Abstract
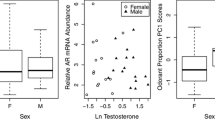
Chemical signaling has been well studied in invertebrates and mammals but less so in birds, due to the longstanding misconception that olfaction is unimportant or even non-existent in this taxon. However, recent findings suggest that olfaction plays an important role in avian mate choice and reproductive behavior, similar to other taxa. The leading candidate source for compounds involved in avian chemical communication is preen oil, a complex mixture secreted from the uropygial gland. Preen oil contains volatile compounds and their potential wax ester precursors, and may act as a reproductive chemosignal. Reproductive signals are generally sexually dimorphic, age-specific, seasonally variable, and may also vary geographically. We tested whether preen oil meets these expectations by using gas chromatography to examine the wax ester composition of preen oil in song sparrows (Melospiza melodia). We found that the wax ester composition of preen oil was significantly different between sexes, age classes, seasons, and populations. Collectively, our results suggest that song sparrow preen oil meets the criteria of a chemical cue that may influence mate choice and reproduction. Our findings in song sparrows, which are sexually monomorphic in plumage, also parallel patterns described for dark-eyed juncos (Junco hyemalis), a closely related songbird with sexually dimorphic plumage. Behavioral tests are needed to confirm that song sparrows attend to the cues present in preen oil, but our findings support the increasingly accepted idea that chemical communication is common and widespread in birds as it is in other taxa.





Similar content being viewed by others
References
Amo L, Avilés JM, Parejo D, Peña A, Rodríguez J, Tomás G (2012a) Sex recognition by odour and variation in the uropygial gland secretion in starlings. J Anim Ecol 81:605–613. https://doi.org/10.1111/j.1365-2656.2011.01940.x
Amo L, López-Rull I, Pagán I, Garcia CM (2012b) Male quality and conspecific scent preferences in the house finch, Carpodacus mexicanus. Anim Behav 84:1483–1489
Andersson MB (1994) Sexual selection. Princeton University Press, Princeton.
Andersson M, Simmons LW (2006) Sexual selection and mate choice. Trends Ecol Evol 21:296–302
Azzani L, Rasmussen JL, Gieseg SP, Briskie JV (2016) An experimental test of the effect of diet on preen wax composition in New Zealand silvereyes (Zosterops lateralis). In: Schulte BA, Goodwin TE, Ferkin MH (eds) Chemical Signals in Vertebrates 12. Springer, New York, pp 511–525
Balthazart J, Schoffeniels E (1979) Pheromones are involved in the control of sexual behaviour in birds. Naturwissenschaften 66:55–56
Balthazart J, Taziaux M (2009) The underestimated role of olfaction in avian reproduction? Behav Brain Res 200:248–259. https://doi.org/10.1016/j.bbr.2008.08.036
Bonadonna F, Nevitt GA (2004) Partner-specific odor recognition in an Antarctic seabird. Science 306:835–835. https://doi.org/10.1126/science.1103001
Campagna S, Mardon J, Celerier A, Bonadonna F (2012) Potential semiochemical molecules from birds: a practical and comprehensive compilation of the last 20 years studies. Chem Senses 37:3–25
Caro SP, Balthazart J (2010) Pheromones in birds: myth or reality? J Comp Physiol A 196:751–766. https://doi.org/10.1007/s00359-010-0534-4
Caro SP, Balthazart J, Bonadonna F (2015) The perfume of reproduction in birds: chemosignaling in avian social life. Horm Behav 68:25–42
Caspers BA, Krause ET (2013) Intraspecific olfactory communication in zebra finches (Taeniopygia guttata): potential information apart from visual and acoustic cues. In: East ML, Dehnhard M (eds) Chemical Signals in Vertebrates 12. Springer, New York, pp 341–351
Clarke KR (1999) Nonmetric multivariate analysis in community-level ecotoxicology. Environ Toxicol Chem 18:118–127. https://doi.org/10.1002/etc.5620180205
Dekker MH, Piersma T, Damsté JSS (2000) Molecular analysis of intact preen waxes of Calidris canutus (Aves: Scolopacidae) by gas chromatography/mass spectrometry. Lipids 35:533–541
Dixon P, Palmer MW (2003) VEGAN, a package of R functions for community ecology. J Veg Sci 14:927–930
Fischer I, Haliński LP, Meissner W, et al (2017) Seasonal changes in the preen wax composition of the Herring gull Larus argentatus. Chemoecology 27:127–139
Gabirot M, Raux L, Dell’Ariccia G et al (2016) Chemical labels differ between two closely related shearwater taxa. J Avian Biol 47:540–551
