Abstract
Purpose
To assess the effect of venovenous extracorporeal membrane oxygenation (ECMO) compared to conventional management in patients with severe acute respiratory distress syndrome (ARDS).
Methods
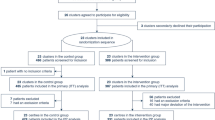
We conducted a systematic review and individual patient data meta-analysis of randomised controlled trials (RCTs) performed after Jan 1, 2000 comparing ECMO to conventional management in patients with severe ARDS. The primary outcome was 90-day mortality. Primary analysis was by intent-to-treat.
Results
We identified two RCTs (CESAR and EOLIA) and combined data from 429 patients. On day 90, 77 of the 214 (36%) ECMO-group and 103 of the 215 (48%) control group patients had died (relative risk (RR), 0.75, 95% confidence interval (CI) 0.6–0.94; P = 0.013; I2 = 0%). In the per-protocol and as-treated analyses the RRs were 0.75 (95% CI 0.6–0.94) and 0.86 (95% CI 0.68–1.09), respectively. Rescue ECMO was used for 36 (17%) of the 215 control patients (35 in EOLIA and 1 in CESAR). The RR of 90-day treatment failure, defined as death for the ECMO-group and death or crossover to ECMO for the control group was 0.65 (95% CI 0.52–0.8; I2 = 0%). Patients randomised to ECMO had more days alive out of the ICU and without respiratory, cardiovascular, renal and neurological failure. The only significant treatment-covariate interaction in subgroups was lower mortality with ECMO in patients with two or less organs failing at randomization.
Conclusions
In this meta-analysis of individual patient data in severe ARDS, 90-day mortality was significantly lowered by ECMO compared with conventional management.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
In this meta-analysis of individual patient data in severe ARDS, 90-day mortality was significantly lowered by ECMO compared with conventional management. Patients randomised to ECMO had more days alive out of the ICU and without respiratory, cardiovascular, renal and neurological failure |
Introduction
Ventilatory management of patients with severe acute respiratory distress syndrome (ARDS) has improved over the last decades with a strategy combining low tidal volume (VT) ventilation [1], high positive end-expiratory pressure (PEEP) [2, 3], neuromuscular blocking agents [4] and prone positioning [5]. However, ventilator-induced lung injury (VILI) may persist in these patients since a recent and large epidemiological study showed that their hospital mortality was still 46% [6]. Recently, even higher mortality was reported for patients with severe acute respiratory syndrome coronavirus2 (SARS-CoV-2) infection who needed invasive mechanical ventilation [7,30]. Our results are consistent with two previous aggregated data meta-analyses in the field: one was a network meta-analysis considering different interventions whose impact is limited by the lack of direct comparisons [31] and the other focused on ECMO [32]. Our IPD meta-analyses goes beyond these two previous studies and provides a stronger evidence on the benefit of ECMO in ARDS for the following reasons. IPD meta-analyses provides a higher level of evidence than aggregated data meta-analyses, because they are independent of the quality of reporting in included studies and allow evaluation of other important outcomes such as time to death and number of days without organ failures [33, 34].
In this study, we showed that, beyond mortality, duration and severity of organ failures also favoured ECMO, and these results were highly consistent between the two studies. This observation provides insights into the potential pathophysiological mechanisms of ECMO-associated benefits in severe ARDS [10]. Although extracorporeal gas exchange may rescue some patients dying of profound hypoxemia or in whom high pressure mechanical ventilation has become dangerous, minimization of lung stress and strain associated with positive pressure ventilation may drive most of the improved outcomes observed under ECMO [10]. Ultraprotective ventilation with very low VTs, driving pressures and respiratory rates [35], and, therefore, minimized overall mechanical power transmitted to lung alveoli [36] may reduce ventilator-induced lung injury, pulmonary and systemic inflammation and ultimately organ failure leading to death. These data also reinforce the recent recommendation of the World Health Organization (WHO) [37], and the Surviving Sepsis Campaign [38] to consider ECMO support in coronavirus disease 2019 (COVID-19)-related ARDS with refractory hypoxemia if lung protective mechanical ventilation was insufficient to support the patient [39].
