therapeutics. J Nucl Med 58(8):1188–1195. https://doi.org/10.2967/jnumed.116.188037
Boerman OC, Oyen WJ (2011) Immuno-PET of cancer: a revival of antibody imaging. J Nucl Med 52(8):1171–1172. https://doi.org/10.2967/jnumed.111.089771
Boros E, Packard AB (2019) Radioactive transition metals for imaging and therapy. Chem Rev 119(2):870–901. https://doi.org/10.1021/acs.chemrev.8b00281
Bouleau A, Lebon V, Truillet C (2021a) PET imaging of immune checkpoint proteins in oncology. Pharmacol Ther 222:107786. https://doi.org/10.1016/j.pharmthera.2020.107786
Bouleau A et al (2021b) Optimizing immunoPET imaging of tumor PD-L1 expression: pharmacokinetics, biodistribution and dosimetric comparisons of (89)Zr-labeled anti-PD-L1 antibody formats. J Nucl Med 63:1259. https://doi.org/10.2967/jnumed.121.262967
Bournazos S, Wang TT, Dahan R, Maamary J, Ravetch JV (2017) Signaling by antibodies: recent progress. Annu Rev Immunol 35:285–311. https://doi.org/10.1146/annurev-immunol-051116-052433
Bridgwater C et al (2020) (89)Zr-labeled anti-PD-L1 antibody fragment for evaluating in vivo PD-L1 levels in melanoma mouse model. Cancer Biother Radiopharm 35(8):549–557. https://doi.org/10.1089/cbr.2019.3056
Bridoux J et al (2020) Anti-human PD-L1 nanobody for immuno-PET imaging: validation of a conjugation strategy for clinical translation. Biomolecules 10(10):1388. https://doi.org/10.3390/biom10101388
Brinkmann U, Kontermann RE (2017) The making of bispecific antibodies. MAbs 9(2):182–212. https://doi.org/10.1080/19420862.2016.1268307
Butte MJ, Keir ME, Phamduy TB, Sharpe AH, Freeman GJ (2007) Programmed death-1 ligand 1 interacts specifically with the B7-1 costimulatory molecule to inhibit T cell responses. Immunity 27(1):111–122. https://doi.org/10.1016/j.immuni.2007.05.016
Cha E, Wallin J, Kowanetz M (2015) PD-L1 inhibition with MPDL3280A for solid tumors. Semin Oncol 42(3):484–487. https://doi.org/10.1053/j.seminoncol.2015.02.002
Chatterjee S et al (2017) Rapid PD-L1 detection in tumors with PET using a highly specific peptide. Biochem Biophys Res Commun 483(1):258–263. https://doi.org/10.1016/j.bbrc.2016.12.156
Cole EL et al (2017) Radiosynthesis and preclinical PET evaluation of (89)Zr-nivolumab (BMS-936558) in healthy non-human primates. Bioorg Med Chem 25(20):5407–5414. https://doi.org/10.1016/j.bmc.2017.07.066
Couzin-Frankel J (2013) Breakthrough of the year 2013. Cancer immunotherapy. Science 342(6165):1432–1433. https://doi.org/10.1126/science.342.6165.1432
de Jong M, Essers J, van Weerden WM (2014) Imaging preclinical tumour models: improving translational power. Nat Rev Cancer 14(7):481–493. https://doi.org/10.1038/nrc3751
De Silva RA et al (2018) Peptide-based (68)Ga-PET radiotracer for imaging PD-L1 expression in cancer. Mol Pharm 15(9):3946–3952. https://doi.org/10.1021/acs.molpharmaceut.8b00399
den Hollander MW et al (2015) TGF-beta antibody uptake in recurrent high-grade glioma imaged with 89Zr-Fresolimumab PET. J Nucl Med 56(9):1310–1314. https://doi.org/10.2967/jnumed.115.154401
Dijkers EC et al (2009) Development and characterization of clinical-grade 89Zr-trastuzumab for HER2/neu immunoPET imaging. J Nucl Med 50(6):974–981. https://doi.org/10.2967/jnumed.108.060392
Dijkers EC et al (2010) Biodistribution of 89Zr-trastuzumab and PET imaging of HER2-positive lesions in patients with metastatic breast cancer. Clin Pharmacol Ther 87(5):586–592. https://doi.org/10.1038/clpt.2010.12
Divgi CR et al (2007) Preoperative characterisation of clear-cell renal carcinoma using iodine-124-labelled antibody chimeric G250 (124I-cG250) and PET in patients with renal masses: a phase I trial. Lancet Oncol 8(4):304–310. https://doi.org/10.1016/S1470-2045(07)70044-X
Divgi CR et al (2013) Positron emission tomography/computed tomography identification of clear cell renal cell carcinoma: results from the REDECT trial. J Clin Oncol 31(2):187–194. https://doi.org/10.1200/JCO.2011.41.2445
Donnelly DJ et al (2018) Synthesis and biologic evaluation of a novel (18)F-labeled adnectin as a PET radioligand for imaging PD-L1 expression. J Nucl Med 59(3):529–535. https://doi.org/10.2967/jnumed.117.199596
Dovedi SJ et al (2021) Design and efficacy of a monovalent bispecific PD-1/CTLA4 antibody that enhances CTLA4 blockade on PD-1(+) activated T cells. Cancer Discov 11(5):1100–1117. https://doi.org/10.1158/2159-8290.CD-20-1445
