Abstract
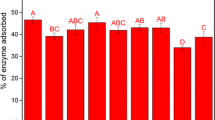
The lignocellulosic biomass, such as provided by the sugarcane, is an abundant source of raw materials for energy production. Milling and pretreatments can be employed to alter the structure of the materials, remove lignin, and hemicellulose. This pretreatment effect exposes the cellulose and raises its accessibility, which is one of the most important properties to ensure enzymatic digestibility. However, the biomass generated from the sugarcane has different physicochemical characteristics, giving different responses to the pretreatments. In this context, this study aimed to verify the effects of lignin and hemicellulose removal from the sugarcane biomass (external fraction, node, internode, and leaf) on cellulose accessibility. Each fraction was pretreated with acid (5, 10, and 20% w/w, at 121 °C/30 min), alkali (5, 10, 20, and 30% NaOH w/w) and oxidative (0.5, 1, 2, and 3 h charged with 30% sodium chlorite). Accessibility was determined by dye adsorption of Direct Orange (external specific surface) and Direct Blue (internal specific surface). Enzymatic hydrolysis was used to verify the effects of pretreatments and cellulose accessibility on the glucose yield. Delignification by sodium chlorite (oxidative) resulted in lignin removal, with almost complete removal from leaf samples. Accessibility determined indicated that pretreatments that are more aggressive improved cellulose accessibility. The less recalcitrant fraction, the internode, showed 1333.3 mg/g of Direct Orange adsorbed and 746.3 mg/g of Direct Blue. Glucose yield during enzymatic hydrolysis improved with higher cellulose accessibility. Lignin and xylan removal (down to 10% and 1%, respectively) resulted in higher glucose yield, with delignified internode samples showing almost complete cellulose conversion. Hemicellulose and lignin removal by the pretreatments directly influenced cellulose accessibility, resulting in better enzymatic hydrolysis across all fractions. This study successfully showed that lignin and hemicellulose removal of 15% and 10%, respectively, resulting in at least 60% of glucose yield, reaching desired accessibility levels based on dye adsorption of 2079.6 mg/g.

Similar content being viewed by others
References
Fernandes JL, Ebecken NFF, Esquerdo JCDM (2017) Sugarcane yield prediction in Brazil using NDVI time series and neural networks ensemble. Int J Remote Sens 38(16):4631–4644
Companhia Nacional De Abastecimento (2019) Acompanhamento da safra brasileira de cana-de-açúcar 2018/2019
Melati RB, Shimizu FL, Oliveira G, Pagnocca FC, De Souza W, Sant’anna C, Brienzo M (2019) Key factors affecting the recalcitrance and conversion process of biomass. BioEnergy Res 12(1):1–20
Zhao X, Zhang L, Liu D (2012) Biomass recalcitrance. Part I: the chemical compositions and physical structures affecting the enzymatic hydrolysis of lignocellulose. Biofuels Bioprod Biorefin 6(4):465–482
Bozell JJ, Holladay JE, Johnson D, White JF (2007) Top value added chemicals from biomass. Volume II–Results of screening for potential candidates from biorefinery lignin. Pacific Northwest National Laboratory, Richland, pp 3552–3599
Miedes E, Vanholme R, Boerjan W, Molina A (2014) The role of the secondary cell wall in plant resistance to pathogens. Front Plant Sci 5:358
Freudenberg K, Chen CL, Harkin JM, Nimz H, Renner H (1965) Observation on lignin. Chem Commun (London) 11:224–225
Naderi A (2017) Nanofibrillated cellulose: properties reinvestigated. Cellulose 24(5):1933–1945
Brienzo M, Tyhoda L, Benjamin Y, Görgens J (2015) Relationship between physicochemical properties and enzymatic hydrolysis of sugarcane bagasse varieties for bioethanol production. New Biotechnol 32(2):253–262
