Abstract
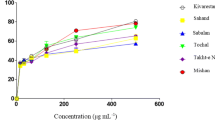
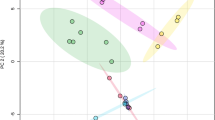
Polyphenolic compounds are electrochemically active components of vegetal matter which were targeted under simple experimental conditions to produce voltammetric profiles characterizing the metabolite composition. Application to bivariate and multivariate chemometric techniques permits to discriminate the species and age of plant leaves, illustrated here for the case of six Pinus species from two different subgenera. Such responses, associated with the electrochemical oxidation of polyphenolic compounds (quercetin, gallic acid, ellagic acid, among others), define a voltammetric profile which varies systematically with the age of the leaves for the different species. The application of this methodology for phylogenetic studies, plant physiology, forensic science, and chemoecology is discussed.

Image of Pinus in a typical Mediterranean forest; Courtesy of the Botanic Garden of the University of Valencia












Similar content being viewed by others
References
Fredrickson H. Applications of methods of chemical analysis in microbial taxonomy. Anal Bioanal Chem. 1992;343:47–8.
Kirk H, Choi YH, Kim HK, Verpoorte R, Van der Meijden E. Comparing metabolomes: the chemical consequences of hybridization in plants. New Phytol. 2005;167:613–22.
Humbert JF, Quiblier C, Gugger M. Molecular approaches for monitoring potentially toxic marine and freshwater phytoplankton species. Anal Bioanal Chem. 2010;397:1723–32.
Dell’Anna R, Lazzeri P, Frisanco M, Monti F, Malvezzi-Campeggi F, Gottardini E, et al. Pollen discrimination and classification by Fourier transform infrared (FT-IR) microspectroscopy and machine learning. Anal Bioanal Chem. 2009;394:1443–52.
Sogawa K, Watanabe M, Sato K, Segawa S, Miyabe A, Murata S, et al. Rapid identification of microorganisms by mass spectrometry: improved performance by incorporation of in-house spectral data into a commercial database. Anal Bioanal Chem. 2012;403:1811–22.
Serra O, Chatterjee S, Huang W, Stark RE. Mini-review: what nuclear magnetic resonance can tell us about protective tissues. Plant Sci. 2012;195:120–4.
Price RA, Liston A, Strauss SH. Evolution, Phylogeny and Systematics of Pinus. In: Richardson DM, editor. Ecology and biogeography of Pinus. Cambridge: Cambridge Univ. Press; 1998. p. 49–68.
Richardson DM, Rundel PW. Ecology and biogeography of Pinus: an introduction. In: Richardson DM, editor. Ecology and Biogeography of Pinus. Cambridge: Cambridge Univ. Press; 1998. p. 3–46.
Farjon A. A natural history of conifers. Portland: Timber Press; 2008.
Li B, Shen YH, He YR, Zhang WD. Chemical constituents and biological activities of Pinus species. Chem Biodiver. 2013;10:2133–60.
Rossbach M, Jayasekera R. Air pollution monitoring at the Environmental Specimen Bank of Germany: spruce and pine shoots as bioindicators. Anal Bioanal Chem. 1996;354:511–4.
Robles C, Greff S, Pasqualini V, Garzino S, Bousquet-Mélou A, Fernandez C, et al. Phenols and flavonoids in Aleppo pine needles as bioindicators of air pollution. J Environ Qual. 2003;32:2265–71.
Warren JM, Bassman JH, Mattinson DS, Fellman JK, Edwards GE, Robberech R. Alteration of foliar flavonoid chemistry induced by enhanced UV-B radiation in field-grown Pinus ponderosa Quercus rubra and Pseudotsuga menziesii. J Photochem Photobiol B: Biol. 2002;66:125–33.
Franich RA, Jakobsson E, Jensen S, Kroese HW, Kylin H. Development of non-destructive methods for the determination of airborne pollutants in pine needles: identification of trace constituents in radiata pine epicuticular wax. Anal Bioanal Chem. 1993;347:337–43.
Hovorka J, Marshall GB. Determination of As, Cd, and Pb in epicuticular waxes of pine and spruce needles by ETAAS. Anal Bioanal Chem. 1997;358:635–40.
Plümacher J, Renner I. Determination of volatile chlorinated hydrocarbons and trichloroacetic acid in conifer needles by headspace gas-chromatography. Anal Bioanal Chem. 1993;347:129–35.
Kylin H, Nordstrand D, Sjödin A, Jensen S. Determination of chlorinated pesticides and PCB in pine needles—improved method for the monitoring of airborne organochlorine pollutants. Anal Bioanal Chem. 1996;356:62–9.
