Abstract
Purpose
The primary purpose of this study was to assess the biodistribution and radiation dose resulting from administration of 18F-EF5, a lipophilic 2-nitroimidazole hypoxia marker in ten cancer patients. For three of these patients (with glioblastoma) unlabeled EF5 was additionally administered to allow the comparative assessment of 18F-EF5 tumor uptake with EF5 binding, the latter measured in tumor biopsies by fluorescent anti-EF5 monoclonal antibodies.
Methods
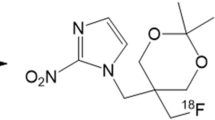
18F-EF5 was synthesized by electrophilic addition of 18F2 gas, made by deuteron bombardment of a neon/fluorine mixture in a high-pressure gas target, to an allyl precursor in trifluoroacetic acid at 0° then purified and administered by intravenous bolus. Three whole-body images were collected for each of ten patients using an Allegro (Philips) scanner. Gamma counts were determined in blood, drawn during each image, and urine, pooled as a single sample. PET images were analyzed to determine radiotracer uptake in several tissues and the resulting radiation dose calculated using OLINDA software and standard phantom. For three patients, 21 mg/kg unlabeled EF5 was administered after the PET scans, and tissue samples obtained the next day at surgery to determine EF5 binding using immunohistochemistry techniques (IHC).
Results
EF5 distributes evenly throughout soft tissue within minutes of injection. Its concentration in blood over the typical time frame of the study (∼3.5 h) was nearly constant, consistent with a previously determined EF5 plasma half-life of ∼13 h. Elimination was primarily via urine and bile. Radiation exposure from labeled EF5 is similar to other 18F-labeled imaging agents (e.g., FDG and FMISO). In a de novo glioblastoma multiforme patient, focal uptake of 18F-EF5 was confirmed by IHC.
Conclusion
These results confirm predictions of biodistribution and safety based on EF5’s characteristics (high biological stability, high lipophilicity). EF5 is a novel hypoxia marker with unique pharmacological characteristics allowing both noninvasive and invasive measurements.





Similar content being viewed by others
Notes
EF5’s partition coefficient was originally determined to be 4 by outside contractors and this value was reported in an initial pharmacology paper [21]. The correct value is 5.7 (at room temperature) and ∼6.25 at 37°.
References
Evans SM, Koch CJ. Prognostic significance of tumor oxygenation in humans. Cancer Lett 2003;195:1–16.
Vaupel P, Mayer A. Hypoxia in cancer: significance and impact on clinical outcome. Cancer Metastasis Rev 2007;26:225–39.
Tatum JL, Kelloff GJ, Gillies RJ, Arbeit JM, Brown JM, Chao KSC, et al. Hypoxia: importance in tumor biology, noninvasive measurement by imaging, and value of its measurement in the management of cancer therapy. Int J Radiat Biol 2006;82:699–757.
Wilson GD. Hypoxia and prognosis: the oxygen tension mounts. Front Biosci 2007;12:3502–18.
Koch CJ. Competition between radiation protectors and radiation sensitizers in mammalian cells. In: Nygaard OF, Simic MG, editors. Radioprotectors and anticarcinogens. New York: Academic; 1983. p. 275–96.
Höckel M, Knoop C, Schlenger K, Vorndran B, Baussmann E, Mitze M, et al. Intratumor pO2 predicts survival in advanced cancer of the uterine cervix. Radiother Oncol 1993;26:45–50.
Brizel DM, Scully SP, Harrelson JM, Layfield LJ, Bean JM, Prosnitz LR, et al. Tissue oxygenation predicts for the likelihood of distant metastases in human soft tissue sarcoma. Cancer Res 1996;56:941–3.
Nordsmark M, Overgaard M, Overgaard J. Pretreatment oxygenation predicts radiation response in advanced squamous cell carcinoma of the head and neck. Radiother Oncol 1996;41:31–9.