Du Y et al (2022) (99m)Tc-labeled peptide targeting interleukin 13 receptor alpha 2 for tumor imaging in a cervical cancer mouse model. Ann Nucl Med. https://doi.org/10.1007/s12149-022-01715-x
Eder M et al (2010) 68Ga-labelled recombinant antibody variants for immuno-PET imaging of solid tumours. Eur J Nucl Med Mol Imaging 37(7):1397–1407. https://doi.org/10.1007/s00259-010-1392-6
Ehlerding EB et al (2019) Antibody and fragment-based PET imaging of CTLA-4+ T-cells in humanized mouse models. Am J Cancer Res 9(1):53–63. [Online]. Available: https://www.ncbi.nlm.nih.gov/pubmed/30755811
England CG et al (2018) (89)Zr-labeled nivolumab for imaging of T-cell infiltration in a humanized murine model of lung cancer. Eur J Nucl Med Mol Imaging 45(1):110–120. https://doi.org/10.1007/s00259-017-3803-4
Esfahani K, Roudaia L, Buhlaiga N, Del Rincon SV, Papneja N, Miller WH Jr (2020) A review of cancer immunotherapy: from the past, to the present, to the future. Curr Oncol 27(Suppl 2):S87–S97. https://doi.org/10.3747/co.27.5223
Farwell MD et al (2021) CD8-targeted PET imaging of tumor infiltrating T cells in patients with cancer: a phase I first-in-human study of (89)Zr-Df-IAB22M2C, a radiolabeled anti-CD8 minibody. J Nucl Med 63:720. https://doi.org/10.2967/jnumed.121.262485
Fridman WH, Zitvogel L, Sautes-Fridman C, Kroemer G (2017) The immune contexture in cancer prognosis and treatment. Nat Rev Clin Oncol 14(12):717–734. https://doi.org/10.1038/nrclinonc.2017.101
Gallamini A, Zwarthoed C, Borra A (2014) Positron emission tomography (PET) in oncology. Cancers (Basel) 6(4):1821–1889. https://doi.org/10.3390/cancers6041821
Gandevia SC, Hobbs SF (1990) Cardiovascular responses to static exercise in man: central and reflex contributions. J Physiol 430:105–117. https://doi.org/10.1113/jphysiol.1990.sp018284
Gerlinger M et al (2012) Intratumor heterogeneity and branched evolution revealed by multiregion sequencing. N Engl J Med 366(10):883–892. https://doi.org/10.1056/NEJMoa1113205
Ghiotto M et al (2010) PD-L1 and PD-L2 differ in their molecular mechanisms of interaction with PD-1. Int Immunol 22(8):651–660. https://doi.org/10.1093/intimm/dxq049
Giesen D et al (2020) Probody therapeutic design of (89)Zr-CX-072 promotes accumulation in PD-L1-expressing tumors Compared to normal murine lymphoid tissue. Clin Cancer Res 26(15):3999–4009. https://doi.org/10.1158/1078-0432.CCR-19-3137
Goldenberg DM, Nabi HA (1999) Breast cancer imaging with radiolabeled antibodies. Semin Nucl Med 29(1):41–48. https://doi.org/10.1016/s0001-2998(99)80028-2
Graff CP, Wittrup KD (2003) Theoretical analysis of antibody targeting of tumor spheroids: importance of dosage for penetration, and affinity for retention. Cancer Res 63(6):1288–1296. [Online]. Available: https://www.ncbi.nlm.nih.gov/pubmed/12649189
Graham MM (2012) Clinical molecular imaging with radiotracers: current status. Med Princ Pract 21(3):197–208. https://doi.org/10.1159/000333552
Griessinger CM et al (2020) The PET-tracer (89)Zr-Df-IAB22M2C enables monitoring of intratumoral CD8 T-cell infiltrates in tumor-bearing humanized mice after T-cell bispecific antibody treatment. Cancer Res 80(13):2903–2913. https://doi.org/10.1158/0008-5472.CAN-19-3269
Hafeez U, Gan HK, Scott AM (2018) Monoclonal antibodies as immunomodulatory therapy against cancer and autoimmune diseases. Curr Opin Pharmacol 41:114–121. https://doi.org/10.1016/j.coph.2018.05.010
Hamid O et al (2013) Safety and tumor responses with lambrolizumab (anti-PD-1) in melanoma. N Engl J Med 369(2):134–144. https://doi.org/10.1056/NEJMoa1305133
Heskamp S et al (2015) Noninvasive imaging of tumor PD-L1 expression using radiolabeled anti-PD-L1 antibodies. Cancer Res 75(14):2928–2936. https://doi.org/10.1158/0008-5472.CAN-14-3477
Hmadcha A, Martin-Montalvo A, Gauthier BR, Soria B, Capilla-Gonzalez V (2020) Therapeutic potential of mesenchymal stem cells for cancer therapy. Front Bioeng Biotechnol 8:43. https://doi.org/10.3389/fbioe.2020.00043
Hodi FS et al (2010) Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med 363(8):711–723. https://doi.org/10.1056/NEJMoa1003466