Meta-analyses of individual patient data can also explore outcomes in important subgroups and suggest which population may derive the greatest benefit of a specific intervention, which is very limited in aggregated data meta-analyses [40]. In this study, the mortality of patients with only one or two organs failing at randomisation was almost halved with ECMO (22% vs. 41%), while it was not substantially different between groups in patients with ≥ 3 organ failures. This finding suggests that veno-venous ECMO may not be able to improve the outcomes of ARDS patients with severe shock and multiple organ failure. In EOLIA, patients with baseline PaO2/FiO2 > 66 mmHg or those enrolled due to severe respiratory acidosis and hypercapnia, seemed to derive the greatest benefit of ECMO [17].
This analysis has several limitations. First, inclusion criteria were more stringent for the EOLIA trial, in which, for example, ventilator optimization (FiO2 > 80%, VT at 6 ml/kg predicted body weight and PEEP > 10 cm H2O) was mandatory before enrolment. However, it should be noted that baseline patient characteristics were comparable regarding ARDS severity at inclusion (eTable 4 in the Supplement). Second patient management was not similar in the two studies. In CESAR, 24% of patients randomised to the ECMO arm did not receive ECMO and there was no standardized protocol for mechanical ventilation in the control group. Conversely, in EOLIA, 98% of patients randomised to ECMO received the intervention, the mechanical ventilation strategy in the control group followed a strict protocol, and rescue ECMO was applied to 28% of control group patients who had developed refractory hypoxemia. However, this meta-analysis showed a significantly lower mortality with ECMO in the per-protocol analysis including only patients in whom ECMO had been initiated in the ECMO arm and patients not having ECMO in the control arm. This analysis minimizes the aforementioned management differences, since the least severe patients who did not receive ECMO after MV optimization in CESAR were excluded from the ECMO arm and the most severe patients who needed rescue ECMO in EOLIA were excluded from the control arm. In contrast, ECMO was not associated with a mortality benefit in the as-treated population, but such an analysis strongly disadvantages the ECMO group, which includes the most severe control patients rescued by ECMO. Second, this meta-analysis does not provide detailed data on ECMO-related safety endpoints, since they were not reported in CESAR. Death directly related to ECMO cannulation was rare in both studies and the rates of stroke and major bleeding were also low in EOLIA, in which a restrictive anticoagulation strategy was applied [17]. Third, no long-term outcomes beyond 90 day post-randomisation were analysed although the CESAR trial [15] and a retrospective cohort of ARDS patients [41] reported satisfactory long-term health-related quality-of-life after ECMO. Fourth, only the CESAR trial provided a cost-effectiveness analysis that suggested a benefit of the transfer of ARDS patients to a centre with an ECMO-based management protocol [15]. Our results, showing improved survival, with more days alive out of the ICU and without the need for major organ support are in line with CESAR’s cost-effectiveness data. Fifth, many conditions such as MV duration > 7 days prior to ECMO or major comorbidities were exclusion criteria for enrolment in both CESAR and EOLIA. The indication to initiate ECMO should, therefore, be carefully evaluated in these situations. Lastly, ECMO should be used in experienced centres and only after proven conventional management of severe ARDS (including lung protective mechanical ventilation and prone positioning) have been applied and failed [42], except when hypoxemia is immediately life-threatening, or when the patient is too unstable for prone positioning [43].
In conclusion, this meta-analysis of individual patient data of the CESAR and EOLIA trials showed strong evidence of a clinically meaningful benefit of early ECMO in severe ARDS patients. Another large study of ECMO appears unlikely in this setting and future research should focus on the identification of patients most likely to benefit from ECMO and optimization of treatment strategies after ECMO initiation [44].
The study was supported by the Direction de la Recherche Clinique et de l’Innovation (DRCI), Assistance Publique—Hopitaux de Paris (APHP), with a grant from the French Ministry of Health (CRC 2018, #18,021).
The EOLIA trial was supported by the Direction de la Recherche Clinique et du Développement (DRCD), Assistance Publique–Hôpitaux de Paris (APHP), with a grant from the French Ministry of Health (Programme Hospitalier de Recherche Clinique number, PHRC 2009 081,224), the EOLIA Trial Group, the Réseau Européen en Ventilation Artificielle (REVA) and the International ECMO Network (ECMONet, https://www.internationalecmonetwork.org).
The CESAR trial was supported by the UK NHS Health Technology Assessment, English National Specialist Commissioning Advisory Group, Scottish Department of Health, and Welsh Department of Health.
See the Supplement for the list of EOLIA and CESAR collaborators.
