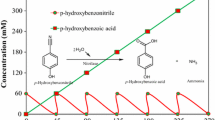
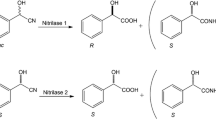
Abstract
Alcaligenes sp. MTCC 10675 has been isolated from soil sample using enrichment method and has nitrilase catalytic system which is highly specific for the hydrolysis of arylaliphatic nitriles. Optimization of culture conditions using response surface methodology and inducer-mediated approach enhanced arylacetonitrilase production significantly (2.4-fold). Isobutyronitrile acted as an effective inducer for the induction of arylacetonitrilase, and it is highly specific for arylacetonitriles (phenyl acetonitrile and mandelonitrile). Arylacetonitrilase has no effect on its relative velocity (V r) up to 20 mM substrate (mandelonitrile) concentration and at 30 mM mandelonitrile, 23.4 % degree of inhibition (I d) was recorded. Half life of arylacetonitrilase of Alcaligenes sp. MTCC 10675 was 27.5 h at 25 °C. Hg2+, Ag+, Pb3+, and Co2+ were strong inhibitor of arylacetonitrilase activity which resulted into 100 %, 91 %, 84 %, and 83 % inhibition, respectively. Polar protic solvent (dichloromethane, dimethylsulphooxide, and n-butanol) reduce arylacetonitrilase activity up to 80–94 % at 10 % concentration. Alcaligenes sp. MTCC 10675 has higher biocatalytic activity, i.e., 3.9 gg-1 dcw, which is highest in comparison to till reported organism. Arylacetonitrilase-mediated hydrolysis of racemic mandelonitrile resulted into R-(-) mandelic acid with 99.0 % enantiomeric excess (e.e.)





Similar content being viewed by others
References
Banerjee A, Dubey S, Kaul P, Barse B, Piotrowski M, Banerjee UC (2009) Enantioselective nitrilase from Pseudomonas putida: cloning, heterologous expression, and bioreactor studies. Mol Biotechnol 41:35–41
Banerjee A, Kaul P, Banerjee UC (2006) Enhancing the catalytic potential of nitrilase from Pseudomonas putida for stereoselective nitrile hydrolysis. Appl Microbiol Biotechnol 72:77–87
Bhalla TC, Miura M, Wakamoto A, Ohba Y, Furuhashi K (1992) Asymmetric hydrolysis of α-aminonitriles to optically active amino acids by a nitrilase of Rhodococcus rhodochrous PA-34. Appl Microbiol Biotechnol 37:184–190
Bhatia SK, Mehta PK, Bhatia RK, Bhalla TC (2013) An isobutyronitrile induced bienzymatic system of Alcaligenes sp. MTCC 10674 for the production of alpha-hydroxyisobutyric acid. Bioprocess Biosyst Eng 36(5):613–625
Fawcett JK, Scott JE (1969) A rapid and precise method for the determination of urea. J Clin Pathol 13:156–159
Fournand D, Arnaud A, Galzy P (1998) Acyl transfer activity of an amidase from Rhodococcus sp. strain R312: formation of a wide range of hydroxamic acids. J Mol Catal B: Enzyme 4:77–90
Gong JS, Lu ZM, Li H, Shi JS, Zhou ZM, Xu ZH (2012) Nitrilases in nitrile biocatalysis: recent progress and forthcoming research. Microb Cell Fact 11:142
He YC, Xu JH, Su JH, Zhou L (2010) Bioproduction of glycolic acid from glycolonitrile with a new bacterial isolate of Alcaligenes sp. ECU0401. Appl Biochem Biotechnol 160:1428–1440
Jain D, Meena VS, Kaushik S, Kamble A, Chisti Y, Banerjee UC (2012) Production of nitrilase by a recombinant Escherichia coli in a laboratory. Fermenta Technol 1(1)
Kaplan O, Vejvoda V, Charvátová-Pinvejcová A, Martínková L (2006) Hyper induction of nitrilases in filamentous fungi. J Ind Microbiol Biotechnol 33:891–896
Kaul P, Banerjee A, Mayilraj S, Banerjee A (2004) Screening for enantioselective nitrilases: kinetic resolution of racemic mandelonitrile to (R)-(-)-mandelic acid by new bacterial isolates. Tetrahedron Asym 15:207–211
