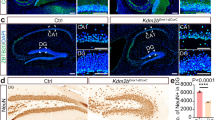
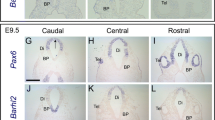
Abstract
The transcription factor KAISO is important for proper development of animal embryos. In the cell, KAISO regulates cell division and apoptosis. KAISO is abundant in the central nervous system. Here we describe the effects of Zbtb33 gene knockout on the transcription of several genes that regulate the development of the central nervous system, including Fgf9, Fgfr3, Sox9, Sox2, c-Myc, NeuroD1 and FoxG1. These genes are related to the Wnt/β-catenin signaling pathway, which is closely connected to KAISO. Hippocampal, frontal cortical, and striatal tissue from C57BL/6j mice with a knockout in the Zbtb33 gene encoding KAISO (ZBTB33-) and wild-type mice (ZBTB33 +) were collected and profiled at different stages of development. Age-dependent and region-specific differences in the mRNA levels of the Fgf9, Fgfr3, c-Myc, FoxG1 genes in the develo** brain of ZBTB33– and ZBTB33+ mice were described and discussed.



Similar content being viewed by others
REFERENCES
Chaudhary R., Pierre C.C., Nanan K., Wojtal D., Morone S., Pinelli C., Wood G.A., Robine S., Daniel J.M. 2013. The POZ-ZF transcription factor Kaiso (ZBTB33) induces inflammation and progenitor cell differentiation in the murine intestine. PLoS One. 8, e74160.
Park J.I., Kim S.W., Lyons J.P., Ji H., Nguyen T.T., Cho K., Barton M.C., Deroo T., Vleminckx K., Moon R.T., McCrea P.D. 2005. Kaiso/p120-catenin and TCF/beta-catenin complexes coordinately regulate canonical Wnt gene targets. Dev. Cell. 8, 843–854.
Ruzov A., Dunican D.S., Prokhortchouk A., Pennings S., Stancheva I., Prokhortchouk E., Meehan R.R. 2004. Kaiso is a genome-wide repressor of transcription that is essential for amphibian development. Development. 131, 6185–6194.
Ruzov A., Savitskaya E., Hackett J.A., Reddington J.P., Prokhortchouk A., Madej M.J., Chekanov N., Li M., Dunican D.S., Prokhortchouk E., Pennings S., Meehan R.R. 2009. The non-methylated DNA-binding function of Kaiso is not required in early Xenopus laevis development. Development. 136, 729–738.
Koh D.I., Yoon J.H., Kim M.K., An H., Kim M.Y., Hur M.W. 2013. Kaiso is a key regulator of spleen germinal center formation by repressing Bcl6 expression in splenocytes. Biochem. Biophys. Res. Commun. 442, 177–182.
Bassey-Archibong B.I., Rayner L.G., Hercules S.M., Aarts C.W., Dvorkin-Gheva A., Bramson J.L., Hassell J.A., Daniel J.M. 2017. Kaiso depletion attenuates the growth and survival of triple negative breast cancer cells. Cell Death Dis. 8, e2689.
Prokhortchouk A., Sansom O., Selfridge J., Caballero I.M., Salozhin S., Aithozhina D., Cerchietti L., Meng F.G., Augenlicht L.H., Mariadason J.M., Hendrich B., Melnick A., Prokhortchouk E., Clarke A., Bird A. 2006. Kaiso-deficient mice show resistance to intestinal cancer. Mol. Cell. Biol. 26, 199–208.
Jones J., Wang H., Zhou J., Hardy S., Turner T., Austin D., He Q., Wells A., Grizzle W.E., Yates C. 2012. Nuclear Kaiso indicates aggressive prostate cancers and promotes migration and invasiveness of prostate cancer cells. Am. J. Pathol. 181, 1836–1846.