Gill FB (2007) Ornithology, 3rd edn. W. H. Freeman, New York
Grieves LA, Kelly TR, Bernards MA, MacDougall-Shackleton EA (2018) Malarial infection alters wax ester composition of preen oil in songbirds: results of an experimental study. Auk 135:767–776. https://doi.org/10.1642/AUK-17-242.1
Griffiths R, Double MC, Orr K, Dawson RJ (1998) A DNA test to sex most birds. Mol Ecol 7:1071–1075
Hagelin JC, Jones IL (2007) Bird odors and other chemical substances: a defense mechanism or overlooked mode of intraspecific communication? Auk 124:741–761. https://doi.org/10.1642/0004-8038(2007)124[741:BOAOCS]2.0.CO;2
Hagelin JC, Jones IL, Rasmussen LEL (2003) A tangerine-scented social odour in a monogamous seabird. Proc R Soc Lond B Biol Sci 270:1323–1329
Haribal M, Dhondt AA, Rosane D, Rodriguez E (2005) Chemistry of preen gland secretions of passerines: different pathways to same goal? Why? Chemoecology 15:251–260
Jacob J, Balthazart J, Schoffeniels E (1979) Sex differences in the chemical composition of uropygial gland waxes in domestic ducks. Biochem Syst Ecol 7:149–153
Johansson BG, Jones TM (2007) The role of chemical communication in mate choice. Biol Rev 82:265–289. https://doi.org/10.1111/j.1469-185X.2007.00009.x
Leclaire S, Merkling T, Raynaud C, Giacinti G, Bessière JM, Hatch SA, Danchin É (2011) An individual and a sex odor signature in kittiwakes? Study of the semiochemical composition of preen secretion and preen down feathers. Naturwissenschaften 98:615–624
Leclaire S, Merkling T, Raynaud C, Mulard H, Bessiere JM, Lhuillier E, Hatch SA, Danchin E (2012) Semiochemical compounds of preen secretion reflect genetic make-up in a seabird species. Proc R Soc B Biol Sci 279:1185–1193. https://doi.org/10.1098/rspb.2011.1611
LeMaster MP, Mason RT (2003) Pheromonally mediated sexual isolation among denning populations of red-sided garter snakes, Thamnophis sirtalis parietalis. J Chem Ecol 29:1027–1043
Martín J, López P (2006) Interpopulational differences in chemical composition and chemosensory recognition of femoral gland secretions of male lizards Podarcis hispanica: implications for sexual isolation in a species complex. Chemoecology 16:31–38
Moreno-Rueda G (2017) Preen oil and bird fitness: a critical review of the evidence. Biol Rev 92:2131–2143
R Development Core Team (2017) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Reneerkens J, Piersma T, Damsté JSS (2002) Sandpipers (Scolopacidae) switch from monoester to diester preen waxes during courtship and incubation, but why? Proc R Soc Lond B Biol Sci 269:2135–2139
Reneerkens J, Piersma T, Damsté JSS (2005) Switch to diester preen waxes may reduce avian nest predation by mammalian predators using olfactory cues. J Exp Biol 208:4199–4202. https://doi.org/10.1242/jeb.01872
Salibian A, Montalti D (2009) Physiological and biochemical aspects of the avian uropygial gland. Braz J Biol 69:437–446
Slade JWG, Watson MJ, Kelly TR, Gloor GB, Bernards MA, MacDougall-Shackleton EA (2016) Chemical composition of preen wax reflects major histocompatibility complex similarity in songbirds. Proc R Soc B Biol Sci 283:20161966
Smadja C, Butlin RK (2009) On the scent of speciation: the chemosensory system and its role in premating isolation. Heredity 102(77):77–97
Soini HA, Schrock SE, Bruce KE, Wiesler D, Ketterson ED, Novotny MV (2007) Seasonal variation in volatile compound profiles of preen gland secretions of the dark-eyed junco (Junco hyemalis). J Chem Ecol 33:183–198. https://doi.org/10.1007/s10886-006-9210-0
Soini HA, Whittaker DJ, Wiesler D, Ketterson ED, Novotny MV (2013) Chemosignaling diversity in songbirds: chromatographic profiling of preen oil volatiles in different species. J Chromatogr A 1317:186–192
Stoffel MA, Caspers BA, Forcada J, Giannakara A, Baier M, Eberhart-Phillips L, Müller C, Hoffman JI (2015) Chemical fingerprints encode mother–offspring similarity, colony membership, relatedness, and genetic quality in fur seals. Proc Natl Acad Sci 112:E5005–E5012
Thomas RH, Price ER, Seewagen CL et al (2010) Use of TLC-FID and GC-MS/FID to examine the effects of migratory state, diet and captivity on preen wax composition in white-throated sparrows Zonotrichia albicollis. Ibis 152:782–792
Tuttle EM, Sebastian PJ, Posto AL, Soini HA, Novotny MV, Gonser RA (2014) Variation in preen oil composition pertaining to season, sex, and genotype in the polymorphic white-throated sparrow. J Chem Ecol 40:1025–1038
