Abstract
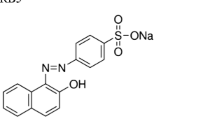
Azo dyes are a class of synthetic dyes with a wide variety of colors and shades with a vast application in industry. Due to their chemical structure, such dyes exert mutagenic and carcinogenic effects on living organisms. Problems caused by environmental contamination with azo dyes highlight the importance of develo** new treatment methods. Thus, biological treatment appears as a low-cost and eco-friendly technology, which uses the enzymatic production of some selected microorganisms for dye decolorization/degradation. Therefore, this work aims to select new bacterial isolates efficient in the mineralization of the azo dyes amaranth and congo red. For this purpose, soil contaminated with azo dyes was collected and 10 bacterial isolates capable of decolorizing both amaranth and congo red were selected. After evaluation of ideal pH and temperature of discoloration, two bacterial isolates belonging to the genus Citrobacter (LAJ01 and LAJ02) were selected aiming to evaluate the degree of dye mineralization. The temperature selected was 35 °C, and the pH was 7.5. LAJ01 reached 91.5% of amaranth discoloration, and LAJ02 and consortium (LAJ01 + LAJ02) reached 90.1% and 90.2% of amaranth discoloration, respectively. Phenol, indole, and N-glycylglycine were the main biodegradation products identified. For congo red dye, LAJ01 reached 93.6% of discoloration, LAJ02 reached 93.0% of discoloration, and the consortium reached 90.6% of discoloration. Phenol, indole, [1,1′-biphenyl]-3-amine, and benzidine were the main biodegradation products identified. Using as reference metabolites formed after discoloration, the isolate LAJ02 achieved a complete mineralization of amaranth. However, no bacterial isolates achieved the complete mineralization of congo red.




Similar content being viewed by others
References
Ahmad A, Sakr WA, Wahidur Rahman K (2010) Anticancer properties of indole compounds: mechanism of apoptosis induction and role in chemotherapy. Curr Drug Targets 11:652–666. https://doi.org/10.2174/138945010791170923
An SY, Min SK, Cha IH, Choi YL, Cho YS, Kim CH, Lee YC (2002) Decolorization of triphenylmethane and azo dyes by Citrobacter sp. Biotechnol Lett 24:1037–1040. https://doi.org/10.1023/A:1015610018103
Azbar N, Yonar T, Kestioglu K (2004) Comparison of various advanced oxidation processes and chemical treatment methods for COD and color removal from a polyester and acetate fiber dyeing effluent. Chemosphere 55:35–43. https://doi.org/10.1016/j.chemosphere.2003.10.046
Borenshtein D, Schauer DB (2006) The genus Citrobacter. Prokaryotes 6:90–98. https://doi.org/10.1007/0-387-30746-x_5
Brillas E, Martínez-Huitle CA (2015) Decontamination of wastewaters containing synthetic organic dyes by electrochemical methods. An updated review. Appl Catal B Environ 166–167:603–643. https://doi.org/10.1016/j.apcatb.2014.11.016
Chen KC, Wu JY, Liou DJ, Hwang SCJ (2003) Decolorization of the textile dyes by newly isolated bacterial strains. J Biotechnol 101:57–68. https://doi.org/10.1016/S0168-1656(02)00303-6
Chun J, Lee JH, Jung Y, Kim M, Kim S, Kim BK, Lim YW (2007) EzTaxon: a web-based tool for the identification of prokaryotes based on 16S ribosomal RNA gene sequences. Int J Syst Evol Microbiol 57:2259–2261
Chung KT (2016) Azo dyes and human health: a review. J Environ Sci Health Part C 34:233–261. https://doi.org/10.1080/10590501.2016.1236602
Gnanasekaran L, Hemamalini R, Saravanan R et al (2017) Synthesis and characterization of metal oxides (CeO2, CuO, NiO, Mn3O4, SnO2 and ZnO) nanoparticles as photo catalysts for degradation of textile dyes. J Photochem Photobiol B Biol 173:43–49. https://doi.org/10.1016/j.jphotobiol.2017.05.027
Gosetti F, Gianotti V, Polati S, Gennaro MC (2005) HPLC-MS degradation study of E110 Sunset Yellow FCF in a commercial beverage. J Chromatogr A 1090:107–115. https://doi.org/10.1016/j.chroma.2005.07.024