Ratola N, Herbert P, Alves A. Microwave-assisted headspace solid-phase microextraction to quantify polycyclic aromatic hydrocarbons in pine trees. Anal Bioanal Chem. 2012;403:1761–9.
Gernandt DS, López GG, Ortiz-García S, Liston A. Phylogeny and classification of Pinus. Taxon. 2005;54:29–42.
Eckert AJ, Hall BD. Phylogeny, historical biogeography, and patterns of diversification for Pinus (Pinaceae): phylogenetic tests of fossil-based hypotheses. Mol Phylogenet Evol. 2006;40:166–82.
Almaraz-Abarca N, González-Elizondo MS, Tena-Flores JA, Ávila-Reyes JA, Herrera-Corral J, Naranjo-Jiménez N. Foliar flavonoids distinguish Pinus leiophylla and Pinus chihuanuana (Coniferales: Pinaceae). Proc Biol Soc. 2006;119:426–36.
Doménech-Carbó A, Ibars AM, Prieto-Mossi J, Estrelles E, Scholz F, Cebrián-Torrejón G, et al. Electrochemistry-based chemotaxonomy in plants using the voltammetry of microparticles methodology. New J Chem. 2015;39:7421–8.
F. Scholz, B. Meyer, in Electroanalytical Chemistry, A Series of Advances (Eds.: A.J. Bard, I. Rubinstein), Marcel Dekker, New York, 1998, pp. 1–87
Doménech-Carbó A, Labuda J, Scholz F. Electroanalytical chemistry for the analysis of solids: characterization and classification (IUPAC Technical Report). Pure Appl Chem. 2013;8:609–31.
F. Scholz, U. Schröder, R. Gulabowski, A. Doménech-Carbó, Electrochemistry of Immobilized Particles and Droplets, 2nd edit. Springer, Berlin-Heidelberg, 2014
Korotkova EI, Karbainov YA, Avramchik OA. Investigation of antioxidant and catalytic properties of some biologically active substances by voltammetry. Anal Bioanal Chem. 2003;375:465–8.
Brainina KZ, Ivanova AV, Sharafutdinova EN, Lozovskaya EL, Shkarina EI. Potentiometry as a method of antioxidant activity investigation. Talanta. 2007;71:13–8.
Bordonaba JG, Terry LA. Electrochemical behaviour of polyphenol rich fruit juices using disposable screen-printed carbon electrodes: towards a rapid sensor for antioxidant capacity and individual antioxidants. Talanta. 2012;90:38–45.
Vilela D, González MC, Escarpa A. Gold-nanosphere formation using food sample endogenous polyphenols for in-vitro assessment of antioxidant capacity. Anal Bioanal Chem. 2012;404:341–9.
Glod BK, Kiersztyn I, Piszcz P. Total antioxidant potential assay with cyclic voltammetry and/or differential pulse voltammetry measurements. J Electroanal Chem. 2014;719:24–9.
Komorsky-Lovrić Š, Novak I. Estimation of antioxidative properties of tea leaves by abrasive strip** electrochemistry using paraffin-impregnated graphite electrode. Collect Czech Chem Commun. 2009;74:1467–75.
Komorsky-Lovrić Š, Novak I. Abrasive strip** square-wave voltammetry of blackberry, raspberry, strawberry, pomegranate, and sweet and blue potatoes. J Food Sci. 2011;76:C916–20.
Doménech-Carbó A, Domínguez I, Hernández-Muñoz P, Gavara R. Electrochemical tomato (Solanum lycopersicum L.) characterization using contact probe in situ voltammetry. Food Chem. 2015;127:318–25.
Domínguez I, Doménech-Carbó A. Screening and authentication of tea varieties based on microextraction-assisted voltammetry of microparticles. Sens Actuator B. 2015;210:491–9.
Doménech-Carbó A, Gavara R, Hernández-Muñoz P, Domínguez I. Contact probe voltammetry for in situ monitoring of the reactivity of phenolic tomato (Solanum lycopersicum L.) compounds with ROS. Talanta. 2015;144:1207–15.
Doménech-Carbó A, Cebrián-Torrejón G, Lopes-Souto A, Martins de Moraes M, Jorge-Kato M, Fechine-Tavares J, et al. Electrochemical ecology: VIMP monitoring of plant defense against external stressors. RSC Adv. 2015;5:61006–11.
Scholz F, Komorsky-Lovrić Š, Lovrić M. A new access to Gibbs energies of transfer of ions across liquid|liquid interfaces and a new method to study electrochemical processes at well-defined three-phase junctions. Electrochem Commun. 2000;2:112–8.