Fyles AW, Milosevic M, Wong R, Kavanagh MC, Pintilie M, Sun A, et al. Oxygenation predicts radiation response and survival in patients with cervix cancer. Radiother Oncol 1998;48:149–56.
Dehdashti F, Grigsby PW, Mintun MA, Lewis JS, Siegel BA, Welch MJ. Assessing tumor hypoxia in cervical cancer by positron emission tomography with 60Cu-ATSM: relationship to therapeutic response—a preliminary report. Int J Radiat Oncol Biol Phys 2003;55:1233–8.
Koh W-J, Rasey JS, Evans ML, Grierson JR, Lewellan TK, Graham MM, et al. Imaging of hypoxia in human tumors with [F-18]fluoromisonidazole. Int J Radiat Oncol Biol Phys 1992;22:199–212.
Krohn KA, Link JM, Mason RP. Molecular imaging of hypoxia. J Nucl Med 2008;49:129S–48.
Eschmann S-M, Paulsen F, Reimold M, Dittmann H, Welz S, Reischl G, et al. Prognostic impact of hypoxia imaging with 18F-misonidazole PET in non-small cell lung cancer and head and neck cancer before radiotherapy. J Nucl Med 2005;46:253–60.
Thorwarth D, Eschmann S-M, Scheiderbauer J, Paulsen F, Alber M. Kinetic analysis of dynamic 18F-fluoromisonidazole PET correlates with radiation treatment outcome in head-and-neck cancer. BMC Cancer 2005;5:152.
Rischin D, Hicks RJ, Fisher R, Binns D, Corry J, Porceddu S, et al. Prognostic significance of [18F]-misonidazole positron emission tomography-detected tumor hypoxia in patients with advanced head and neck cancer randomly assigned to chemoradiation with or without tirapazamine: a substudy of Trans-Tasman Radiation Oncology Group Study 98.02. J Clin Oncol 2006;24:2098–104.
Ballinger JR. Imaging hypoxia in tumors. Semin Nucl Med 2001;31:321–9.
Chapman JD, Zanzonico P, Ling CC. On measuring of hypoxia in individual tumors with radiolabeled agents. J Nucl Med 2001;42:1653–5.
Beck R, Röper B, Carlsen JM, Huisman MC, Lebschi JA, Andratschke M, et al. Pretreatment 18F-FAZA PET predicts success of hypoxia-directed radiochemotherapy using tirapazamine. J Nucl Med 2007;48:973–80.
Grosu A-L, Souvatzoglou M, Röper B, Dobritz M, Wiedenmann N, Jacob V, et al. Hypoxia imaging with FAZA-PET and theoretical considerations with regard to dose painting for individualization of radiotherapy in patients with head and neck cancer. Int J Radiat Oncol Biol Phys 2007;69:541–51.
Koch CJ, Shuman AL, Jenkins WT, Kachur AV, Karp JS, Freifelder R, et al. The radiation response of cells from 9L gliosarcoma tumours is correlated with [F18]-EF5 uptake. Int J Radiat Biol 2009;85:1137–47.
Koch CJ, Hahn SM, Rockwell KJ, Covey JM, McKenna WK, Evans SM. Pharmacokinetics of EF5 [2-(2-nitro-1-H-imidazol-1-yl)-N-(2,2,3,3,3-pentafluoropropyl) acetamide] in human patients: implications for hypoxia measurements in vivo. Cancer Chemother Pharmacol 2001;48:177–87.
Coleman CN, Urtasun RC, Wasserman TH, Hancock S, Harris JW, Halsey J, et al. Initial report of the phase I trial of the hypoxic cell radiosensitizer SR-2508. Int J Radiat Oncol Biol Phys 1984;10:1749–53.
Koch CJ. Measurement of absolute oxygen levels in cells and tissues using oxygen sensors and the 2-nitroimidazole EF5. In: Sen CK, Packer L, editors. Methods in enzymology—antioxidants & redox cycling. San Diego: Academic; 2002. p. 3–31.