Granada CE, Beneduzi A, Lisboa BB et al (2015) Multilocus sequence analysis reveals taxonomic differences among Bradyrhizobium sp. symbionts of Lupinus albescens plants growing in arenized and non-arenized areas. Syst Appl Microbiol 38:323–329. https://doi.org/10.1016/j.syapm.2015.03.009
Gupta VK, Saravanan R, Agarwal S et al (2017) Degradation of azo dyes under different wavelengths of UV light with chitosan-SnO2 nanocomposites. J Mol Liq 232:423–430. https://doi.org/10.1016/j.molliq.2017.02.095
Hall TA (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucl Acids Symp Ser 4:95–98
Hong Y, Guo J, Xu Z, Mo C, Xu M, Sun G (2007) Reduction and partial degradation mechanisms of naphthylaminesulfonic azo dye amaranth by Shewanella decolorationis S12. Appl Microbiol Biotechnol 75:647–654. https://doi.org/10.1007/s00253-007-0838-7
Imran M, Arshad M, Negm F, Khalid A, Shaharoona B, Hussain S, Mahmood Nadeem S, Crowley DE (2016) Yeast extract promotes decolorization of azo dyes by stimulating azoreductase activity in Shewanella sp. strain IFN4. Ecotoxicol Environ Saf 124:42–49. https://doi.org/10.1016/j.ecoenv.2015.09.041
Isik M, Sponza DT (2007) Fate and toxicity of azo dye metabolites under batch long-term anaerobic incubations. Enzyme Microb Technol 40:934–939. https://doi.org/10.1016/j.enzmictec.2006.07.032
Jadhav SB, Patil NS, Watharkar AD, Apine OA, Jadhav JP (2013) Batch and continuous biodegradation of amaranth in plain distilled water by P. aeruginosa BCH and toxicological scrutiny using oxidative stress studies. Environ Sci Pollut Res 20:2854–2866. https://doi.org/10.1007/s11356-012-1155-y
Kimura K, Inoue T, Kato DI, Negoro S, Ike M, Takeo M (2013) Distribution of chitin/chitosan-like bioflocculant-producing potential in the genus Citrobacter. Appl Microbiol Biotechnol 97:9569–9577. https://doi.org/10.1007/s00253-012-4668-x
Mandal SM, Sharma S, Pinnaka AK, Kumari A, Korpole S (2013) Isolation and characterization of diverse antimicrobial lipopeptides produced by Citrobacter and Enterobacter. BMC Microbiol. https://doi.org/10.1186/1471-2180-13-152
Meerbergen K, Willems KA, Dewil R, Van Impe J, Appels L, Lievens B (2018) Isolation and screening of bacterial isolates from wastewater treatment plants to decolorize azo dyes. J Biosci Bioeng 125:448–456. https://doi.org/10.1016/j.jbiosc.2017.11.008
Michałowicz J, Duda W (2007) Phenols—sources and toxicity. Pol J Environ Stud 16:347–362
Pandey A, Singh P, Iyengar L (2007) Bacterial decolorization and degradation of azo dyes. Int Biodeterior Biodegrad 59:73–84. https://doi.org/10.1016/j.ibiod.2006.08.006
Pandiyarajan T, Saravanan R, Karthikeyan B et al (2017) Sonochemical synthesis of CuO nanostructures and their morphology dependent optical and visible light driven photocatalytic properties. J Mater Sci: Mater Electron 28:2448–2457. https://doi.org/10.1007/s10854-016-5817-2
Patel DK, Archana G, Kumar GN (2008) Variation in the nature of organic acid secretion and mineral phosphate solubilization by Citrobacter sp. DHRSS in the presence of different sugars. Curr Microbiol 56:168–174. https://doi.org/10.1007/s00284-007-9053-0
Popli S, Patel UD (2015) Destruction of azo dyes by anaerobic–aerobic sequential biological treatment: a review. Int J Environ Sci Technol 12:405–420. https://doi.org/10.1007/s13762-014-0499-x
Rawat D, Mishra V, Sharma RS (2016) Detoxification of azo dyes in the context of environmental processes. Chemosphere 155:591–605. https://doi.org/10.1016/j.chemosphere.2016.04.068
Rawat D, Sharma RS, Karmakar S, Arora LS, Mishra V (2018) Ecotoxic potential of a presumably non-toxic azo dye. Ecotoxicol Environ Saf 148:528–537. https://doi.org/10.1016/j.ecoenv.2017.10.049
Romero-Sáez M, Jaramillo LY, Saravanan R et al (2017) Notable photocatalytic activity of TiO2-polyethylene nanocomposites for visible light degradation of organic pollutants. Express Polym Lett 11:899–909. https://doi.org/10.3144/expresspolymlett.2017.86
Sambrook J, Russel DW (2001) Molecular cloning: a laboratory manual. Ed. Cold Spring Harbor Laboratory Press, New York