Liu Y., Dong Q.Z., Wang S., Xu H.T., Miao Y., Wang L., Wang E.H. 2014. Kaiso interacts with p120-catenin to regulate beta-catenin expression at the transcriptional level. PLoS One. 9, e87537.
van de Ven R.A., Tenhagen M., Meuleman W., van Riel J.J., Schackmann R.C., Derksen P.W. 2015. Nuclear p120-catenin regulates the anoikis resistance of mouse lobular breast cancer cells through Kaiso-dependent Wnt11 expression. Dis. Model Mech. 8, 373–384.
Pierre C.C., Hercules S.M., Yates C., Daniel J.M. 2019. Dancing from bottoms up: Roles of the POZ-ZF transcription factor Kaiso in cancer. Biochim. Biophys. Acta Rev. Cancer. 1871, 64–74.
Buck-Koehntop B.A., Stanfield R.L., Ekiert D.C., Martinez-Yamout M.A., Dyson H.J., Wilson I.A., Wright P.E. 2012. Molecular basis for recognition of methylated and specific DNA sequences by the zinc finger protein Kaiso. Proc. Natl. Acad. Sci. U. S. A. 109, 15229–15234.
Prokhortchouk A., Hendrich B., Jorgensen H., Ruzov A., Wilm M., Georgiev G., Bird A., Prokhortchouk E. 2001. The p120 catenin partner Kaiso is a DNA methylation-dependent transcriptional repressor. Genes Dev. 15, 1613–1618.
Daniel J.M., Spring C.M., Crawford H.C., Reynolds A.B., Baig A. 2002. The p120(ctn)-binding partner Kaiso is a bi-modal DNA-binding protein that recognizes both a sequence-specific consensus and methylated CpG dinucleotides. Nucleic Acids Res. 30, 2911–2919.
Zhigalova N.A., Sokolov A.S., Prokhorchuk E.B., Zhenilo S.V. 2015. S100A3 is a novel target gene of Kaiso in mouse skin. Mol. Biol. (Moscow). 49 (2), 322–325.
Pozner A., Terooatea T.W., Buck-Koehntop B.A. 2016. Cell-specific Kaiso (ZBTB33) regulation of cell cycle through cyclin D1 and cyclin E1. J. Biol. Chem. 291, 24538–24550.
Donaldson N.S., Pierre C.C., Anstey M.I., Robinson S.C., Weerawardane S.M., Daniel J.M. 2012. Kaiso represses the cell cycle gene cyclin D1 via sequence-specific and methyl-CpG-dependent mechanisms. PLoS One. 7, e50398.
Spring C.M., Kelly K.F., O’Kelly I., Graham M., Crawford H.C., Daniel J.M. 2005. The catenin p120ctn inhibits Kaiso-mediated transcriptional repression of the beta-catenin/TCF target gene matrilysin. Exp. Cell Res. 305, 253–265.
Del Valle-Perez B., Casagolda D., Lugilde E., Valls G., Codina M., Dave N., de Herreros A.G., Dunach M. 2011. Wnt controls the transcriptional activity of Kaiso through CK1epsilon-dependent phosphorylation of p120-catenin. J. Cell. Sci. 124, 2298–2309.
Kim S.W., Park J.I., Spring C.M., Sater A.K., Ji H., Otchere A.A., Daniel J.M., McCrea P.D. 2004. Non-canonical Wnt signals are modulated by the Kaiso transcriptional repressor and p120-catenin. Nat. Cell. Biol. 6, 1212–1220.
Liebner S., Corada M., Bangsow T., Babbage J., Taddei A., Czupalla C.J., Reis M., Felici A., Wolburg H., Fruttiger M., Taketo M.M., von Melchner H., Plate K.H., Gerhardt H., Dejana E. 2008. Wnt/beta-catenin signaling controls development of the blood–brain barrier. J. Cell. Biol. 183, 409–417.
Okuda T., Yu L.M., Cingolani L.A., Kemler R., Goda Y. 2007. beta-Catenin regulates excitatory postsynaptic strength at hippocampal synapses. Proc. Natl. Acad. Sci. U. S. A. 104, 13479–13484.
