Abstract
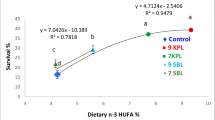
A study was conducted to determine the effects of dietary phospholipid (PL) levels in cobia (Rachycentron canadum) larvae with regard to growth, survival, plasma lipids and enzymes of lipid metabolism. Fish with an average weight of 0.4 g were fed diets containing four levels of PL (0, 20, 40 and 80 g kg−1dry matter: purity 97%) for 42 days. Final body weight (FBW), weight gain (WG) and survival ratio were highest in the 8% PL diet group and mortality was highest in PL-free diet group. We examined the activities of lipoprotein lipase (LPL) and hepatic lipase (HL) in liver, lecithin-cholesterolacyltransferase (LCAT) in plasma as well as plasma lipids and lipoprotein. LCAT activity showed a decrease of more than two-fold in PL-supplemented diet groups compared with the PL-free diet group. HL activity was highest in the 8% PL diet group and the other three groups showed no difference. LPL activity was significantly higher in the PL-supplemented diet groups than in the PL-free diet group. The dietary intervention significantly increased plasma phospholipids and total cholesterol (TC) levels, and the higher free cholesterol (FC) level contributed to the TC level. However, the fish fed PL exhibited a significantly decreased plasma triglyceride (TG) level. The lipoprotein fractions were also affected significantly by the PL. The PL-supplemented diet groups had significantly higher high-density lipoprotein (HDL) compared with the PL-free diet group, but showed a marked decrease in very low-density lipoprotein (VLDL). The results suggested that PL could modify plasma lipoprotein metabolism and lipid profile, and that the optimal dietary PL level may well exceed 80 g kg−1 for cobia larvae according to growth and survival.
Similar content being viewed by others
Abbreviations
- FBW:
-
Final body weight
- FC:
-
Free cholesterol
- HDL:
-
High-density lipoprotein
- HL:
-
Hepatic lipase
- IBW:
-
Initial body weight
- LCAT:
-
Lecithin-cholesterolacyltransferase
- LDL:
-
Low-density lipoprotein
- LPL:
-
Lipoprotein lipase
- PC:
-
Phosphatidylcholine
- PL:
-
Phospholipids
- TC:
-
Total cholesterol
- TG:
-
Triglyceride
- VLDL:
-
Very low-density lipoprotein
- WG:
-
Weight gain
- DHA:
-
22:6n−3
- EPA:
-
20:5n−3
References
Babin PJ, Vernier JM (1989) Plasma lipoprotein in fish. J Lipid Res 30:467–489
Bartlett GR (1959) Phosphorus assay in column chromatography. J Biol Chem 234:466–468
Brown-Peterson NJ, Grier HJ, Overstreet RM (2002) Annual changes in germinal epithelium determine male reproductive classes of the cobia. J Fish Biol 60:178–202
Burnett J, Wilcox LJ, Huff MW (1999) Acyl coenzyme A: cholesterol acyltransferase inhibition and hepatic apolipoprotein B secretion. Clin Chim Acta 286:231–242
Cahu CL, Zambonino Infante JL (2001) Substitution of live food by formulated diets in marine fish larvae. Aquaculture 200:161–180
Cahu CL, Zambonino Infante JL, Quazuguel P, Le Gall MM (1999) Protein hydrolysate vs. fish meal in compound diets for 10-day old sea bass Dicentrarchus labrax larvae. Aquaculture 171:109–119
Cahu CL, Zambonino Infante JL, Barbosa V (2003) Effect of dietary phospholipid level and phospholipid:neutral lipid value on the development of sea bass (Dicentrarchus labrax) larvae fed a compound diet. Br J Nutr 90:21–28
Chou RL, Su MS, Chen HY (2001) Optimal dietary protein and lipid levels for juvenile cobia (Rachycentron canadum). Aquaculture 193:81–89
Chou RL, Her BY, Su MS, Hwang G, Wu YH, Chen HY (2004) Substituting fish meal with soybean meal in diets of juvenile cobia Rachycentron canadum. Aquaculture 229:325–333
Coutteau P, Geurden I, Camara MR, Bergot P, Sorgeloos P (1997) Review on the dietary of phospholipids in fish and crustacean larviculture. Aquaculture 155:149–164
D’Abramo LR, Bordner CE, Conklin DE (1982) Relationship between phosphatidylcholine and serum cholesterol in the lobster Homarus sp. Mar Biol 67:231–235
Dietschy JM, Turley SD, Spady DK (1993) Role of the liver in the maintenance of cholesterol and low density lipoprotein homeostasis in different animal species, including humans. J Lipid Res 34:1637–1659
Eisenberg S, Levy RI (1975) Lipoprotein metabolism. Adv Lipid Res 13:2–80
Fontagné S, Geurden I, Escaffre A-M, Bergot P (1998) Histological changes induced by dietary phospholipids in intestine and liver of common carp (Cyprinus carpio L.) larvae. Aquaculture 161:213–223
Francone OL, Gong EL, Ng DS, Fielding CJ, Rubino EM (1995) Expression of human lecithin-cholesterol acyltransferase in transgenic mice. J Clin Invest 96:1440–1448
Frederickson DS, Ono K, Davis LL (1963) Lipolytic activity of post-heparin plasma in hyperglyceridemia. J Lipid Res 4:24–33
Garling DL, Wilson RP (1976) Optimum dietary protein to energy ratio for channel catfish fingerlings, Ictalurus punctatus. J Nutr 106:1368–1375
