Abstract
Purpose
Nivolumab is a human monoclonal antibody specific for programmed cell death-1 (PD-1), a negative regulator of T-cell activation and response. Acting as an immune checkpoint inhibitor, nivolumab binds to PD-1 expressed on the surface of many immune cells and prevents ligation by its natural ligands. Nivolumab is only effective in a subset of patients, and there is limited evidence supporting its use for diagnostic, monitoring, or stratification purposes.
Methods
89Zr-Df-nivolumab was synthesized to map the biodistribution of PD-1-expressing tumor infiltrating T-cells in vivo using a humanized murine model of lung cancer. The tracer was developed by radiolabeling the antibody with the positron emitter zirconium-89 (89Zr). Imaging results were validated by ex vivo biodistribution studies, and PD-1 expression was validated by immunohistochemistry. Data obtained from PET imaging were used to determine human dosimetry estimations.
Results
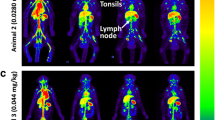
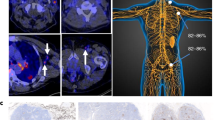
The tracer showed elevated binding to stimulated PD-1 expressing T-cells in vitro and in vivo. PET imaging of 89Zr-Df-nivolumab allowed for clear delineation of subcutaneous tumors through targeting of localized activated T-cells expressing PD-1 in the tumors and salivary glands of humanized A549 tumor-bearing mice. In addition to tumor uptake, salivary and lacrimal gland infiltration of T-cells was noticeably visible and confirmed via histological analysis.
Conclusions
These data support our claim that PD-1-targeted agents allow for tumor imaging in vivo, which may assist in the design and development of new immunotherapies. In the future, noninvasive imaging of immunotherapy biomarkers may assist in disease diagnostics, disease monitoring, and patient stratification.







Similar content being viewed by others
References
Philips GK, Atkins M. Therapeutic uses of anti-PD-1 and anti-PD-L1 antibodies. Int Immunol. 2015;27(1):39–46. doi:10.1093/intimm/dxu095.
Patel SP, Kurzrock R. PD-L1 expression as a predictive biomarker in cancer immunotherapy. Mol Cancer Ther. 2015;14(4):847–56. doi:10.1158/1535-7163.MCT-14-0983.
Keir ME, Butte MJ, Freeman GJ, Sharpe AH. PD-1 and its ligands in tolerance and immunity. Annu Rev Immunol. 2008;26:677–704. doi:10.1146/annurev.immunol.26.021607.090331.
Wang J, Yuan R, Song W, Sun J, Liu D, Li Z. PD-1, PD-L1 (B7-H1) and tumor-site immune modulation therapy: the historical perspective. J Hematol Oncol. 2017;10(1):34. doi:10.1186/s13045-017-0403-5.
Zou W, Wolchok JD, Chen L. PD-L1 (B7-H1) and PD-1 pathway blockade for cancer therapy: mechanisms, response biomarkers, and combinations. Sci Transl Med. 2016;8(328):328rv4. doi:10.1126/scitranslmed.aad7118.
Yuzefpolskiy Y, Baumann FM, Penny LA, Kalia V, Sarkar S. Signaling through PD-1 on CD8 T cells is critical for antigen-independent maintenance of immune memory. J Immunol. 2016;196(1 Supplement):129.6-.6.
Guo Y, Feng X, Jiang Y, Shi X, **ng X, Liu X, et al. PD1 blockade enhances cytotoxicity of in vitro expanded natural killer cells towards myeloma cells. Oncotarget. 2016;7(30):48360–74. doi:10.18632/oncotarget.10235.
Dai C, Lin F, Geng R, Ge X, Tang W, Chang J, et al. Implication of combined PD-L1/PD-1 blockade with cytokine-induced killer cells as a synergistic immunotherapy for gastrointestinal cancer. Oncotarget. 2016;7(9):10332–44. doi:10.18632/oncotarget.7243.
Ehlerding EB, England CG, McNeel DG, Cai W. Molecular imaging of immunotherapy targets in cancer. J Nucl Med. 2016;57(10):1487–92. doi:10.2967/jnumed.116.177493.
Heinzerling L, Kirchberger MC, Walter L, Schuler G. Predicting the response to anti-PD1 therapy in metastatic melanoma. Transl Cancer Res. 2016;5(3):S576–S9.
Daud AI, Loo K, Pauli ML, Sanchez-Rodriguez R, Sandoval PM, Taravati K, et al. Tumor immune profiling predicts response to anti-PD-1 therapy in human melanoma. J Clin Invest. 2016;126(9):3447–52. doi:10.1172/JCI87324.
Diggs LP, Hsueh EC. Utility of PD-L1 immunohistochemistry assays for predicting PD-1/PD-L1 inhibitor response. Biomark Res. 2017;5(1):12. doi:10.1186/s40364-017-0093-8.
Gangadhar TC, Salama AK. Clinical applications of PD-1-based therapy: a focus on pembrolizumab (MK-3475) in the management of melanoma and other tumor types. Onco Targets Ther. 2015;8:929–37. doi:10.2147/OTT.S53164.
Gettinger SN, Horn L, Gandhi L, Spigel DR, Antonia SJ, Rizvi NA, et al. Overall survival and long-term safety of nivolumab (anti-programmed death 1 antibody, BMS-936558, ONO-4538) in patients with previously treated advanced non-small cell lung cancer. J Clin Oncol. 2015;33(18):2004–12. doi:10.1200/JCO.2014.58.3708.
Michot JM, Bigenwald C, Champiat S, Collins M, Carbonnel F, Postel-Vinay S, et al. Immune-related adverse events with immune checkpoint blockade: a comprehensive review. Eur J Cancer. 2016;54:139–48. doi:10.1016/j.ejca.2015.11.016.