References
Brower RG, Matthay MA, Morris A, Schoenfeld D, Thompson BT, Wheeler A (2000) Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. N Engl J Med 342:1301–1308
Briel M, Meade M, Mercat A, Brower RG, Talmor D, Walter SD, Slutsky AS, Pullenayegum E, Zhou Q, Cook D, Brochard L, Richard JC, Lamontagne F, Bhatnagar N, Stewart TE, Guyatt G (2010) Higher vs lower positive end-expiratory pressure in patients with acute lung injury and acute respiratory distress syndrome: systematic review and meta-analysis. JAMA 303:865–873
Franchineau G, Brechot N, Lebreton G, Hekimian G, Nieszkowska A, Trouillet JL, Leprince P, Chastre J, Luyt CE, Combes A, Schmidt M (2017) Bedside contribution of electrical impedance tomography to setting positive end-expiratory pressure for extracorporeal membrane oxygenation-treated patients with severe acute respiratory distress syndrome. Am J Respir Crit Care Med 196:447–457
Papazian L, Forel JM, Gacouin A, Penot-Ragon C, Perrin G, Loundou A, Jaber S, Arnal JM, Perez D, Seghboyan JM, Constantin JM, Courant P, Lefrant JY, Guerin C, Prat G, Morange S, Roch A (2010) Neuromuscular blockers in early acute respiratory distress syndrome. N Engl J Med 363:1107–1116
Guerin C, Reignier J, Richard JC, Beuret P, Gacouin A, Boulain T, Mercier E, Badet M, Mercat A, Baudin O, Clavel M, Chatellier D, Jaber S, Rosselli S, Mancebo J, Sirodot M, Hilbert G, Bengler C, Richecoeur J, Gainnier M, Bayle F, Bourdin G, Leray V, Girard R, Baboi L, Ayzac L (2013) Prone positioning in severe acute respiratory distress syndrome. N Engl J Med 368:2159–2168
Bellani G, Laffey JG, Pham T, Fan E, Brochard L, Esteban A, Gattinoni L, van Haren F, Larsson A, McAuley DF, Ranieri M, Rubenfeld G, Thompson BT, Wrigge H, Slutsky AS, Pesenti A (2016) Epidemiology, patterns of care, and mortality for patients with acute respiratory distress syndrome in intensive care units in 50 countries. JAMA 315:788–800
Cummings MJ, Baldwin MR, Abrams D, Jacobson SD, Meyer BJ, Balough EM, Aaron JG, Claassen J, Rabbani LE, Hastie J, Hochman BR, Salazar-Schicchi J, Yip NH, Brodie D, O'Donnell MR, (2020) Epidemiology, clinical course, and outcomes of critically ill adults with COVID-19 in New York City: a prospective cohort study. Lancet 395(10239):1763–1770. https://doi.org/10.1016/S0140-6736(20)31189-2
Yang X, Yu Y, Xu J, Shu H, **a J, Liu H, Wu Y, Zhang L, Yu Z, Fang M, Yu T, Wang Y, Pan S, Zou X, Yuan S, Shang Y (2020) Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. Lancet Respiratory Med 8:475–481
Wang D, Hu B, Hu C, Zhu F, Liu X, Zhang J, Wang B, **ang H, Cheng Z, **ong Y, Zhao Y, Li Y, Wang X, Peng Z (2020) Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA 323:1061–1069
Brodie D, Slutsky AS, Combes A (2019) Extracorporeal life support for adults with respiratory failure and related indications: a review. JAMA 322:557–568
Schmidt M, Pham T, Arcadipane A, Agerstrand C, Ohshimo S, Pellegrino V, Vuylsteke A, Guervilly C, McGuinness S, Pierard S, Breeding J, Stewart C, Ching SSW, Camuso JM, Stephens RS, King B, Herr D, Schultz MJ, Neuville M, Zogheib E, Mira JP, Roze H, Pierrot M, Tobin A, Hodgson C, Chevret S, Brodie D, Combes A (2019) Mechanical ventilation management during extracorporeal membrane oxygenation for acute respiratory distress syndrome. an international multicenter prospective cohort. Am J Respir Crit Care Med 200:1002–1012
Pham T, Combes A, Roze H, Chevret S, Mercat A, Roch A, Mourvillier B, Ara-Somohano C, Bastien O, Zogheib E, Clavel M, Constan A, Marie Richard JC, Brun-Buisson C, Brochard L (2013) Extracorporeal membrane oxygenation for pandemic influenza A(H1N1)-induced acute respiratory distress syndrome: a cohort study and propensity-matched analysis. Am J Respir Crit Care Med 187:276–285