Maguschak K.A., Ressler K.J. 2012. The dynamic role of beta-catenin in synaptic plasticity. Neuropharmacology. 62, 78–88.
Chenn A., Walsh C.A. 2003. Increased neuronal production, enlarged forebrains and cytoarchitectural distortions in beta-catenin overexpressing transgenic mice. Cereb. Cortex. 13, 599–606.
Backman M., Machon O., Mygland L., van den Bout C.J., Zhong W., Taketo M.M., Krauss S. 2005. Effects of canonical Wnt signaling on dorso-ventral specification of the mouse telencephalon. Dev. Biol. 279, 155–168.
Rodova M., Kelly K.F., VanSaun M., Daniel J.M., Werle M.J. 2004. Regulation of the rapsyn promoter by kaiso and delta-catenin. Mol. Cell. Biol. 24, 7188–7196.
Falcone C., Filippis C., Granzotto M., Mallamaci A. 2015. Emx2 expression levels in NSCs modulate astrogenesis rates by regulating EgfR and Fgf9. Glia. 63, 412–422.
Lin Y., Chen L., Lin C., Luo Y., Tsai R.Y., Wang F. 2009. Neuron-derived FGF9 is essential for scaffold formation of Bergmann radial fibers and migration of granule neurons in the cerebellum. Dev. Biol. 329, 44–54.
Hecht D., Zimmerman N., Bedford M., Avivi A., Yayon A. 1995. Identification of fibroblast growth factor 9 (FGF9) as a high affinity, heparin dependent ligand for FGF receptors 3 and 2 but not for FGF receptors 1 and 4. Growth Factors. 12, 223–233.
Sears R.C., Nevins J.R. 2002. Signaling networks that link cell proliferation and cell fate. J. Biol. Chem. 277, 11617–11620.
Boyer L.A., Lee T.I., Cole M.F., Johnstone S.E., Levine S.S., Zucker J.P., Guenther M.G., Kumar R.M., Murray H.L., Jenner R.G., Gifford D.K., Melton D.A., Jaenisch R., Young R.A. 2005. Core transcriptional regulatory circuitry in human embryonic stem cells. Cell. 122, 947–956.
Mercurio S., Serra L., Motta A., Gesuita L., Sanchez-Arrones L., Inverardi F., Foglio B., Barone C., Kaimakis P., Martynoga B., Ottolenghi S., Studer M., Guillemot F., Frassoni C., Bovolenta P., Nicolis S.K. 2019. Sox2 acts in thalamic neurons to control the development of retina-thalamus-cortex connectivity. Science. 15, 257–273.
Mercurio S., Serra L., Nicolis S.K. 2019. More than just stem cells: Functional roles of the transcription factor Sox2 in differentiated glia and neurons. Int. J. Mol. Sci. 20, e4540.
Kang P., Lee H.K., Glasgow S.M., Finley M., Donti T., Gaber Z.B., Graham B.H., Foster A.E., Novitch B.G., Gronostajski R.M., Deneen B. 2012. Sox9 and NFIA coordinate a transcriptional regulatory cascade during the initiation of gliogenesis. Neuron. 74, 79–94.
Cheng L.C., Pastrana E., Tavazoie M., Doetsch F. 2009. miR-124 regulates adult neurogenesis in the subventricular zone stem cell niche. Nat. Neurosci. 12, 399–408.
Ambrozkiewicz M.C., Schwark M., Kishimoto-Suga M., Borisova E., Hori K., Salazar-Lazaro A., Rusanova A., Altas B., Piepkorn L., Bessa P., Schaub T., Zhang X., Rabe T., Ripamonti S., Rosario M., et al. 2018. Polarity acquisition in cortical neurons is driven by synergistic action of Sox9-regulated Wwp1 and Wwp2 E3 ubiquitin ligases and intronic miR-140. Neuron. 100, 1097–1115, e1015.