Samonis G, Karageorgopoulos DE, Kofteridis DP, Matthaiou DK, Sidiropoulou V, Maraki S, Falagas ME (2009) Citrobacter infections in a general hospital: characteristics and outcomes. Eur J Clin Microbiol Infect Dis 28:61–68. https://doi.org/10.1007/s10096-008-0598-z
Saratale RG, Saratale GD, Chang JS, Govindwar SP (2011) Bacterial decolorization and degradation of azo dyes: a review. J Taiwan Inst Chem Eng 42:138–157. https://doi.org/10.1016/j.jtice.2010.06.006
Saravanan R, Agarwal S, Gupta VK et al (2018a) Line defect Ce3+ induced Ag/CeO2/ZnO nanostructure for visible-light photocatalytic activity. J Photochem Photobiol A Chem 353:499–506. https://doi.org/10.1016/j.jphotochem.2017.12.011
Saravanan R, Aviles J, Gracia F et al (2018b) Crystallinity and lowering band gap induced visible light photocatalytic activity of TiO2/CS (chitosan) nanocomposites. Int J Biol Macromol 109:1239–1245. https://doi.org/10.1016/j.ijbiomac.2017.11.125
Sen SK, Raut S, Bandyopadhyay P, Raut S (2016) Fungal decolouration and degradation of azo dyes: a review. Fungal Biol Rev 30:112–133. https://doi.org/10.1016/j.fbr.2016.06.003
Singh RL, Singh PK, Singh RP (2015) Enzymatic decolorization and degradation of azo dyes—a review. Int Biodeterior Biodegrad 104:21–31. https://doi.org/10.1016/j.ibiod.2015.04.027
Song L, Shao Y, Ning S, Tan L (2017) Performance of a newly isolated salt-tolerant yeast strain Pichia occidentalis G1 for degrading and detoxifying azo dyes. Bioresour Technol 233:21–29. https://doi.org/10.1016/j.biortech.2017.02.065
Stolz A (2001) Basic and applied aspects in the microbial degradation of azo dyes. Appl Microbiol Biotechnol 56:69–80. https://doi.org/10.1007/s002530100686
Tan L, He M, Song L, Fu X, Shi S (2016) Aerobic decolorization, degradation and detoxification of azo dyes by a newly isolated salt-tolerant yeast Scheffersomyces spartinae TLHS-SF1. Bioresour Technol 203:287–294. https://doi.org/10.1016/j.biortech.2015.12.058
Tang BP, Wang YQ, Zhang DZ (2009) Studies on the interaction between benzidine and hemocyanin from Chinese mitten crab Eriocheir japonica sinensis (Decapoda, Grapsidae). Spectrochim Acta Part A Mol Biomol Spectrosc 73:676–681. https://doi.org/10.1016/j.saa.2009.03.019
Telke AA, Joshi SM, Jadhav SU, Tamboli DP, Govindwar SP (2010) Decolorization and detoxification of congo red and textile industry effluent by an isolated bacterium Pseudomonas sp. SU-EBT. Biodegradation 21:283–296. https://doi.org/10.1007/s10532-009-9300-0
Van Der Zee FP, Villaverde S (2005) Combined anaerobic-aerobic treatment of azo dyes—a short review of bioreactor studies. Water Res 39:1425–1440. https://doi.org/10.1016/j.watres.2005.03.007
Wang H, Su JQ, Zheng XW, Tian Y, **ong XJ, Zheng TL (2009) Bacterial decolorization and degradation of the reactive dye Reactive Red 180 by Citrobacter sp. CK3. Int Biodeterior Biodegrad 63:395–399. https://doi.org/10.1016/j.ibiod.2008.11.006
Xu H, Heinze TM, Chen S, Cerniglia CE, Chen H (2007) Anaerobic metabolism of 1-amino-2-naphthol-based azo dyes (Sudan dyes) by human intestinal microflora. Appl Environ Microbiol 73:7759–7762. https://doi.org/10.1128/AEM.01410-07
Zamani H, Grakoee SR, Rakhshaee R (2016) Microbial degradation of Paclitaxel using Citrobacter amalonaticus Rashtia isolated from pharmaceutical wastewater: kinetic and thermodynamic study. World J Microbiol Biotechnol 32:1–10. https://doi.org/10.1007/s11274-016-2087-3
Acknowledgements
The authors would like to thank the University of Taquari Valley—UNIVATES, cost center number 10901314, for the financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Additional information
Editorial responsibility: Necip Atar.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Schmidt, C., Berghahn, E., Ilha, V. et al. Biodegradation potential of Citrobacter cultures for the removal of amaranth and congo red azo dyes. Int. J. Environ. Sci. Technol. 16, 6863–6872 (2019). https://doi.org/10.1007/s13762-019-02274-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-019-02274-x