Abstract
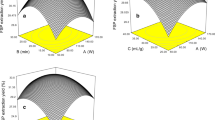
Plantago ovata seed is comprises two distinct layers: a non-adherent outer layer that can easily extracted by gentle agitation, and an adherent inner layer, which is extremely difficult to separate from the seed coat. This study aims to develop and optimize an efficient method for extracting adherent and non-adherent mucilage from P. ovata seeds and to evaluate its biological activities. The extraction process variables were optimized using the response surface methodology (RSM) based on Box-behnken Design with three factors at three levels, to test different experimental conditions and achieve the highest yield. Our results demonstrate that ultrasound treatment was an efficient method for the rapid extraction of both mucilage layers. The aqueous treatment involved a seed-to-water ratio of 1:60, an extraction time of 1.4 h with constant stirring at 45 °C and pH 8, followed by sonication at 100% amplitude for 60 min with pulses of 10 s ON and 20 s OFF for non-adherent mucilage extraction. For the adherent mucilage, the extraction time was adjusted to 1 h with constant stirring at 45 °C and a seed-to-water ratio of 1:50. This process facilitated the recovery of nearly all P. ovata seed mucilage, estimated at approximately 40 mg/g of seeds (39.85% ~ 40%). Evaluation of antioxidant activity in vitro showed that aqueous extracts of P.ovata mucilage had antiradical activity (IC50 = 202 µg/ml), ferric reducing power (IC50 = 310 µg/ml), important total antioxidant activity (16.84 mg EAG/g MS), inhibitory effect on alpha amylase (IC50 = 2.13 mg/ml), and exhibits strong antiproliferative effects on Caco2 cells (IC50 = 90 µg/ml).



Similar content being viewed by others
References
R. Pandita, X. **ao, W. Yang, W. Enck, T. **e, Towards automating risk assessment of mobile applications. In 22nd Security SympoWashington, D.C. (2013), pp. 527–542
R. Ashwini, M. Madgulkar, R.P. Rao, D. Warrier, Polysaccharides (2014). https://doi.org/10.1007/978-3-319-03751-6_49-1
K.M. Huerta, J.S. Alves, A.F.C. Silva, E.H. Kubota, C.S. Rosa, Food Sci. Technol. (2016). https://doi.org/10.1590/1678-457X.0032
L. Yu, G.E. Yakubov, W. Zeng, X. **ng, J. Stenson, V. Bulone, J.R. Stokes, Carbohydr. Polym. (2017). https://doi.org/10.1016/j.carbpol.2017.02.038
F. Chemat, M.A. Vian, G. Cravotto, Int. J. Mol. Sci. (2012). https://doi.org/10.3390/ijms13078615
Q. Guo, S.W. Cui, Q. Wang, J.C. Young, Carbohydr. Polym. (2008). https://doi.org/10.1016/j.carbpol.2007.11.001
Y. Yu, M. Shen, Q. Song, J. **e, Carbohydr. Polym. (2018). https://doi.org/10.1016/j.carbpol.2017.12.009
N. Addoun, Z. Boual, C. Delattre, A.V. Ursu, J. Desbrières, D. Le Cerf, G. Pierre, Int. J. Biol. Macromol. (2020). https://doi.org/10.1016/j.ijbiomac.2019.11.106
J.C. Gasparetto, C.A.F. Martins, S.S. Hayashi, J. Pharm. Phamacol. (2012). https://doi.org/10.1111/j.2042-7158.2011.01383.x
A.E. Al-Snafi, Int. J. Pharm. Tech. Res. 5, 1378–1385 (2013)
M.K. Patel, A. Mishra, B. Jha, Front. Plant. Sci. (2016). https://doi.org/10.3389/fpls.2016.00431
M.T. Jabbar, A. Yasin, A. Khurshid, Q.H. Rashid, Q. Muhammad, S.N. Nazif, A. Imrana, S.A.I. ElKinza, Bokhari, Am. J. P Sci. (2020). https://doi.org/10.5281/zenodo.3749294
A.B. Samuelsen, J. Ethnopharmacol, (2000). https://doi.org/10.1016/s0378-8741(00)00212-9
P. Talukder, S. Talapatra, N. Ghoshal, S.S. Raychaudhuri, J. Sci. Food Agric. (2015). https://doi.org/10.1002/jsfa.7086
Avicenna, Canon of Medicine. (New Delhi: S. Waris Awab, 1998) Lamia Hamdard Printing Press
A. Ameri, G. Heydarirad, J.M. Jafari, A. Ghobadi, H. Rezaeizadeh, R. Choopani, Pharm. Biol. (2015). https://doi.org/10.3109/13880209.2014.928330