Cheng C.C., Uchiyama Y., Hiyama A., Gajghate S., Shapiro I.M., Risbud M.V. 2009. PI3K/AKT regulates aggrecan gene expression by modulating Sox9 expression and activity in nucleus pulposus cells of the intervertebral disc. J. Cell. Physiol. 221, 668–676.
Pataskar A., Jung J., Smialowski P., Noack F., Calegari F., Straub T., Tiwari V.K. 2016. NeuroD1 reprograms chromatin and transcription factor landscapes to induce the neuronal program. EMBO J. 35, 24–45.
Chen Y.C., Ma N.X., Pei Z.F., Wu Z., Do-Monte F.H., Keefe S., Yellin E., Chen M.S., Yin J.C., Lee G., Minier-Toribio A., Hu Y., Bai Y.T., Lee K., Quirk G.J., Chen G. 2020. A NeuroD1 AAV-based gene therapy for functional brain repair after ischemic injury through in vivo astrocyte-to-neuron conversion. Mol. Ther. 28, 217–234.
Kumamoto T., Hanashima C. 2017. Evolutionary conservation and conversion of Foxg1 function in brain development. Dev. Growth Differ. 59, 258–269.
Hanashima C., Li S.C., Shen L., Lai E., Fishell G. 2004. Foxg1 suppresses early cortical cell fate. Science. 303, 56–59.
Kumamoto T., Toma K., Gunadi, McKenna W.L., Kasukawa T., Katzman S., Chen B., Hanashima C. 2013. FoxG1 coordinates the switch from nonradially to radially migrating glutamatergic subtypes in the neocortex through spatiotemporal repression. Cell Rep. 3, 931–945.
Reemst K., Noctor S.C., Lucassen P.J., Hol E.M. 2016. The indispensable roles of microglia and astrocytes during brain development. Front. Hum. Neurosci. 10, 566.
Ahlgren S., Vogt P., Bronner-Fraser M. 2003. Excess FoxG1 causes overgrowth of the neural tube. J. Neurobiol. 57, 337–349.
Hatton B.A., Knoepfler P.S., Kenney A.M., Ro-witch D.H., de Alboran I.M., Olson J.M., Eisenman R.N. 2006. N-myc is an essential downstream effector of Shh signaling during both normal and neoplastic cerebellar growth. Cancer Res. 66, 8655–8661.
Wey A., Knoepfler P.S. 2010. C-myc and N-myc promote active stem cell metabolism and cycling as architects of the develo** brain. Oncotarget. 1, 120–130.
Bird C.M., Burgess N. 2008. The hippocampus and memory: Insights from spatial processing. Nat. Rev. Neurosci. 9, 182–194.
Funding
The experimental part of the work was supported by a grant from the Russian Foundation for Basic Research (no. 18-04-00869 A, brain dissection, RNA isolation and real-time PCR) and budget project no. 0259-2021-0015 (housing and breeding animals) and was carried out using equipment belonging to the Shared Center for Genetic Resources of Laboratory Animals, Federal Research Center of ICG SB RAS, supported by the Ministry of Education and Science of Russia (Unique project identifier RFMEFI62119X0023).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests. The authors declare they have no conflict of interest.
Statement on the welfare of animals. The treatment of animals was in accordance with the guidelines for the care and use of laboratory animals of the Russian National Center for Genetic Resources of Laboratory Animals in a specific pathogens free vivarium . All procedures involving animals were in accordance with the FELASA ethical standards.
Rights and permissions
About this article
Cite this article
Illarionova, N.B., Borisova, M.A., Bazhenova, E.Y. et al. Zbtb33 Gene Knockout Changes Transcription of the Fgf9, Fgfr3, c-Myc and FoxG1 Genes in the Develo** Mouse Brain. Mol Biol 55, 363–371 (2021). https://doi.org/10.1134/S0026893321020230
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0026893321020230