Abstract
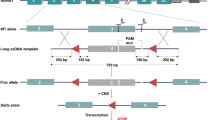
Tiam-1 has been implicated in the development of the central nervous system. However, the in vivo function of Tiam-1 has not been fully determined in the develo** mouse brain. In this study, we generated Tiam-1 knockout mice using a Tiam-1 gene-trapped embryonic stem cell line. Insertion of a gene trap vector into a genomic site downstream of exon 5 resulted in a mutant allele encoding a truncated protein fused with the β-geo LacZ gene. Primary mouse embryonic fibroblasts lacking Tiam-1 revealed a significant decrease in Rac activity and cell proliferation. In addition, whole-mount embryonic LacZ expression analysis demonstrated that Tiam-1 is specifically expressed in regions of the develo** brain, such as the caudal telencephalon and rostral diencephalon. More importantly, mouse embryos deficient in Tiam-1 gene expression displayed a severe defect in embryonic brain development, including neural tube closure defects or a dramatic decrease in brain size. These findings suggest that embryonic Tiam-1 expression plays a critical role during early brain development in mice.
Similar content being viewed by others
References
Engers, R., Springer, E., Michiels, F., Collard, J.G., and Gabbert, H. E. (2001). Rac affects invasion of human renal cell carcinomas by up-regulating tissue inhibitor of metalloproteinases (TIMP)-1 and TIMP-2 expression. J. Biol. Chem. 276, 41889–41897.
Habets, G.G., Scholtes, E.H., Zuydgeest, D., van der Kammen, R.A., Stam, J.C., Berns, A., and Collard, J.G. (1994). Identification of an invasion-inducing gene, Tiam-1, that encodes a protein with homology to GDP-GTP exchangers for Rho-like proteins. Cell 77, 537–549.
Hogan, B. (1994). Manipulating the Mouse Embryo: A Laboratory Manual, 2nd ed. (Plainview, N.Y.: Cold Spring Harbor Laboratory Press).
Iden, S., and Collard, J.G. (2008). Crosstalk between small GTPases and polarity proteins in cell polarization. Nat. Rev. Mol. Cell Biol. 9, 846–859.
Kawauchi, T., Chihama, K., Nabeshima, Y., and Hoshino, M. (2003). The in vivo roles of STEF/Tiam1, Rac1 and JNK in cortical neuronal migration. EMBO J. 22, 4190–4201.
Kim, J., Lee, H., Kim, Y., Yoo, S., Park, E., and Park, S. (2010). The SAM domains of Anks family proteins are critically involved in modulating the degradation of EphA receptors. Mol. Cell. Biol. 30, 1582–1592.
Kunda, P., Paglini, G., Quiroga, S., Kosik, K., and Caceres, A. (2001). Evidence for the involvement of Tiam1 in axon formation. J. Neurosci. 21, 2361–2372.
Malliri, A., van der Kammen, R.A., Clark, K., van der Valk, M., Michiels, F., and Collard, J.G. (2002). Mice deficient in the Rac activator Tiam1 are resistant to Ras-induced skin tumours. Nature 417, 867–871.
Mertens, A.E., Pegtel, D.M., and Collard, J.G. (2006). Tiam1 takes PARt in cell polarity. Trends Cell Biol. 16, 308–316.
Michiels, F., Habets, G.G., Stam, J.C., van der Kammen, R.A., and Collard, J.G. (1995). A role for Rac in Tiam1-induced membrane ruffling and invasion. Nature 375, 338–340.
Minard, M.E., Kim, L.S., Price, J.E., and Gallick, G.E. (2004). The role of the guanine nucleotide exchange factor Tiam1 in cellular migration, invasion, adhesion and tumor progression. Breast Cancer Res. Treat. 84, 21–32.
Miyamoto, Y., Yamauchi, J., Tanoue, A., Wu, C., and Mobley, W.C. (2006). TrkB binds and tyrosine-phosphorylates Tiam1, leading to activation of Rac1 and induction of changes in cellular morphology. Proc. Natl. Acad. Sci. USA 103, 10444–10449.
Nishimura, T., Yamaguchi, T., Kato, K., Yoshizawa, M., Nabeshima, Y., Ohno, S., Hoshino, M., and Kaibuchi, K. (2005). PAR-6-PAR-3 mediates Cdc42-induced Rac activation through the Rac GEFs STEF/Tiam1. Nat. Cell Biol. 7, 270–277.
Shim, S., Kim, Y., Shin, J., Kim, J., and Park, S. (2007). Regulation of EphA8 gene expression by TALE homeobox transcription factors during development of the mesencephalon. Mol. Cell. Biol. 27, 1614–1630.
Tanaka, M., Ohashi, R., Nakamura, R., Shinmura, K., Kamo, T., Sakai, R., and Sugimura, H. (2004). Tiam1 mediates neurite outgrowth induced by ephrin-B1 and EphA2. EMBO J. 23, 1075–1088.
Tolias, K.F., Bikoff, J.B., Burette, A., Paradis, S., Harrar, D., Tavazoie, S., Weinberg, R.J., and Greenberg, M.E. (2005). The Rac1-GEF Tiam1 couples the NMDA receptor to the activity-dependent development of dendritic arbors and spines. Neuron 45, 525–538.
Tolias, K.F., Bikoff, J.B., Kane, C.G., Tolias, C.S., Hu, L., and Greenberg, M.E. (2007). The Rac1 guanine nucleotide exchange factor Tiam1 mediates EphB receptor-dependent dendritic spine development. Proc. Natl. Acad. Sci. USA 104, 7265–7270.
Woodcock, S.A., Rushton, H.J., Castaneda-Saucedo, E., Myant, K., White, G.R., Blyth, K., Sansom, O.J., and Malliri, A. (2010). Tiam1-Rac signaling counteracts Eg5 during bipolar spindle assembly to facilitate chromosome congression. Curr. Biol. 20, 669–675.
Yoo, S., Shin, J., and Park, S. (2010). EphA8-ephrinA5 signaling and clathrin-mediated endocytosis is regulated by Tiam-1, a Rac-specific guanine nucleotide exchange factor. Mol. Cells 29, 603–609.
Zhang, H., and Macara, I.G. (2006). The polarity protein PAR-3 and TIAM1 cooperate in dendritic spine morphogenesis. Nat. Cell Biol. 8, 227–237.
Author information
Authors and Affiliations
Corresponding author
Additional information
These authors contributed equally to this work.
About this article
Cite this article
Yoo, S., Kim, Y., Lee, H. et al. A gene trap knockout of the Tiam-1 protein results in malformation of the early embryonic brain. Mol Cells 34, 103–108 (2012). https://doi.org/10.1007/s10059-012-0119-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10059-012-0119-x