J. Huang, D. Bowles, E. Esfandiari, G. Dean, N.C. Carpita, G.W. Haughn, Plant. Physiol. (2011). https://doi.org/10.1104/pp.111.172023
C. Voiniciuc, B. Yang, M. Schmidt, M. Günl, B. Usadel, Int. J. Mol. Sci. (2015). https://doi.org/10.3390/ijms16023452
N. Castejon, P. Luna, F.J. Señoráns, J. Agric. Food Chem. (2017). https://doi.org/10.1021/acs.jafc.6b05726
X. Zhao, L. Qiao, A.M. Wu, Sci. Rep. (2017). https://doi.org/10.1038/srep40672
B.K. Tiwari, K. Muthukumarappan, C.P. O’Donnell, P.J. Cullen, Int. J. Food Prop. (2010). https://doi.org/10.1080/10942910802317610
K. Vilkhu, R. Mawson, L. Simons, D. Bates, Innov. Food Sci. Emerg. Technol. (2008). https://doi.org/10.1016/j.ifset.2007.04.014
M. Ashokkumar, R. Rink, S. Shestakov, Electron. J. Tech. Acoust. (2011). https://www.ejta.org, 9
A.C. Soria, M. Villamiel, Trends Food Sci. Technol. (2010). https://doi.org/10.1016/j.tifs.2010.04.003
F. Chemat, N. Rombaut, A.G. Sicaire, A. Meullemiestre, A.S. Fabiano-Tixier, Abert-Vian, a review. Ultrason. Sonochem. (2017). https://doi.org/10.1016/j.ultsonch.2016.06.035
A. Koocheki, S.A. Mortazavi, F. Shahidi, S.M.A. Razavi, A.R. Taherian, J. Food Eng. (2009). https://doi.org/10.1016/j.jfoodeng.2008.09.028
T. Kaewmanee, L. Bagnasco, S. Benjakul, S. Lanteri, C.F. Morelli, G. Speranza, M.E. Cosulich, Food Chem. (2014). https://doi.org/10.1016/j.foodchem.2013.10.022
G.S. Souza, R.C. Bergamasco, A.P. Stafussa, G.S. Madrona, Emir J. Food Agric. (2020). https://doi.org/10.9755/ejfa.2020.v32.i4.2089
R. Myers, D.C. Montgomery, Response Surface Methodology: Product and Process Optimization Using Designed Experiments, 2nd edn. (John Wiley & Sons, New York, 2002), p. 824
C.J. Patel, J. Bhattacharya, A.J. Butte, PloS One. (2010). https://doi.org/10.1002/jps.2600540727
S.C. Ferreira, R.E. Bruns, H.S. Ferreira, G.D. Matos, J.M. David, G.C. Brandão, W.N.L. Dos, Santos, Anal. Chim. Acta. (2007). https://doi.org/10.1016/j.aca.2007.07.011
S.K. Ozdemir, J. Zhu, L. He, L. Yang, Phys. Rev. A (2011). https://doi.org/10.1103/PhysRevA.83.033817
E. Alpizar-Reyes, H. Carrillo-Navas, R. Gallardo-Rivera, V. Varela-Guerrero, J. Alvarez-Ramirez, C. Pérez-Alons, J. Food Eng. (2017). https://doi.org/10.1016/j.jfoodeng.2017.04.021
M. Dubois, K.A. Gilles, J.K. Hamilton, P.A. Rebers, F. Smith, Anal. Chem. (1956). https://doi.org/10.1021/ac60111a017
P. Prieto, M. Pineda, M. Aguilar, Anal. Biochem. (1999). https://doi.org/10.1006/abio.1999.4019
G. Tel, M. Apaydın, M.E. Duru, Food Anal. Methods. (2012). https://doi.org/10.1007/s12161-011-9275-4
S.P. Wong, P.L. Lai, H.W.K. Jen, Food Chem. 99, 775–783 (2006)
P. Sudha, S.S. Zinjarde, S.Y. Bhargava, A.R. Kumar, BMC Complement. Altern. Med. (2011). https://doi.org/10.1186/1472-6882-11-5
T.L. Western, D.J. Skinner, G.W. Haughn, Plant Physiol. (2000). https://doi.org/10.1104/pp.122.2.345
M.I. Capitani, V.Y. Ixtaina, S.M. Nolasco, M.C. Tomás, J. Sci. Food Agric. (2013). https://doi.org/10.1002/jsfa.6327
S.C. Orifici, M.I. Capitani, M.C. Tomás, S.M. Nolasco, J. Sci. Food Agric. (2018). https://doi.org/10.1002/jsfa.8974
M.H.F. Felisberto, A.L. Wahanik, C.R. Gomes-Ruffi, M.T.P.S. Clerici, Y.K. Chang, C.J. Steel, LWT-Food Sci. Technol. (2015). https://doi.org/10.1016/j.lwt.2015.03.114
D.L. Woods, R.K. Downey, Mucilage from yellow mustard. Can. J. Plant. Sci. 60, 10311033 (1980). https://doi.org/10.4141/cjps80-146
W. Cui, M.N.A. Eskin, C.G. Biliaderis, Food Chem. (1993). https://doi.org/10.1016/0308-8146(93)90032-B
J. Zubr, Nutr. Food Sci. (2010). https://doi.org/10.1108/00346651011077036