Hu S et al (1996) Minibody: a novel engineered anti-carcinoembryonic antigen antibody fragment (single-chain Fv-CH3) which exhibits rapid, high-level targeting of xenografts. Cancer Res 56(13):3055–3061. [Online]. Available: https://www.ncbi.nlm.nih.gov/pubmed/8674062
Huisman MC et al (2020) Quantification of PD-L1 expression with (18)F-BMS-986192 PET/CT in patients with advanced-stage non-small cell lung cancer. J Nucl Med 61(10):1455–1460. https://doi.org/10.2967/jnumed.119.240895
Hutloff A et al (1999) ICOS is an inducible T-cell co-stimulator structurally and functionally related to CD28. Nature 397(6716):263–266. https://doi.org/10.1038/16717
Inthagard J, Edwards J, Roseweir AK (2019) Immunotherapy: enhancing the efficacy of this promising therapeutic in multiple cancers. Clin Sci (Lond) 133(2):181–193. https://doi.org/10.1042/CS20181003
Jagoda EM et al (2019) Immuno-PET imaging of the programmed cell death-1 ligand (PD-L1) using a zirconium-89 labeled therapeutic antibody, avelumab. Mol Imaging 18:1536012119829986. https://doi.org/10.1177/1536012119829986
Jandus C et al (2009) Tumor antigen-specific FOXP3+ CD4 T cells identified in human metastatic melanoma: peptide vaccination results in selective expansion of Th1-like counterparts. Cancer Res 69(20):8085–8093. https://doi.org/10.1158/0008-5472.CAN-09-2226
Jauw YW et al (2016) Immuno-positron emission tomography with zirconium-89-labeled monoclonal antibodies in oncology: what can we learn from initial clinical trials? Front Pharmacol 7:131. https://doi.org/10.3389/fphar.2016.00131
Jiang J et al (2021) Noninvasive evaluation of PD-L1 expression using Copper 64 labeled peptide WL12 by micro-PET imaging in Chinese hamster ovary cell tumor model. Bioorg Med Chem Lett 40:127901. https://doi.org/10.1016/j.bmcl.2021.127901
Joseph RW et al (2018) Baseline tumor size is an independent prognostic factor for overall survival in patients with melanoma treated with pembrolizumab. Clin Cancer Res 24(20):4960–4967. https://doi.org/10.1158/1078-0432.CCR-17-2386
Jung KH, Park JW, Lee JH, Moon SH, Cho YS, Lee KH (2021) (89)Zr-labeled anti-PD-L1 antibody PET monitors gemcitabine therapy-induced modulation of tumor PD-L1 expression. J Nucl Med 62(5):656–664. https://doi.org/10.2967/jnumed.120.250720
Kaira K, Kuji I, Kagamu H (2021) Value of (18)F-FDG-PET to predict PD-L1 expression and outcomes of PD-1 inhibition therapy in human cancers. Cancer Imaging 21(1):11. https://doi.org/10.1186/s40644-021-00381-y
Kantoff PW et al (2010) Sipuleucel-T immunotherapy for castration-resistant prostate cancer. N Engl J Med 363(5):411–422. https://doi.org/10.1056/NEJMoa1001294
Kather JN et al (2018) Topography of cancer-associated immune cells in human solid tumors. Elife 7:e36967. https://doi.org/10.7554/eLife.36967
Kelly MP et al (2021) Preclinical PET imaging with the novel human antibody (89)Zr-DFO-REGN3504 sensitively detects PD-L1 expression in tumors and normal tissues. J Immunother Cancer 9(1):e002025. https://doi.org/10.1136/jitc-2020-002025
Kenanova V, Wu AM (2006) Tailoring antibodies for radionuclide delivery. Expert Opin Drug Deliv 3(1):53–70. https://doi.org/10.1517/17425247.3.1.53
Keu KV et al (2017) Reporter gene imaging of targeted T cell immunotherapy in recurrent glioma. Sci Transl Med 9(373):eaag2196. https://doi.org/10.1126/scitranslmed.aag2196
Kimura RH, Cheng Z, Gambhir SS, Cochran JR (2009) Engineered knottin peptides: a new class of agents for imaging integrin expression in living subjects. Cancer Res 69(6):2435–2442. https://doi.org/10.1158/0008-5472.CAN-08-2495
Kist de Ruijter L et al (2021) First-in-human study of the biodistribution and pharmacokinetics of (89)Zr-CX-072, a novel immunopet tracer based on an anti-PD-L1 probody. Clin Cancer Res. https://doi.org/10.1158/1078-0432.CCR-21-0453
Kluger HM et al (2015) Characterization of PD-L1 expression and associated T-cell infiltrates in metastatic melanoma samples from variable anatomic sites. Clin Cancer Res 21(13):3052–3060. https://doi.org/10.1158/1078-0432.CCR-14-3073
Kluger HM et al (2017) PD-L1 studies across tumor types, its differential expression and predictive value in patients treated with immune checkpoint inhibitors. Clin Cancer Res 23(15):4270–4279. https://doi.org/10.1158/1078-0432.CCR-16-3146