Kobayashi M, Yanaka N, Nagasawa T, Yamada H (1991) Hyper induction of an aliphatic nitrilase by Rhodococcus rhodochrous K22. FEMS Microbiol Lett 77:121–124
Nagasawa T, Kobayashi M, Yamada H (1988) Optimum culture conditions for the production of benzonitrilase by Rhodococcus rhodochrous J1. Arch Microbiol 150:89–94
Nagasawa T, Mauger J, Yamada H (2010) A novel nitrilase, arylacetonitrilase, of Alcaligenes faecalis JM3-purification and characterization. Eur J Biochem 194:765–777
Rustler S, Stolz A (2007) Isolation and characterization of a nitrile hydrolyzing acidotolerant black yeast-Exophiala oligosperma R1. Appl Microbiol Biotechnol 75:899–908
Sosedov O, Baum S, Bürger S, Matzer K, Kiziak C, Stolz A (2010) Construction and application of variants of the Pseudomonas fluorescens EBC191 arylacetonitrilase for increased production of acids or amides. Appl Environ Microbiol 76:3668–3674
Vejvoda V, Kaplan O, Bezouska K, Pompach P, Sulc M, Cantarella M, Benada O, Uhnakova B, Rinagelova A, Lutz-Wahl S, Fischer L, Kren V, Martınkova L (2008) Purification and characterization of a nitrilase from Fusarium solani O1. J Mol Catal B Enzym 50:99–106
Watanabe I, Satoh Y, Enomoto K, Seki S, Sakashita K (1987) Optimal conditions for cultivation of Rhodococcus sp. N-774 and for conversion of acrylonitrile to acrylamide by resting cells. Agric Biol Chem 51:3201–3206
Xue YP, Liua ZQ, Xub M, Wanga YJ, Zhenga YG, Shen YC (2010) Enhanced biotransformation of (R, S)-mandelonitrile to (R)-(-)-mandelic acid with in situ production removal by addition of resin. Biochem Eng J 53:143–149
Yamamoto K, Fujimatsu I, Komatsu K (1992) Purification and characterization of the nitrilase from Alcaligenes faecalis ATCC 8750 responsible for enantioselective hydrolysis of mandelonitrile. J Ferment Bioeng 73:425–430
Yamamoto K, Komatsu K (1991) Purification and characterization of nitrilase responsible for the enantioselective hydrolysis from Acinetobacter sp. AK226. Agric Biol Chem 55:1459–1466
Yamazaki Y, Maeda H (1986) (R)-(-) Mandelic acid and other 2-hydroxycarboxylic acids: screening for the enzyme, and its purification, characterization and use. Agric Biol Chem 50:2621–2631
Yang L, Dordil JS, Gade S (2004) Hydration of enzyme in nonaqueous media is consistent with solvent dependence of its activity. Biophys J 87(2):812–821
Zhang Z, Xu JH, He YC, Ouyang LM, Liu YY (2011) Cloning and biochemical properties of a highly thermostable and enantioselective nitrilase from Alcaligenes sp. ECU0401 and its potential for (R)-(-)-mandelic acid production. Bioprocess Biosyst Eng 34:39
Zhu D, Mukherjee C, Yang Y, Rios BE, Gallagher DT, Smith NN, Biehl ER, Hua L (2008) A new nitrilase from Bradyrhizobium japonicum USDA 110 gene cloning, biochemical characterization and substrate specificity. J Biotechnol 133:327–333
Acknowledgments
The authors acknowledge the Department of Biotechnology and University Grant Commission, India for financial support in the form of Senior Research Fellowship to Mr. Shashi Kant Bhatia, Praveen Kumar Mehta, and Ravi Kant Bhatia. The computational facility availed at Bioinformatics Centre, Himachal Pradesh University is also duly acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bhatia, S.K., Mehta, P.K., Bhatia, R.K. et al. Optimization of arylacetonitrilase production from Alcaligenes sp. MTCC 10675 and its application in mandelic acid synthesis. Appl Microbiol Biotechnol 98, 83–94 (2014). https://doi.org/10.1007/s00253-013-5288-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-013-5288-9