Davies A, Jones D, Bailey M, Beca J, Bellomo R, Blackwell N, Forrest P, Gattas D, Granger E, Herkes R, Jackson A, McGuinness S, Nair P, Pellegrino V, Pettila V, Plunkett B, Pye R, Torzillo P, Webb S, Wilson M, Ziegenfuss M (2009) Extracorporeal membrane oxygenation for 2009 influenza A(H1N1) acute respiratory distress syndrome. JAMA 302:1888–1895
Noah MA, Peek GJ, Finney SJ, Griffiths MJ, Harrison DA, Grieve R, Sadique MZ, Sekhon JS, McAuley DF, Firmin RK, Harvey C, Cordingley JJ, Price S, Vuylsteke A, Jenkins DP, Noble DW, Bloomfield R, Walsh TS, Perkins GD, Menon D, Taylor BL, Rowan KM (2011) Referral to an extracorporeal membrane oxygenation center and mortality among patients with severe 2009 influenza A(H1N1). JAMA 306:1659–1668
Peek GJ, Mugford M, Tiruvoipati R, Wilson A, Allen E, Thalanany MM, Hibbert CL, Truesdale A, Clemens F, Cooper N, Firmin RK, Elbourne D (2009) Efficacy and economic assessment of conventional ventilatory support versus extracorporeal membrane oxygenation for severe adult respiratory failure (CESAR): a multicentre randomised controlled trial. Lancet 374:1351–1363
Peek GJ, Elbourne D, Mugford M, Tiruvoipati R, Wilson A, Allen E, Clemens F, Firmin R, Hardy P, Hibbert C, Jones N, Killer H, Thalanany M, Truesdale A (2010) Randomised controlled trial and parallel economic evaluation of conventional ventilatory support versus extracorporeal membrane oxygenation for severe adult respiratory failure (CESAR). Health Technol Assessment (Winchester, England) 14:1–46
Combes A, Hajage D, Capellier G, Demoule A, Lavoue S, Guervilly C, Da Silva D, Zafrani L, Tirot P, Veber B, Maury E, Levy B, Cohen Y, Richard C, Kalfon P, Bouadma L, Mehdaoui H, Beduneau G, Lebreton G, Brochard L, Ferguson ND, Fan E, Slutsky AS, Brodie D, Mercat A (2018) Extracorporeal membrane oxygenation for severe acute respiratory distress syndrome. N Engl J Med 378:1965–1975
Bernard GR, Artigas A, Brigham KL, Carlet J, Falke K, Hudson L, Lamy M, Legall JR, Morris A, Spragg R (1994) The American-European consensus conference on ARDS. Definitions, mechanisms, relevant outcomes, and clinical trial coordination. Am J Respir Crit Care Med 149:818–824
Ranieri VM, Rubenfeld GD, Thompson BT, Ferguson ND, Caldwell E, Fan E, Camporota L, Slutsky AS (2012) Acute respiratory distress syndrome: the Berlin Definition. JAMA 307:2526–2533
Combes A, Brodie D, Bartlett R, Brochard L, Brower R, Conrad S, De Backer D, Fan E, Ferguson N, Fortenberry J, Fraser J, Gattinoni L, Lynch W, MacLaren G, Mercat A, Mueller T, Ogino M, Peek G, Pellegrino V, Pesenti A, Ranieri M, Slutsky A, Vuylsteke A (2014) Position paper for the organization of extracorporeal membrane oxygenation programs for acute respiratory failure in adult patients. Am J Respir Crit Care Med 190:488–496
Combes A, Pesenti A, Ranieri VM (2017) Fifty years of research in ARDS. Is extracorporeal circulation the future of acute respiratory distress syndrome management? Am J Respir Crit Care Med 195:1161–1170
Sterne JAC, Savovic J, Page MJ, Elbers RG, Blencowe NS, Boutron I, Cates CJ, Cheng HY, Corbett MS, Eldridge SM, Emberson JR, Hernan MA, Hopewell S, Hrobjartsson A, Junqueira DR, Juni P, Kirkham JJ, Lasserson T, Li T, McAleenan A, Reeves BC, Shepperd S, Shrier I, Stewart LA, Tilling K, White IR, Whiting PF, Higgins JPT (2019) RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ 366:l4898
Tierney JF, Stewart LA, Clarke M (2019) Chapter 26: Individual participant data. In: Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA (eds) Cochrane handbook for systematic reviews of interventions version 6–0 (updated July 2019) Cochrane, www.training.cochrane.org/handbook