Knowles SM, Wu AM (2012) Advances in immuno-positron emission tomography: antibodies for molecular imaging in oncology. J Clin Oncol 30(31):3884–3892. https://doi.org/10.1200/JCO.2012.42.4887
Kontermann RE (2011) Strategies for extended serum half-life of protein therapeutics. Curr Opin Biotechnol 22(6):868–876. https://doi.org/10.1016/j.copbio.2011.06.012
Kostelnik TI, Orvig C (2019) Radioactive main group and rare earth metals for imaging and therapy. Chem Rev 119(2):902–956. https://doi.org/10.1021/acs.chemrev.8b00294
Kraeber-Bodere F et al (2015) Tumor immunotargeting using innovative radionuclides. Int J Mol Sci 16(2):3932–3954. https://doi.org/10.3390/ijms16023932
Kramer-Marek G, Kiesewetter DO, Capala J (2009) Changes in HER2 expression in breast cancer xenografts after therapy can be quantified using PET and (18)F-labeled affibody molecules. J Nucl Med 50(7):1131–1139. https://doi.org/10.2967/jnumed.108.057695
Krasniqi A et al (2018) Same-day imaging using small proteins: clinical experience and translational prospects in oncology. J Nucl Med 59(6):885–891. https://doi.org/10.2967/jnumed.117.199901
Kristensen LK et al (2019) CD4(+) and CD8a(+) PET imaging predicts response to novel PD-1 checkpoint inhibitor: studies of Sym021 in syngeneic mouse cancer models. Theranostics 9(26):8221–8238. https://doi.org/10.7150/thno.37513
Lamberts LE et al (2015) Antibody positron emission tomography imaging in anticancer drug development. J Clin Oncol 33(13):1491–1504. https://doi.org/10.1200/JCO.2014.57.8278
Larkin J et al (2015) Combined nivolumab and ipilimumab or monotherapy in untreated melanoma. N Engl J Med 373(1):23–34. https://doi.org/10.1056/NEJMoa1504030
Lee L, Gupta M, Sahasranaman S (2016) Immune checkpoint inhibitors: an introduction to the next-generation cancer immunotherapy. J Clin Pharmacol 56(2):157–169. https://doi.org/10.1002/jcph.591
Levi J et al (2019) Imaging of activated T cells as an early predictor of immune response to anti-PD-1 therapy. Cancer Res 79(13):3455–3465. https://doi.org/10.1158/0008-5472.CAN-19-0267
Li D et al (2018) Immuno-PET imaging of (89)Zr labeled anti-PD-L1 domain antibody. Mol Pharm 15(4):1674–1681. https://doi.org/10.1021/acs.molpharmaceut.8b00062
Li M et al (2020) In vivo characterization of PD-L1 expression in breast cancer by immuno-PET with (89)Zr-labeled avelumab. Am J Transl Res 12(5):1862–1872. [Online]. Available: https://www.ncbi.nlm.nih.gov/pubmed/32509182
Li W et al (2021) PET/CT imaging of (89)Zr-N-sucDf-pembrolizumab in healthy cynomolgus monkeys. Mol Imaging Biol 23(2):250–259. https://doi.org/10.1007/s11307-020-01558-w
Liberini V et al (2021) The future of cancer diagnosis, treatment and surveillance: a systemic review on immunotherapy and immuno-PET radiotracers. Molecules 26(8):2201. https://doi.org/10.3390/molecules26082201
Lipovsek D (2011) Adnectins: engineered target-binding protein therapeutics. Protein Eng Des Sel 24(1–2):3–9. https://doi.org/10.1093/protein/gzq097
Liu Q et al (2021) Immuno-PET imaging of (68)Ga-labeled nanobody Nb109 for dynamic monitoring the PD-L1 expression in cancers. Cancer Immunol Immunother 70(6):1721–1733. https://doi.org/10.1007/s00262-020-02818-y
Liu H et al (2022) A novel small cyclic peptide-based (68)Ga-radiotracer for positron emission tomography imaging of PD-L1 expression in tumors. Mol Pharm 19(1):138–147. https://doi.org/10.1021/acs.molpharmaceut.1c00694
Lohrmann C et al (2019) Retooling a blood-based biomarker: phase I assessment of the high-affinity CA19–9 antibody HuMab-5B1 for immuno-PET imaging of pancreatic cancer. Clin Cancer Res 25(23):7014–7023. https://doi.org/10.1158/1078-0432.CCR-18-3667
Long NE et al (2018a) Linker engineering in anti-TAG-72 antibody fragments optimizes biophysical properties, serum half-life, and high-specificity tumor imaging. J Biol Chem 293(23):9030–9040. https://doi.org/10.1074/jbc.RA118.002538
Long GV et al (2018b) Combination nivolumab and ipilimumab or nivolumab alone in melanoma brain metastases: a multicentre randomised phase 2 study. Lancet Oncol 19(5):672–681. https://doi.org/10.1016/S1470-2045(18)30139-6
Luke JJ, Flaherty KT, Ribas A, Long GV (2017) Targeted agents and immunotherapies: optimizing outcomes in melanoma. Nat Rev Clin Oncol 14(8):463–482. https://doi.org/10.1038/nrclinonc.2017.43