Shimizu FL, Monteiro PQ, Ghiraldi PHC, Melati RB, Pagnocca FC, De Souza W, Brienzo M (2018) Acid, alkali and peroxide pretreatments increase the cellulose accessibility and glucose yield of banana pseudostem. Ind Crop Prod 115:62–68
Brienzo M, Ferreira S, Vicentim MP, De Souza W, Sant’anna C (2014) Comparison study on the biomass recalcitrance of different tissue fractions of sugarcane culm. BioEnergy Res 7(4):1454–1465
Siqueira G, Milagres AM, Carvalho W, Koch G, Ferraz A (2011) Topochemical distribution of lignin and hydroxycinnamic acids in sugar-cane cell walls and its correlation with the enzymatic hydrolysis of polysaccharides. Biotechnol Biofuels 4(1):1
Brienzo M, Abud Y, Ferreira S, Corrales RC, Ferreira-Leitão VS, De Souza W, Sant’anna C (2016) Characterization of anatomy, lignin distribution, and response to pretreatments of sugarcane culm node and internode. Ind Crop Prod 84:305–313
Zheng Y, Zhao J, Xu F, Li Y (2014) Pretreatment of lignocellulosic biomass for enhanced biogas production. Prog Energy Combust Sci 42:35–53
Fl S (1950) A stain for use in the microscopy of beaten fibers. TAPPI J 33:312–314
Chandra RP, Esteghlalian AR, Saddler JN (2008) Assessing substrate accessibility to enzymatic hydrolysis by cellulases. Biofuels 60–80
Ghose TK (1987) Measurement of cellulase activities. Pure Appl Chem 59(2):257–268
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31(3):426–428
Sluiter JB, Ruiz RO, Scarlata CJ, Sluiter AD, Templeton DW (2010) Compositional analysis of lignocellulosic feedstocks. 1. Review and description of methods. J Agric Food Chem 58(16):9043–9053
Liu Z, Wang L, Zhang Y, Li Y, Li Z, Cai H (2017) Cellulose-lignin and xylan-lignin interactions and the formation of lignin-derived phenols in pyrolysis oil. BioResources 12(3):4958–4971
Santos CC, De Souza W, Sant’anna C, Brienzo M (2018) Elephant grass leaves have lower recalcitrance to acid pretreatment than stems, with higher potential for ethanol production. Ind Crop Prod 111:193–200
Brienzo M, Fikizolo S, Benjamin Y, Tyhoda L, Görgens J (2017) Influence of pretreatment severity on structural changes, lignin content and enzymatic hydrolysis of sugarcane bagasse samples. Renew Energy 104:271–280
Rocha GJ, Nascimento VM, Goncalves AR, Silva VFN, And Martín C (2015) Influence of mixed sugarcane bagasse samples evaluated by elemental and physical–chemical composition. Ind Crop Prod 64:52–58
Aguilar R, Ram_irez JA, Garrote G, Vazquez M (2002) Kinetic study of the acid hydrolysis of sugar cane bagasse. J Food Eng 55(4):309–318
Maryana R, Ma’rifatun D, Wheni AI, Satriyo KW, Rizal WA (2014) Alkaline pretreatment on sugarcane bagasse for bioethanol production. Energy Procedia 47:250–254
Siqueira G, Várnai A, Ferraz A, Milagres AM (2013) Enhancement of cellulose hydrolysis in sugarcane bagasse by the selective removal of lignin with sodium chlorite. Appl Energy 102:399–402
Yu X, Minor JL, Atalla RH (1995) Mechanism of action of Simons' stain. TAPPI J 78(6):175–180
Siqueira G, Arantes V, Saddler JN, Ferraz A, Milagres AM (2017) Limitation of cellulose accessibility and unproductive binding of cellulases by pretreated sugarcane bagasse lignin. Biotechnol Biofuels 10(1):176
Alam A, Zhang R, Liu P, Huang J, Wang Y, Hu Z, Tu Y (2019) A finalized determinant for complete lignocellulose enzymatic saccharification potential to maximize bioethanol production in bioenergy Miscanthus. Biotechnol Biofuels 12(1):99
Funding
This study was supported by the Brazilian Council for Research and Development (CNPq, process 401900/2016–9).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shimizu, F.L., de Azevedo, G.O., Coelho, L.F. et al. Minimum Lignin and Xylan Removal to Improve Cellulose Accessibility. Bioenerg. Res. 13, 775–785 (2020). https://doi.org/10.1007/s12155-020-10120-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12155-020-10120-z