Li X, Scales DC, Kavanagh BP (2018) Unproven and expensive before proven and cheap: extracorporeal membrane oxygenation versus prone position in acute respiratory distress syndrome. Am J Respir Crit Care Med 197:991–993
Schmidt M, Combes A, Shekar K (2019) ECMO for immunosuppressed patients with acute respiratory distress syndrome: drawing a line in the sand. Intensive Care Med 45:1140–1142
Fernando SM, Qureshi D, Tanuseputro P, Fan E, Munshi L, Rochwerg B, Talarico R, Scales DC, Brodie D, Dhanani S, Guerguerian AM, Shemie SD, Thavorn K, Kyeremanteng K (2019) Mortality and costs following extracorporeal membrane oxygenation in critically ill adults: a population-based cohort study. Intensive Care Med 45:1580–1589
Fan E, Karagiannidis C (2019) Less is more: not (always) simple-the case of extracorporeal devices in critical care. Intensive Care Med 45:1451–1453
Zapol WM, Snider MT, Hill JD, Fallat RJ, Bartlett RH, Edmunds LH, Morris AH, Peirce EC 2nd, Thomas AN, Proctor HJ, Drinker PA, Pratt PC, Bagniewski A, Miller RG Jr (1979) Extracorporeal membrane oxygenation in severe acute respiratory failure. A randomized prospective study JAMA 242:2193–2196
Morris AH, Wallace CJ, Menlove RL, Clemmer TP, Orme JF Jr, Weaver LK, Dean NC, Thomas F, East TD, Pace NL et al (1994) Randomized clinical trial of pressure-controlled inverse ratio ventilation and extracorporeal CO2 removal for adult respiratory distress syndrome. Am J Respir Crit Care Med 149:295–305
Goligher EC, Tomlinson G, Hajage D, Wijeysundera DN, Fan E, Juni P, Brodie D, Slutsky AS, Combes A (2018) Extracorporeal membrane oxygenation for severe acute respiratory distress syndrome and posterior probability of mortality benefit in a post hoc bayesian analysis of a randomized clinical trial. JAMA 320:2251–2259
Aoyama H, Uchida K, Aoyama K, Pechlivanoglou P, Englesakis M, Yamada Y, Fan E (2019) Assessment of therapeutic interventions and lung protective ventilation in patients with moderate to severe acute respiratory distress syndrome: a systematic review and network meta-analysis. JAMA network open 2:e198116
Munshi L, Walkey A, Goligher E, Pham T, Uleryk EM, Fan E (2019) Venovenous extracorporeal membrane oxygenation for acute respiratory distress syndrome: a systematic review and meta-analysis. The Lancet Respiratory medicine 7:163–172
Tierney JF, Vale C, Riley R, Smith CT, Stewart L, Clarke M, Rovers M (2015) Individual participant data (IPD) meta-analyses of randomised controlled trials: guidance on their use. PLoS Med 12:e1001855
Tierney JF, Fisher DJ, Burdett S, Stewart LA, Parmar MKB (2020) Comparison of aggregate and individual participant data approaches to meta-analysis of randomised trials: an observational study. PLoS Med 17:e1003019
Abrams D, Schmidt M, Pham T, Beitler JR, Fan E, Goligher EC, McNamee JJ, Patroniti N, Wilcox ME, Combes A, Ferguson ND, McAuley DF, Pesenti A, Quintel M, Fraser J, Hodgson CL, Hough CL, Mercat A, Mueller T, Pellegrino V, Ranieri VM, Rowan K, Shekar K, Brochard L, Brodie D (2020) Mechanical ventilation for acute respiratory distress syndrome during extracorporeal life support. research and practice. Am J Respir Crit Care Med 201:514–525
Serpa Neto A, Deliberato RO, Johnson AEW, Bos LD, Amorim P, Pereira SM, Cazati DC, Cordioli RL, Correa TD, Pollard TJ, Schettino GPP, Timenetsky KT, Celi LA, Pelosi P, Gama de Abreu M, Schultz MJ (2018) Mechanical power of ventilation is associated with mortality in critically ill patients: an analysis of patients in two observational cohorts. Intensive Care Med 44:1914–1922