Luo H, Hong H, Yang SP, Cai W (2014) Design and applications of bispecific heterodimers: molecular imaging and beyond. Mol Pharm 11(6):1750–1761. https://doi.org/10.1021/mp500115x
Lv G et al (2020) PET imaging of tumor PD-L1 expression with a highly specific nonblocking single-domain antibody. J Nucl Med 61(1):117–122. https://doi.org/10.2967/jnumed.119.226712
Lv G et al (2021) Promising potential of a (18)F-labelled small-molecular radiotracer to evaluate PD-L1 expression in tumors by PET imaging. Bioorg Chem 115:105294. https://doi.org/10.1016/j.bioorg.2021.105294
Marofi F et al (2021) CAR T cells in solid tumors: challenges and opportunities. Stem Cell Res Ther 12(1):81. https://doi.org/10.1186/s13287-020-02128-1
Martins F et al (2019) Adverse effects of immune-checkpoint inhibitors: epidemiology, management and surveillance. Nat Rev Clin Oncol 16(9):563–580. https://doi.org/10.1038/s41571-019-0218-0
Mastracci L et al (2020) Response to ipilimumab therapy in metastatic melanoma patients: potential relevance of CTLA-4(+) tumor infiltrating lymphocytes and their in situ localization. Cancer Immunol Immunother 69(4):653–662. https://doi.org/10.1007/s00262-020-02494-y
McQuerry JA, Chang JT, Bowtell DDL, Cohen A, Bild AH (2017) Mechanisms and clinical implications of tumor heterogeneity and convergence on recurrent phenotypes. J Mol Med (Berl) 95(11):1167–1178. https://doi.org/10.1007/s00109-017-1587-4
Menke-van der Houven CW et al (2015) 89Zr-cetuximab PET imaging in patients with advanced colorectal cancer. Oncotarget 6(30):30384–30393. https://doi.org/10.18632/oncotarget.4672
Minn I et al (2019) Imaging CAR T cell therapy with PSMA-targeted positron emission tomography. Sci Adv 5(7):eaaw5096. https://doi.org/10.1126/sciadv.aaw5096
Minutoli F, Parisi S, Laudicella R, Pergolizzi S, Baldari S (2022) 18F-FDG PET/CT imaging of immune checkpoint inhibitor-related “retroperitoneal panniculitis”. Clin Nucl Med 47(1):e39–e40. https://doi.org/10.1097/RLU.0000000000003806
Morris O, Fairclough M, Grigg J, Prenant C, McMahon A (2019) A review of approaches to (18) F radiolabelling affinity peptides and proteins. J Labelled Comp Radiopharm 62(1):4–23. https://doi.org/10.1002/jlcr.3634
Muylle K et al (2015) Tumour targeting and radiation dose of radioimmunotherapy with (90)Y-rituximab in CD20+ B-cell lymphoma as predicted by (89)Zr-rituximab immuno-PET: impact of preloading with unlabelled rituximab. Eur J Nucl Med Mol Imaging 42(8):1304–1314. https://doi.org/10.1007/s00259-015-3025-6
Namavari M, Chang YF, Kusler B, Yaghoubi S, Mitchell BS, Gambhir SS (2011) Synthesis of 2′-deoxy-2′-[18F]fluoro-9-beta-D-arabinofuranosylguanine: a novel agent for imaging T-cell activation with PET. Mol Imaging Biol 13(5):812–818. https://doi.org/10.1007/s11307-010-0414-x
Natarajan A, **ong CY, Albrecht H, DeNardo GL, DeNardo SJ (2005) Characterization of site-specific ScFv PEGylation for tumor-targeting pharmaceuticals. Bioconjug Chem 16(1):113–121. https://doi.org/10.1021/bc0498121
Natarajan A, Du W, **ong CY, DeNardo GL, DeNardo SJ, Gervay-Hague J (2007) Construction of di-scFv through a trivalent alkyne-azide 1,3-dipolar cycloaddition. Chem Commun (Camb) 7:695–697. https://doi.org/10.1039/b611636a
Natarajan A, Hackel BJ, Gambhir SS (2013) A novel engineered anti-CD20 tracer enables early time PET imaging in a humanized transgenic mouse model of B-cell non-Hodgkins lymphoma. Clin Cancer Res 19(24):6820–6829. https://doi.org/10.1158/1078-0432.CCR-13-0626
Natarajan A, Patel CB, Habte F, Gambhir SS (2018) Dosimetry prediction for clinical translation of (64)Cu-pembrolizumab immunoPET targeting human PD-1 expression. Sci Rep 8(1):633. https://doi.org/10.1038/s41598-017-19123-x
Natarajan A, Patel CB, Ramakrishnan S, Panesar PS, Long SR, Gambhir SS (2019) A novel engineered small protein for positron emission tomography imaging of human programmed death ligand-1: validation in mouse models and human cancer tissues. Clin Cancer Res 25(6):1774–1785. https://doi.org/10.1158/1078-0432.CCR-18-1871
Niemeijer AN et al (2018) Whole body PD-1 and PD-L1 positron emission tomography in patients with non-small-cell lung cancer. Nat Commun 9(1):4664. https://doi.org/10.1038/s41467-018-07131-y