Workman P. Pharmacokinetics of hypoxic cell radiosensitizers: a review. Cancer Clin Trials 1980;3:237–51.
Coleman CN, Halsey J, Cox RS, Hirst K, Blaschke T, Howes AE, et al. Relationship between the neurotoxicity of the hypoxic cell radiosensitizer SR 2508 and the pharmacokinetic profile. Cancer Res 1987;47:319–22.
Lord EM, Harwell LW, Koch CJ. Detection of hypoxic cells by monoclonal antibody recognizing 2-nitroimidazole adducts. Cancer Res 1993;53:5721–6.
Southwick PL, Ernst LA, Tauriello EW, Parker SR, Mujumdar RB, Mujumdar SR, et al. Cyanine dye labeling reagents—carboxymethylindocyanine succinimidyl esters. Cytometry 1990;11:418–30.
Evans SM, Judy KD, Dunphy I, Jenkins WT, Hwang W-T, Nelson PT, et al. Hypoxia is important in the biology and aggression of human glial brain tumors. Clin Cancer Res 2004;10:8177–84.
Evans SM, Du KL, Chalian AA, Mick R, Zhang PJ, Hahn SM, et al. Patterns and levels of hypoxia in head and neck squamous cell carcinomas and their relationship to patient outcome. Int J Radiat Oncol Biol Phys 2007;69:1024–31.
Evans SM, Jenkins KW, Chen HI, Jenkins WT, Judy KD, Hwang W-T, et al. The relationship between hypoxia, proliferation and outcome in patients with de novo glioblastoma: a pilot study. Transl Oncol 2009; In press.
Dolbier WR, Li A-R, Koch CJ, Shiue C-Y, Kachur AV. [18F]-EF5, a marker for PET detection of hypoxia: synthesis of precursor and a new fluorination procedure. Appl Radiat Isot 2001;54:73–80.
Ziemer LS, Evans SM, Kachur AV, Shuman AL, Cardi CA, Jenkins WT, et al. Noninvasive imaging of tumor hypoxia in rats using the 2-nitroimidazole 18F-EF5. Eur J Nucl Med Mol Imaging 2003;30:259–66.
Allemann K, Wyss MT, Wergin M, Ohlerth S, Rohrer-Bley C, Evans SM, et al. Measurements of hypoxia ([(18)F]-FMISO, [(18)F]-EF5) with positron emission tomography (PET) and perfusion using PET ([(15)O]-H(2)O) and power Doppler ultrasonography in feline fibrosarcomas. Vet Comp Oncol 2005;3:211–21.
Komar G, Seppänen M, Eskola O, Lindholm P, Grönroos JT, Forsback S, et al. 18F-EF5: a new PET tracer for imaging hypoxia in head and neck cancer. J Nucl Med 2008;49:1944–51.
Surti S, Karp JS. Imaging characteristics of a 3-dimensional GSO whole-body PET camera. J Nucl Med 2004;45:1040–9.
Stabin MG, Sparks RB, Crowe E. OLINDA/EXM: the second-generation personal computer software for internal dose assessment in nuclear medicine. J Nucl Med 2005;46:1023–7.
Cloutier RJ, Smith SA, Watson EE, Snyder WS, Warner GG. Dose to the fetus from radionuclides in the bladder. Health Phys 1973;25:147–61.
Evans SM, Hahn SM, Magarelli DP, Zhang PJ, Jenkins WT, Fraker DL, et al. Hypoxia in human intraperitoneal and extremity sarcomas. Int J Radiat Oncol Biol Phys 2001;49:587–96.
Evans SM, Judy KD, Dunphy I, Jenkins WT, Nelson PT, Collins R, et al. Comparative measurements of hypoxia in human brain tumors using needle electrodes and EF5 binding. Cancer Res 2004;64:1886–92.
Evans SM, Jenkins KW, Jenkins WT, Dilling T, Judy KD, Schrlau A, et al. Imaging and analytical methods for the evaluation of vasculature and hypoxia in human brain tumors. Radiat Res 2008;170:677–90.