World Health Organization: Clinical management of severe acute respiratory infection when COVID-19 disease is suspected. Last accessed July, 10 2020: https://www.who.int/publications/i/item/clinical-management-of-covid-19
Alhazzani W, Møller MH, Arabi YM, Loeb M, Gong MN, Fan E, Oczkowski S, Levy MM, Derde L, Dzierba A, Du B, Aboodi M, Wunsch H, Cecconi M, Koh Y, Chertow DS, Maitland K, Alshamsi F, Belley-Cote E, Greco M, Laundy M, Morgan JS, Kesecioglu J, McGeer A, Mermel L, Mammen MJ, Alexander PE, Arrington A, Centofanti JE, Citerio G, Baw B, Memish ZA, Hammond N, Hayden FG, Evans L, Rhodes A (2020) Surviving sepsis campaign: guidelines on the management of critically ill adults with coronavirus disease 2019 (COVID-19). Intensive Care Med 46:854–887
Schmidt M, Hajage D, Lebreton G, Monsel A, Voiriot G, Levy D, Baron E, Beurton A, Chommeloux J, Meng P, Nemlaghi S, Bay P, Leprince P, Demoule A, Guidet B, Constantin JM, Fartoukh M, Dres M, Combes A (2020) Extracorporeal membrane oxygenation for severe acute respiratory distress syndrome associated with COVID-19: a retrospective cohort study. Lancet Respiratory Med. https://doi.org/10.1016/S2213-2600(20)30328-3
Fisher DJ, Carpenter JR, Morris TP, Freeman SC, Tierney JF (2017) Meta-analytical methods to identify who benefits most from treatments: daft, deluded, or deft approach? BMJ 356:j573
Schmidt M, Zogheib E, Roze H, Repesse X, Lebreton G, Luyt CE, Trouillet JL, Brechot N, Nieszkowska A, Dupont H, Ouattara A, Leprince P, Chastre J, Combes A (2013) The PRESERVE mortality risk score and analysis of long-term outcomes after extracorporeal membrane oxygenation for severe acute respiratory distress syndrome. Intensive Care Med 39:1704–1713
Abrams D, Ferguson ND, Brochard L, Fan E, Mercat A, Combes A, Pellegrino V, Schmidt M, Slutsky AS, Brodie D (2019) ECMO for ARDS: from salvage to standard of care? Lancet Respiratory Med 7:108–110
MacLaren G, Combes A, Brodie D (2020) Saying no until the moment is right: initiating ECMO in the EOLIA era. Intensive Care Med. https://doi.org/10.1007/s00134-020-06185-1
Guervilly C, Prud'homme E, Pauly V, Bourenne J, Hraiech S, Daviet F, Adda M, Coiffard B, Forel JM, Roch A, Persico N, Papazian L (2019) Prone positioning and extracorporeal membrane oxygenation for severe acute respiratory distress syndrome: time for a randomized trial? Intensive Care Med 45:1040–1042
Acknowledgements
We thank Mrs Elizabeth Allen for her help in preparing the data of the CESAR trial.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
Alain Combes reports grants from Getinge, personal fees from Getinge, Baxter and Xenios outside the submitted work. Matthieu Schmidt reports lecture fees from Getinge, Drager and Xenios outside the submitted work. The other authors declare that they have no conflicts of interest related to the purpose of this manuscript. The study protocol for the systematic review and IPD meta-analysis was approved by the relevant independent ethics committees: in France, Comité de Protection des Personnes CPP Ile de France VI, Pitié-Salpêtrière, on 04/19/2018, Ref #12 and in the UK by the Ethics committee of the London School of Hygiene and Tropical Medicine, on 04/12/2019, LSHTM Ethics Ref: 17159. Only patient characteristics and outcomes already evaluated in the trials were combined in this systematic review and meta-analysis.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Combes, A., Peek, G.J., Hajage, D. et al. ECMO for severe ARDS: systematic review and individual patient data meta-analysis. Intensive Care Med 46, 2048–2057 (2020). https://doi.org/10.1007/s00134-020-06248-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-020-06248-3