Nienhuis PH et al (2021) (18)F-BMS986192 PET imaging of PD-L1 in metastatic melanoma patients with brain metastases treated with immune checkpoint inhibitors. A pilot study. J Nucl Med. https://doi.org/10.2967/jnumed.121.262368
Oehlke E et al (2015) Production of Y-86 and other radiometals for research purposes using a solution target system. Nucl Med Biol 42(11):842–849. https://doi.org/10.1016/j.nucmedbio.2015.06.005
Olafsen T et al (2004) Covalent disulfide-linked anti-CEA diabody allows site-specific conjugation and radiolabeling for tumor targeting applications. Protein Eng Des Sel 17(1):21–27. https://doi.org/10.1093/protein/gzh009
Orlova A, Wallberg H, Stone-Elander S, Tolmachev V (2009) On the selection of a tracer for PET imaging of HER2-expressing tumors: direct comparison of a 124I-labeled affibody molecule and trastuzumab in a murine xenograft model. J Nucl Med 50(3):417–425. https://doi.org/10.2967/jnumed.108.057919
Pandit-Taskar N et al (2020) First-in-humans imaging with (89)Zr-Df-IAB22M2C anti-CD8 minibody in patients with solid malignancies: preliminary pharmacokinetics, biodistribution, and lesion targeting. J Nucl Med 61(4):512–519. https://doi.org/10.2967/jnumed.119.229781
Pardoll DM (2012) The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer 12(4):252–264. https://doi.org/10.1038/nrc3239
Philpott GW et al (1995) RadioimmunoPET: detection of colorectal carcinoma with positron-emitting copper-64-labeled monoclonal antibody. J Nucl Med 36(10):1818–1824. [Online]. Available: https://www.ncbi.nlm.nih.gov/pubmed/7562049
Postow MA et al (2015) Nivolumab and ipilimumab versus ipilimumab in untreated melanoma. N Engl J Med 372(21):2006–2017. https://doi.org/10.1056/NEJMoa1414428
Price EW, Orvig C (2014) Matching chelators to radiometals for radiopharmaceuticals. Chem Soc Rev 43(1):260–290. https://doi.org/10.1039/c3cs60304k
Qin S et al (2021) A preclinical study: correlation between PD-L1 PET imaging and the prediction of therapy efficacy of MC38 tumor with (68)Ga-labeled PD-L1 targeted nanobody. Aging (Albany NY) 13(9):13006–13022. https://doi.org/10.18632/aging.202981
Rashidian M et al (2017) Predicting the response to CTLA-4 blockade by longitudinal noninvasive monitoring of CD8 T cells. J Exp Med 214(8):2243–2255. https://doi.org/10.1084/jem.20161950
Rashidian M et al (2019) Immuno-PET identifies the myeloid compartment as a key contributor to the outcome of the antitumor response under PD-1 blockade. Proc Natl Acad Sci U S A 116(34):16971–16980. https://doi.org/10.1073/pnas.1905005116
Receptor CA (2016) Chimeric antigen receptor-modified T cells in chronic lymphoid leukemia; chimeric antigen receptor-modified T cells for acute lymphoid leukemia; chimeric antigen receptor T cells for sustained remissions in leukemia. N Engl J Med 374(10):998. https://doi.org/10.1056/NEJMx160005
Ren J et al (2021) PET imaging facilitates antibody screening for synergistic radioimmunotherapy with a (177)Lu-labeled alphaPD-L1 antibody. Theranostics 11(1):304–315. https://doi.org/10.7150/thno.45540
Ribas A, Wolchok JD (2018) Cancer immunotherapy using checkpoint blockade. Science 359(6382):1350–1355. https://doi.org/10.1126/science.aar4060
Rios X et al (2019) Immuno-PET imaging and pharmacokinetics of an Anti-CEA scFv-based trimerbody and its monomeric counterpart in human gastric carcinoma-bearing mice. Mol Pharm 16(3):1025–1035. https://doi.org/10.1021/acs.molpharmaceut.8b01006
Robert C et al (2011) Ipilimumab plus dacarbazine for previously untreated metastatic melanoma. N Engl J Med 364(26):2517–2526. https://doi.org/10.1056/NEJMoa1104621
Robu S et al (2021) Synthesis and preclinical evaluation of a (68)Ga-labeled adnectin, (68)Ga-BMS-986192, as a PET agent for imaging PD-L1 expression. J Nucl Med 62(9):1228–1234. https://doi.org/10.2967/jnumed.120.258384
Rosell R et al (2013) Adaptive resistance to targeted therapies in cancer. Transl Lung Cancer Res 2(3):152–159. https://doi.org/10.3978/j.issn.2218-6751.2012.12.08
Rowe SP, Pomper MG (2021) Molecular imaging in oncology: current impact and future directions. CA Cancer J Clin 72:333. https://doi.org/10.3322/caac.21713
Rowshanravan B, Halliday N, Sansom DM (2018) CTLA-4: a moving target in immunotherapy. Blood 131(1):58–67. https://doi.org/10.1182/blood-2017-06-741033