J.M. Cowley, L. Herliana, K.A. Neumann, Plant Methods. (2020). https://doi.org/10.1186/s13007-020-00569-6
C. Soukoulis, C. Gaiani, L. Hoffmann, Curr. Opin. Food Sci. (2018). https://doi.org/10.1016/j.cofs.2018.01.004
F.J. Sánchez, M.A. Manzanares, E.F. De andres, J.L. Tenorio, L. Ayerbe, Field Crops Res. (1998). https://doi.org/10.1016/S0378-4290(98)00125-7
E. Bukhsh, S.A. Malik, S.S. Pak. J. Bot. 39, 1181 (2007)
Z.F. Ahmed, A.M. Rizk, F.M. Hammouda, J. Pharm. Sci. (1965). https://doi.org/10.1002/jps.2600540727
E. Souri, G. Amin, H. Farsam, M.T. Barazandeh, DARU. 16, 83–87 (2008)
N. Beara, M.M. Lesjak, E.D. Jovin, K.J. Balog, G.T. Anaˇckov, D.Z. Orˇci´c, N.M. Mimica-Duki´c, J. Agricult Food Chem. (2009). https://doi.org/10.1021/jf902205m
Al-Achi, Women’s Health in Primary Care. 8(7), 325 (2005)
S. Foster, Acta Hort,. (1993). https://doi.org/10.17660/ActaHortic.1993.330.6
A.S.J. Garcia, M.J. Verde-Star, N.L. Heredia, J. Herbs Spices Med. Plants. (2001). https://doi.org/10.1300/J044v08n02_02
J.K. Grover, S. Yadav, V. Vats, J. Ethnopharmacol. (2002). https://doi.org/10.1016/s0378-8741(02)00059-4
S.R. Mentreddy, J. Sci, Food Agric. (2007). https://doi.org/10.1002/jsfa.2811
R. Sultana, A.M. Alashi, K. Islam, M. Saifullah, C.E. Haque, R.E. Aluko, Foods, (2020). https://doi.org/10.3390/foods9070844
B.A. Kumar, K. Lakshman, R. Nandeesh, P.A. Kumar, B. Manoj, V. Kumar, D.S. Shekar, Saudi J. boil sci. (2011). https://doi.org/10.1016/j.sjbs.2010.08.002
S.B. Shah, L. Sartaj, F. Ali, MOJ. Bioequiv. Availab. (2018). https://doi.org/10.15406/mojbb.2018.05.00113
I. Funke, M. Melzig, Rev. bras. Pharmacogn. (2006). https://doi.org/10.1590/S0102-695X2006000100002
M. Sangeethapriya, P. Siddhuraju, Food Sci. Hum. Wellness. (2014). https://doi.org/10.1016/j.fshw.2014.05.003
C.I. Chukwuma, M.S. Islam, E.O. Amonsou, J. Food Biochem. (2018). https://doi.org/10.1111/jfbc.12601
F. Nazir, Q. Fariduddin, A. Hussain, T.A. Khan, Ecotoxicol. Environ. Saf. (2021). https://doi.org/10.1016/j.ecoenv.2020.111081
J.L. Hartwell, Plants used against cancer: A Survey (Quarterman Publications, Lawrence, 1982)
M. Juarranz, M.E. Calle-Puron, A. Gonzalez-Navarro, E. Regidor-Poyatos, T. Soriano, D. Martinez-Hernandez, V.D. Rojas, V.F. Guinee, Eur. J. Cancer Prev. (2002). https://doi.org/10.1097/00008469-200210000-00009
M. Sierra, J. Garcia, N. Fernández, Eur. J. Clin. Nutr. (2001). https://doi.org/10.1038/sj.ejcn.1601147
B. Singh, N. Chauhan, S. Kumar, Carbohydr. Polym. (2008). https://doi.org/10.1016/j.carbpol.2007.12.009
M.K. Patel, B. Tanna, A. Mishra, B. Jha, Int. J. Biol. Macromol. (2018). https://doi.org/10.1016/j.ijbiomac.2018.06.139
A. Wahid, S.M.N. Mahmoud, E.Z. Attia, A.E.S.A. Yousef, A.M.M. Okasha, H.A. Soliman, S Afr. J. Bot. (2020). https://doi.org/10.1016/j.sajb.2020.01.007
M.K. Patel, B. Tanna, H. Gupta, A. Mishra, B. Jha, Int. J. Biol. Macromol. (2019). https://doi.org/10.1016/j.ijbiomac.2019.04.062
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Hassen Gouja, Abdelkarim Ben Arfa, H?dia Hannachi, Hajer Tlili, Hanen Najjaa and Mohamed Neffati. The first draft of the manuscript was written by Hassen Gouja and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The author states that there is no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gouja, H., Arfa, A.B., Hajer, T. et al. Optimization of the rapid effective extraction, antioxidant, antiproliferative and alpha-amylase activities in Plantago ovata seed non-adherent and adherent mucilage by RSM. Food Measure 18, 2450–2460 (2024). https://doi.org/10.1007/s11694-024-02363-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11694-024-02363-7