Cascieri JJ, Graham MM, Rasey JS. A modeling approach for quantifying tumor hypoxia with [F-18]fluoromisonidazole PET time-activity data. Med Phys 1995;22:1127–39.
Rajendran JG, Schwartz DL, O’Sullivan J, Peterson LM, Ng P, Scharnhorst J, et al. Tumor hypoxia imaging with [F-18] fluoromisonidazole positron emission tomography in head and neck cancer. Clin Cancer Res 2006;12:5435–41.
Postema EJ, McEwan AJB, Riauka TA, Kumar P, Richmond DA, Abrams DN, et al. Initial results of hypoxia imaging using 1-alpha-D-(5-deoxy-5-[18F]-fluoroarabinofuranosyl)-2-nitroimidazole (18F-FAZA). Eur J Nucl Med Mol Imaging 2009;36:1565–73.
Raleigh JA, Koch CJ. Importance of thiols in the reductive binding of 2-nitroimidazoles to macromolecules. Biochem Pharmacol 1990;40:2457–64.
Koch CJ. Importance of antibody concentration in the assessment of cellular hypoxia by flow cytometry: EF5 and pimonidazole. Radiat Res 2008;169:677–88.
Evans SM, Schrlau A, Chalian AA, Zhang P, Koch CJ. Oxygen levels in normal and previously irradiated human skin as assessed by EF5 binding. J Invest Dermatol 2006;126:2596–606.
Graham MM, Peterson LM, Link JM, Evans ML, Rasey JS, Koh W-J, et al. Fluorine-18-fluoromisonidazole radiation dosimetry in imaging studies. J Nucl Med 1997;38:1631–6.
Koch CJ, Evans SM. Non-invasive PET and SPECT imaging of tissue hypoxia using isotopically labeled 2-nitroimidazoles. Adv Exp Med Biol 2003;510:285–92.
Bergman J, Solin O. Fluorine-18-labeled fluorine gas for synthesis of tracer molecules. Nucl Med Biol 1997;24:677–83.
Mahy P, Geets X, Lonneux M, Levêque P, Christian N, De Bast M, et al. Determination of tumour hypoxia with [18F]EF3 in patients with head and neck tumours: a phase I study to assess the tracer pharmacokinetics, biodistribution and metabolism. Eur J Nucl Med Mol Imaging 2008;35:1282–9.
Oppenheimer JH, Levy ML, Sinha U, el-Kadi H, Apuzzo ML, Luxton G, et al. Radionecrosis secondary to interstitial brachytherapy: correlation of magnetic resonance imaging and histopathology. Neurosurgery 1992;31:336–43.
De Saint-Hubert M, Prinsen K, Mortelmans L, Verbruggen A, Mottaghy FM. Molecular imaging of cell death. Methods 2009;48:178–87.
Qayum N, Muschel RJ, Im JH, Balathasan L, Koch CJ, Patel S, et al. Tumor vascular changes mediated by inhibition of oncogenic signaling. Cancer Res 2009;69:6347–54.
Nickles RJ, Daube ME, Ruth TJ. An 18O2 target for the production of [18F]F2. Int J Appl Radiat Isot 1984;35:117–22.
Acknowledgement
This work was supported by grants from the NIH/NCI RO1-75284, RO1-87645
Conflicts of interest
EF5 and its labeled counterpart (18F-EF5) are patented (CJK and AVK and others not part of this manuscript, co-inventors) with patents owned by several universities. These patents have been licensed to Varian Biosynergy but this work was entirely supported by grants as indicated in the acknowledgement.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Koch, C.J., Scheuermann, J.S., Divgi, C. et al. Biodistribution and dosimetry of 18F-EF5 in cancer patients with preliminary comparison of 18F-EF5 uptake versus EF5 binding in human glioblastoma. Eur J Nucl Med Mol Imaging 37, 2048–2059 (2010). https://doi.org/10.1007/s00259-010-1517-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-010-1517-y