Rubins DJ et al (2021) In vivo evaluation and dosimetry estimate for a high affinity affibody PET tracer targeting PD-L1. Mol Imaging Biol 23(2):241–249. https://doi.org/10.1007/s11307-020-01544-2
Rudnick SI et al (2011) Influence of affinity and antigen internalization on the uptake and penetration of Anti-HER2 antibodies in solid tumors. Cancer Res 71(6):2250–2259. https://doi.org/10.1158/0008-5472.CAN-10-2277
Runcie K, Budman DR, John V, Seetharamu N (2018) Bi-specific and tri-specific antibodies- the next big thing in solid tumor therapeutics. Mol Med 24(1):50. https://doi.org/10.1186/s10020-018-0051-4
Sato N et al (2020) In vivo tracking of adoptively transferred natural killer cells in rhesus macaques using (89)zirconium-oxine cell labeling and PET imaging. Clin Cancer Res 26(11):2573–2581. https://doi.org/10.1158/1078-0432.CCR-19-2897
Sato M et al (2021) Predictive value of 3′-deoxy-3′-(18)F-fluorothymidine PET in the early response to anti-programmed death-1 therapy in patients with advanced non-small cell lung cancer. J Immunother Cancer 9(7):e003079. https://doi.org/10.1136/jitc-2021-003079
Schiefner A, Skerra A (2015) The menagerie of human lipocalins: a natural protein scaffold for molecular recognition of physiological compounds. Acc Chem Res 48(4):976–985. https://doi.org/10.1021/ar5003973
Sehlin D, Syvanen S (2019) Engineered antibodies: new possibilities for brain PET? Eur J Nucl Med Mol Imaging 46(13):2848–2858. https://doi.org/10.1007/s00259-019-04426-0
Seo JW et al (2018) CD8(+) T-cell density imaging with (64)cu-labeled Cys-diabody informs immunotherapy protocols. Clin Cancer Res 24(20):4976–4987. https://doi.org/10.1158/1078-0432.CCR-18-0261
Shaffer TM, Aalipour A, Schurch CM, Gambhir SS (2020) PET imaging of the natural killer cell activation receptor NKp30. J Nucl Med 61(9):1348–1354. https://doi.org/10.2967/jnumed.119.233163
Shaffer T, Natarajan A, Gambhir SS (2021) PET imaging of TIGIT expression on tumor-infiltrating lymphocytes. Clin Cancer Res 27(7):1932–1940. https://doi.org/10.1158/1078-0432.CCR-20-2725
Stahl S, Graslund T, Eriksson Karlstrom A, Frejd FY, Nygren PA, Lofblom J (2017) Affibody molecules in biotechnological and medical applications. Trends Biotechnol 35(8):691–712. https://doi.org/10.1016/j.tibtech.2017.04.007
Sun H et al (2016) ImmunoPET for assessing the differential uptake of a CD146-specific monoclonal antibody in lung cancer. Eur J Nucl Med Mol Imaging 43(12):2169–2179. https://doi.org/10.1007/s00259-016-3442-1
Sun C, Mezzadra R, Schumacher TN (2018) Regulation and function of the PD-L1 checkpoint. Immunity 48(3):434–452. https://doi.org/10.1016/j.immuni.2018.03.014
Suzuki T et al (2010) Importance of neonatal FcR in regulating the serum half-life of therapeutic proteins containing the Fc domain of human IgG1: a comparative study of the affinity of monoclonal antibodies and Fc-fusion proteins to human neonatal FcR. J Immunol 184(4):1968–1976. https://doi.org/10.4049/jimmunol.0903296
Tabassum DP, Polyak K (2015) Tumorigenesis: it takes a village. Nat Rev Cancer 15(8):473–483. https://doi.org/10.1038/nrc3971
Tamura K et al (2013) 64Cu-DOTA-trastuzumab PET imaging in patients with HER2-positive breast cancer. J Nucl Med 54(11):1869–1875. https://doi.org/10.2967/jnumed.112.118612
Tarhini AA (2013) Tremelimumab: a review of development to date in solid tumors. Immunotherapy 5(3):215–229. https://doi.org/10.2217/imt.13.9
Tavare R et al (2014) Engineered antibody fragments for immuno-PET imaging of endogenous CD8+ T cells in vivo. Proc Natl Acad Sci U S A 111(3):1108–1113. https://doi.org/10.1073/pnas.1316922111
Thurber GM, Zajic SC, Wittrup KD (2007) Theoretic criteria for antibody penetration into solid tumors and micrometastases. J Nucl Med 48(6):995–999. https://doi.org/10.2967/jnumed.106.037069
Thurber GM, Schmidt MM, Wittrup KD (2008) Antibody tumor penetration: transport opposed by systemic and antigen-mediated clearance. Adv Drug Deliv Rev 60(12):1421–1434. https://doi.org/10.1016/j.addr.2008.04.012
Tunger A et al (2019) The evolving landscape of biomarkers for anti-PD-1 or anti-PD-L1 therapy. J Clin Med 8(10):1534. https://doi.org/10.3390/jcm8101534