Gniadek TJ, Li QK, Tully E, Chatterjee S, Nimmagadda S, Gabrielson E. Heterogeneous expression of PD-L1 in pulmonary squamous cell carcinoma and adenocarcinoma: implications for assessment by small biopsy. Mod Pathol. 2017;30(4):530–8. doi:10.1038/modpathol.2016.213.
Brehm MA, Wiles MV, Greiner DL, Shultz LD. Generation of improved humanized mouse models for human infectious diseases. J Immunol Methods. 2014;410:3–17. doi:10.1016/j.jim.2014.02.011.
Blazar BR, Carreno BM, Panoskaltsis-Mortari A, Carter L, Iwai Y, Yagita H, et al. Blockade of programmed death-1 engagement accelerates graft-versus-host disease lethality by an IFN-gamma-dependent mechanism. J Immunol. 2003;171(3):1272–7.
Schilbach K, Schick J, Wehrmann M, Wollny G, Simon P, Schlegel PG, et al. PD-1-PD-L1 pathway is involved in suppressing alloreactivity of heart infiltrating t cells during murine gvhd across minor histocompatibility antigen barriers. Transplantation. 2007;84(2):214–22. doi:10.1097/01.tp.0000268074.77929.54.
Habicht A, Kewalaramani R, Vu MD, Demirci G, Blazar BR, Sayegh MH, et al. Striking dichotomy of PD-L1 and PD-L2 pathways in regulating alloreactive CD4(+) and CD8(+) T cells in vivo. Am J Transplant. 2007;7(12):2683–92. doi:10.1111/j.1600-6143.2007.01999.x.
Hettich M, Braun F, Bartholoma MD, Schirmbeck R, Niedermann G. High-resolution PET imaging with therapeutic antibody-based PD-1/PD-L1 checkpoint tracers. Theranostics. 2016;6(10):1629–40. doi:10.7150/thno.15253.
England CG, Ehlerding EB, Hernandez R, Rekoske BT, Graves SA, Sun H, et al. Preclinical pharmacokinetics and biodistribution studies of 89Zr-labeled pembrolizumab. J Nucl Med. 2017;58(1):162–8. doi:10.2967/jnumed.116.177857.
Hernandez R, Sun H, England CG, Valdovinos HF, Ehlerding EB, Barnhart TE, et al. CD146-targeted immunoPET and NIRF imaging of hepatocellular carcinoma with a dual-labeled monoclonal antibody. Theranostics. 2016;6(11):1918–33. doi:10.7150/thno.15568.
The 2007 Recommendations of the international commission on radiological protection. ICRP publication 103. Ann ICRP. 2007;37(2–4):1–332. doi:10.1016/j.icrp.2007.10.003.
Ikebuchi R, Konnai S, Okagawa T, Yokoyama K, Nakajima C, Suzuki Y, et al. Blockade of bovine PD-1 increases T cell function and inhibits bovine leukemia virus expression in B cells in vitro. Vet Res. 2013;44(1):59. doi:10.1186/1297-9716-44-59.
Soret M, Bacharach SL, Buvat I. Partial-volume effect in PET tumor imaging. J Nucl Med. 2007;48(6):932–45. doi:10.2967/jnumed.106.035774.
Cancer Facts & Figures 2016. American Cancer Society. 2016;Atlanta, GA.
Garon EB, Rizvi NA, Hui R, Leighl N, Balmanoukian AS, Eder JP, et al. Pembrolizumab for the treatment of non-small-cell lung cancer. N Engl J Med. 2015;372(21):2018–28. doi:10.1056/NEJMoa1501824.
Natarajan A, Mayer AT, Xu L, Reeves RE, Gano J, Gambhir SS. Novel radiotracer for ImmunoPET imaging of PD-1 checkpoint expression on tumor infiltrating lymphocytes. Bioconjug Chem. 2015;26(10):2062–9. doi:10.1021/acs.bioconjchem.5b00318.
Hayashi T. Dysfunction of lacrimal and salivary glands in Sjogren's syndrome: nonimmunologic injury in preinflammatory phase and mouse model. J Biomed Biotechnol. 2011;2011:407031. doi:10.1155/2011/407031.
Poluektova LY, Makarov E. Human peripheral blood lymphocyte reconstituion as a model of neuroinflammation associated with graft-versus-host disease. In: **ong H, Gendelman HE, editors. Current laboratory methods in neuroscience research. New York: Springer; 2014. p. 487–90.
Reynders K, De Ruysscher D. Tumor infiltrating lymphocytes in lung cancer: a new prognostic parameter. J Thorac Dis. 2016;8(8):E833-5. doi:10.21037/jtd.2016.07.75.
Acknowledgements
This work was supported, in part, by the University of Wisconsin - Madison, the National Institutes of Health (NIBIB/NCI 1R01CA169365, 1R01CA205101, 1R01EB021336, T32CA009206, T32GM008505, 5T32GM08349, P30CA014520), the National Science Foundation (DGE-1256259), the American Cancer Society (125246-RSG-13-099-01-CCE), the National Science Foundation of China (81401465, 51573096), and the Basic Research Program of Shenzhen (JCYJ20170412111100742, JCYJ20160422091238319).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Electronic supplementary material
ESM 1
(DOCX 14 kb)
Rights and permissions
About this article
Cite this article
England, C.G., Jiang, D., Ehlerding, E.B. et al. 89Zr-labeled nivolumab for imaging of T-cell infiltration in a humanized murine model of lung cancer. Eur J Nucl Med Mol Imaging 45, 110–120 (2018). https://doi.org/10.1007/s00259-017-3803-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-017-3803-4