Whittaker DJ, Soini HA, Atwell JW, Hollars C, Novotny MV, Ketterson ED (2010) Songbird chemosignals: volatile compounds in preen gland secretions vary among individuals, sexes, and populations. Behav Ecol 21:608–614. https://doi.org/10.1093/beheco/arq033
Whittaker DJ, Richmond KM, Miller AK, Kiley R, Bergeon Burns C, Atwell JW, Ketterson ED (2011) Intraspecific preen oil odor preferences in dark-eyed juncos (Junco hyemalis). Behav Ecol 22:1256–1263. https://doi.org/10.1093/beheco/arr122
Zhang J-X, Wei W, Zhang J-H, Yang W-H (2010) Uropygial gland-secreted alkanols contribute to olfactory sex signals in budgerigars. Chem Senses 35:375–382
Zink RM, Dittmann DL (1993) Gene flow, refugia, and evolution of geographic variation in the song sparrow (Melospiza melodia). Evolution 47:717–729
Acknowledgments
We thank the rare Charitable Research Reserve and the Queen’s University Biological Station (QUBS) for access to their land and research facilities. We further acknowledge that the land we used is on the traditional territories of the Anishinabek and Haudenosauneega (Iroquois) peoples, Haldimand Treaty and Simcoe Patent Treaty lands (Cambridge) and on the traditional territories of the Anishinabek, Huron-Wendat, and Mohawk peoples and Crawford Purchase lands (Newboro). We thank Tosha Kelly and Ross Dickson for field assistance and sample collection. This research was supported by an Ontario Graduate Scholarship and a Vanier Scholarship to LAG, and NSERC Discovery Grants to MAB and EAMS.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All birds were captured under permission from the Canadian Wildlife Service (banding subpermits 10691B, E, F). All animal procedures were approved by The University of Western Ontario Animal Use Subcommittee (protocol # 2016–017).
Electronic supplementary material
Table S1
(DOCX 42 kb)
Fig. S1
Complete wax ester composition of breeding stage adult song sparrow preen oil at Newboro (Nfemales = 3, Nmales = 3, mean ± SD). Peaks that were at least 0.1% of the total chromatogram area were retained for analysis, while peaks that were <0.1% were counted as zero (see text for details). (PNG 30 kb)
Fig. S2
Selected wax ester composition of breeding stage adult song sparrow preen oil at Newboro (Nfemales = 3, Nmales = 3, mean ± SD). For complete wax ester composition, see Supplementary Material, Fig. S1. (PNG 43 kb)
Fig. S3
Complete wax ester composition of song sparrow preen oil from post-breeding stage adults and juveniles (sexes pooled for juveniles; sampled at Newboro). Peaks that were at least 0.1% of the total chromatogram area were retained for analysis, while peaks that were <0.1% were counted as zero (see text for details). (PNG 25 kb)
Fig. S4
Complete wax ester composition of breeding-stage song sparrow preen oil at Newboro and Cambridge (NNewboro males = 3, NNewboro females = 3, NCambridge males = 3, NCambridge females = 3, mean ± SD). Peaks that were at least 0.1% of the total chromatogram area were retained for analysis, while peaks that were <0.1% were counted as zero (see text for details). (PNG 31 kb)
Fig. S5
Selected wax ester composition of breeding-stage song sparrow preen oil at Newboro and Cambridge (NNewboro males = 3, NNewboro females = 3, NCambridge males = 3, NCambridge females = 3, mean ± SD). For complete wax ester composition, see Supplementary Material, Fig. S4. (PNG 85 kb)
Fig. S6
Complete wax ester composition of adult song sparrow preen oil sampled during breeding and post-breeding stages at Newboro (Nearly-season females = 3, Nlate-season females = 2, Nearly-season males = 3, Nlate-season males = 3, mean ± SD). Peaks that were at least 0.1% of the total chromatogram area were retained for analysis, while peaks that were <0.1% were counted as zero (see text for details). (PNG 31 kb)
Fig. S7
Selected wax ester composition of adult song sparrow preen oil sampled during breeding and post-breeding stages at Newboro (Nearly-season females = 3, Nlate-season females = 2, Nearly-season males = 3, Nlate-season males = 3, mean ± SD). For complete wax ester composition, see Supplementary Material, Fig. S6. (PNG 101 kb)
Rights and permissions
About this article
Cite this article
Grieves, L.A., Bernards, M.A. & MacDougall-Shackleton, E.A. Wax Ester Composition of Songbird Preen Oil Varies Seasonally and Differs between Sexes, Ages, and Populations. J Chem Ecol 45, 37–45 (2019). https://doi.org/10.1007/s10886-018-1033-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-018-1033-2