Vaddepally RK, Kharel P, Pandey R, Garje R, Chandra AB (2020) Review of indications of FDA-approved immune checkpoint inhibitors per NCCN guidelines with the level of evidence. Cancers (Basel) 12(3):738. https://doi.org/10.3390/cancers12030738
van Asselt SJ et al (2014) Everolimus reduces (89)Zr-bevacizumab tumor uptake in patients with neuroendocrine tumors. J Nucl Med 55(7):1087–1092. https://doi.org/10.2967/jnumed.113.129056
van der Veen EL, Giesen D, Pot-de Jong L, Jorritsma-Smit A, De Vries EGE, Lub-de Hooge MN (2020) (89)Zr-pembrolizumab biodistribution is influenced by PD-1-mediated uptake in lymphoid organs. J Immunother Cancer 8(2). https://doi.org/10.1136/jitc-2020-000938
Vento J et al (2019) PD-L1 detection using (89)Zr-atezolizumab immuno-PET in renal cell carcinoma tumorgrafts from a patient with favorable nivolumab response. J Immunother Cancer 7(1):144. https://doi.org/10.1186/s40425-019-0607-z
Ward ES, Zhou J, Ghetie V, Ober RJ (2003) Evidence to support the cellular mechanism involved in serum IgG homeostasis in humans. Int Immunol 15(2):187–195. https://doi.org/10.1093/intimm/dxg018
Weide B et al (2016) Baseline biomarkers for outcome of melanoma patients treated with pembrolizumab. Clin Cancer Res 22(22):5487–5496. https://doi.org/10.1158/1078-0432.CCR-16-0127
Weist MR et al (2018) PET of adoptively transferred chimeric antigen receptor T cells with (89)Zr-oxine. J Nucl Med 59(10):1531–1537. https://doi.org/10.2967/jnumed.117.206714
Wittrup KD, Thurber GM, Schmidt MM, Rhoden JJ (2012) Practical theoretic guidance for the design of tumor-targeting agents. Methods Enzymol 503:255–268. https://doi.org/10.1016/B978-0-12-396962-0.00010-0
Wu AM (2014) Engineered antibodies for molecular imaging of cancer. Methods 65(1):139–147. https://doi.org/10.1016/j.ymeth.2013.09.015
Wu R et al (2012) Adoptive T-cell therapy using autologous tumor-infiltrating lymphocytes for metastatic melanoma: current status and future outlook. Cancer J 18(2):160–175. https://doi.org/10.1097/PPO.0b013e31824d4465
Xenaki KT, Oliveira S, van Bergen En Henegouwen PMP (2017) Antibody or antibody fragments: implications for molecular imaging and targeted therapy of solid tumors. Front Immunol 8:1287. https://doi.org/10.3389/fimmu.2017.01287
**ao Z, Mayer AT, Nobashi TW, Gambhir SS (2020) ICOS is an indicator of T-cell-mediated response to cancer immunotherapy. Cancer Res 80(14):3023–3032. https://doi.org/10.1158/0008-5472.CAN-19-3265
Yu S et al (2017) Recent advances of bispecific antibodies in solid tumors. J Hematol Oncol 10(1):155. https://doi.org/10.1186/s13045-017-0522-z
Zhang Y, Zhang Z (2020) The history and advances in cancer immunotherapy: understanding the characteristics of tumor-infiltrating immune cells and their therapeutic implications. Cell Mol Immunol 17(8):807–821. https://doi.org/10.1038/s41423-020-0488-6
Zhao H et al (2021) ImmunoPET imaging of human CD8(+) T cells with novel (68)Ga-labeled nanobody companion diagnostic agents. J Nanobiotechnology 19(1):42. https://doi.org/10.1186/s12951-021-00785-9
Zhou X et al (2021) First-in-human evaluation of a PD-L1-binding peptide radiotracer in non-small cell lung cancer patients with PET. J Nucl Med 63:536. https://doi.org/10.2967/jnumed.121.262045
Ziv E, Durack JC, Solomon SB (2016) The importance of biopsy in the era of molecular medicine. Cancer J 22(6):418–422. https://doi.org/10.1097/PPO.0000000000000228
Zou Y et al (2021) Discordance of immunotherapy response predictive biomarkers between primary lesions and paired metastases in tumours: a systematic review and meta-analysis. EBioMedicine 63:103137. https://doi.org/10.1016/j.ebiom.2020.103137
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 Springer Nature Switzerland AG
About this entry
Cite this entry
Wynter, C., Murugesan, N., Natarajan, A. (2023). ImmunoPET Imaging of Immune Checkpoints to Assess Their Cancer Therapeutic Potential. In: Rezaei, N. (eds) Handbook of Cancer and Immunology. Springer, Cham. https://doi.org/10.1007/978-3-030-80962-1_170-1
Download citation
DOI: https://doi.org/10.1007/978-3-030-80962-1_170-1
Received:
Accepted:
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-80962-1
Online ISBN: 978-3-030-80962-1
eBook Packages: Springer Reference Biomedicine and Life SciencesReference Module Biomedical and Life Sciences