Scholz F, Gulaboski R. Gibbs energies of transfer of chiral anions across the interface water/chiral organic solvent determined with the help of three-phase electrodes. Faraday Discuss. 2005;6:16–28.
Gunckel S, Santander P, Cordano G, Ferreira J, Muñoz S, Nuñez-Vergara LJ, et al. Antioxidant activity of gallates: an electrochemical study in aqueous media. Chem Biol Interact. 1998;114:45–59.
Janeiro P, Oliveira-Brett AM. Solid state electrochemical oxidation mechanisms of Morin in aqueous media. Electroanalysis. 2005;17:733–8.
Timbola AK, de Souza CD, Giacomelli C, Spinelli A. Electrochemical oxidation of quercetin in hydro-alcoholic solution. J Braz Chem Soc. 2006;17:139–48.
Novak I, Seruga M, Komorsky-Lovric S. Electrochemical characterization of epigallocatechin gallate using square-wave voltammetry. Electroanalysis. 2009;21:1019–25.
Ramesova S, Sokolova R, Tarabek J, Degano I. The oxidation of luteolin, the natural flavonoid dye. Electrochim Acta. 2013;110:646–54.
Lovric M, Scholz F. A model for the propagation of a redox reaction through microcrystals. J Solid State Electrochem. 1997;1:108–13.
Oldham KB. Voltammetry at a three-phase junction. J Solid State Electrochem. 1998;2:367–77.
Lovric M, Scholz F. A model for the coupled transport of ions and electrons in redox conductive microcrystals. J Solid State Electrochem. 1999;3:172–5.
Schröder U, Oldham KB, Myland JC, Mahon PJ, Scholz F. Modelling of solid state voltammetry of immobilized microcrystals assuming an initiation of the electrochemical reaction at a three-phase junction. J Solid State Electrochem. 2000;4:314–24.
Doménech-Carbó A, Doménech-Carbó MT. Chronoamperometric study of proton transfer/electron transfer in solid state electrochemistry of organic dyes. J Solid State Electrochem. 2006;10:949–58.
Doménech-Carbó A, Doménech-Carbó MT. In situ AFM study of proton-assisted electrochemical oxidation/reduction of microparticles of organic dyes. Electrochem Commun. 2008;10:1238–41.
Scampicchio M, Mannino S, Zima J, Wang J. Chemometrics on microchips: towards the classification of wines. Electroanalysis. 2005;17:1215–21.
Klaus W. Mediterranean pines and their history. Plant Syst Evol. 1989;162:133–63.
Wang XR, Tsumura Y, Yoshimaru H, Nagasaka K, Szmidt AD. Phylogenetic Relationships of Eurasian Pines (Pinus, Pinaceae) Based on Chloroplast RBCL, MATK, RPL20-RPS18 Spacer, and TRNV Intron Sequences. Am J Bot. 2000;86:1742–53.
Virtanen V, Korpelainen H, Kostamo K. Forensic botany: usability of bryophyte material in forensic studies. Forensic Sci Int. 2007;172:161–3.
Wiltshire PEJ, Hawksworth DL, Webb JA, Edwards KJ. Palynology and mycology provide separate classes of probative evidence from the same forensic samples: a rape case from southern England. Forensic Sci Int. 2014;244:186–95.
Schnitzler J, Jungblut TP, Feicht C, Köfferlein M, Langebartels C, Heller W, et al. UV-B induction of flavonoid biosynthesis in Scots pine (Pinus sylvestris L.) seedlings. Trees. 1997;11:162–8.
Rousseaux MC, Ballaré CL, Scopel AL, Searles PS, Caldwell WW. Solar UV-B radiation affects plant-insect interactions in a natural ecosystem of Tierra del Fuego (southern Argentina). Oecologia. 1998;116:528–35.
Ratola N, Homem V, Silva JA, Araújo R, Amigo JM, Santos L, et al. Biomonitoring of pesticides by pine needles—chemical scoring, risk of exposure, levels and trends. Sci Total Environ. 2014;476:114–24.
Sensula B, Wilczyński S, Opala M. Tree Growth and climate relationship: dynamics of Scots Pine (Pinus sylvestris L.) growing in the near-source region of the combined heat and power plant during the development of the pro-ecological strategy in Poland. Water, Air, Soil Pollut.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare compliance with ethical standards.
Conflict of interest
No existing conflict of interest related to the procedures, contents, etc. of this article.
Rights and permissions
About this article
Cite this article
Ortiz-Miranda, A.S., König, P., Kahlert, H. et al. Voltammetric analysis of Pinus needles with physiological, phylogenetic, and forensic applications. Anal Bioanal Chem 408, 4943–4952 (2016). https://doi.org/10.1007/s00216-016-9588-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-016-9588-7