Geurden I, Radunz-Neto J, Bergot P (1995) Essentiality of dietary phospholipids for carp (Cyprinus carpio L.) larvae. Aquaculture 131:303–314
Glomset JA (1968) The plasma lecithin-cholesterol acyltransferase reaction. J Lipid Res 9:155–167
Glomset JA, Janssen ET, Kennedy R, Dobbius J (1966) Role of plasma lecithin:cholesterol acyltransferase in the metabolism of high density lipoproteins. J Lipid Res 7:638–648
Glueck CJ, Kaplan AP, Levy RI, Greten H, Gralnick H, Fredrickson DS (1969) A new mechanism of exogenous hyperglyceridemia. Ann Intern Med 71:1051–1062
Hung SS (1989) Choline requirement of hatchery-produced juvenile white sturgeon (Acipenser transmontanus). Aquaculture 78:183–194
Joans A, Swaney SA, Herbert PN (1984) Discoidal complexes of A and C apolipoproteins with lipids and their reactions with lecithin:cholesterol acyltransferase. J Biol Chem 259:6369–6375
Kanazawa A (1993) Essential phospholipids of fish and crustaceans. In: Kaushik SJ, Luquet P (eds) Fish nutrition in practice, Biarritz (France), June 24–27. INRA, Paris, Les Colloques nr. 61:519–530
Kanazawa A, Teshima S, Inamori S, Iwashita T, Nagao A (1981) Effects of phospholipids on growth, survival rate and incidence of malformation in the larval ayu. Mem Fac Fish Kagoshima Univ 30:301–309
Kanazawa A, Teshima S, Sakamoto M (1985) Effects of dietary lipids, fatty acids, and phospholipids on growth and survival of prawn (Penaeus japonicus) larvae. Aquaculture 50:39–49
Levy RI, Thibault L, Garofalo C, Messier M, Lepage G, Ronco N, Roy CC (1990) Combined (n−3 and n−6) essential fatty acid deficiency is a potent modulator of plasma lipids, lipoprotein composition, and lipolytic enzymes. J Lipid Res 31:2009–2017
Marzolo MP, Rigotti A, Nervi F (1990) Secretion of biliary lipids from the hepatocyte. Hepatology 12:134S–142S
Nicoll A, Lewis B (1980) Evaluation of the roles of lipoprotein lipase and hepatic lipase in lipoprotein metabolism: in vivo and in vitro studies in man. Eur J Clin Invest 10:487–495
Poston HA (1991a) Response of rainbow trout to soy lecithin, choline, and autoclaved isolated soy protein. Prog Fish Cult 53:85–90
Poston HA (1991) Response of Atlantic salmon fry to feed-grade lecithin and choline. Prog Fish Cult 53:224–228
Rubistein A, Gibson JC, Paterniti JR, Kakis JG, Little A, Ginsberg HN, Brown WV (1985) Effect of heparin-induced lipolysis on the distribution of apolipoprotein E among lipoprotein subclasses. J Clin Invest 75:710–721
Rumsey GL (1991) Choline-betaine requirements of rainbow trout (Oncorhynchus mykiss). Aquaculture 95:107–116
Salhi M, Hernández-Cruz CM, Bessonart M, Izquierdo MS, Fernández-Palacios H (1999) Effect of different dietary polar lipid levels and different –3 HUFA content in polar lipids on gut and liver histological structure of gilthead seabream (Sparus aurata) larvae. Aquaculture 179:253–263
Santulli A, Modica A, Cutatolo A, D’Amelio V (1993) Lipid and apoprotein composition of lipoprotein of sea bass (Dicentrarchus labrax L.). Proceeding of World Aquaculture 1993, from discovery to commercialization. Torremolinos, Spain. Eur Aquacult Soc (Spec Publ) 19:461
Sargent JR, Henderson RJ, Tocher DR (1989) The lipids. In: Halver J (ed) Fish nutrition, 2nd edn. Academic, London, pp 153–217
Sargent JR, Mceovy LA, Estevez A, Bell JG, Bell MV, Henderson RJ, Tocher DJ (1999) Lipid nutrition of marine fish during early development: current status and future directions. Aquaculture 179:217–229
Sheridan MA (1988) Lipid dynamics in fish: aspects of absorption, transportation, deposition and mobilization. Comp Biochem Physiol 90B:670–690
Suckling KE, Stange EF (1985) Role of acyl-CoA: cholesterol acyltransferase in cellular cholesterol metabolism. J Lipid Res 26:647–671
Teshima S, Kanazawa A, Horinouchi K, Yamasaki S, Hirata H (1987) Phospholipids of the rotifers, prawn and larval fish. Nipon Suisan Gakkaishi 53:609–615
Zheng WH, Liu YJ, Tian LX (2002) A simple method for fatty acid analysis of fish liver. Chin J Health Lab Tech 2:159
Zhou QC, Tan BP, Mai KS, Liu YJ (2004) Apparent digestibility of selected feed ingredients for juvenile cobia Rachycentron canadum. Aquaculture 241:441–451
Acknowledgements
The authors are grateful for the financial support of grant No. 2001DA505D/06 from the National High Technology Research and Development Program (863 Program) during the 10th 5-year plan, China, and to the co-workers for their help in sampling. Special thanks go to teacher C.B. Li for her help in the analysis of the plasma index.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Niu, J., Liu, Y.J., Tian, L.X. et al. Effects of dietary phospholipid level in cobia (Rachycentron canadum) larvae: growth, survival, plasma lipids and enzymes of lipid metabolism. Fish Physiol Biochem 34, 9–17 (2008). https://doi.org/10.1007/s10695-007-9140-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-007-9140-y