Abstract
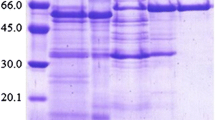
1-O-Sinapoyl-β-glucose:l-malate O-sinapoyltransferase (SMT; EC 2.3.1.) from cotyledons of red radish (Raphanus sativus L. var. sativus) was purified to apparent homogeneity with a 2100-fold enrichment and a 4% recovery. Apparent Mrs of 52 and 51, respectively, were determined by gel filtration and by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE). On isoelectric focusing, the SMT resolved into two isoforms which, on SDS-PAGE, showed, slightly different Mrs (SMT I: Mr/isoelectric point = 51/5.75; SMT II: Mr/isoelectric point = 51.5/5.9). The highest activity of SMT was found at pH 6.0 (50% at pH 5.5 and pH 6.5). The temperature maxima in the presence of 10, 50, 100 and 250 mM malate were 22, 30, 35 and 37° C, respectively, with energies of activation of 55, 81, 96 and 121 kJ · mol-1. The enzyme accepted all the hydroxycinnamic acid-glucose esters tested with relative ratios of initial velocity values of 100∶85∶45∶26∶2.6 of 1-O-sinapoyl-, 1-O-feruloyl-, 1-O-caffeoyl-, 1,2-di-O-sinapoyl-, and 1-O-(4-coumaroyl)-β-glucose. It showed an absolute acceptor specificity for l-malate. d-Malate as second acceptor molecule in standard assays with l-malate inhibited the reaction velocity noncompetitively (K i = 215 mM). The substrate saturation curves were not hyperbolic. The data for sinapoylglucose indicated substrate activation; those for l-malate, substrate inhibition. Kinetic analysis suggests a random bi bi mechanism within two ranges of substrate concentrations, with a kinetically preferred pathway via the enzyme-sinapoylglucose complex indicating a slow-transition mechanism. This may be interpreted as hysteretic cooperativity with sinapoylglucose.
Similar content being viewed by others
Abbreviations
- IEF:
-
isoelectric focusing
- Mal:
-
l-malate
- pI:
-
isoelectric point
- SinGlc:
-
1-O-sinapoyl-β-glucose
- SinMal:
-
O-sinapoyl-l-malate
- SMT:
-
1-O-sinapoyl-β-glucose: l-malate sinapoyltransferase
- SMT I and SMT II:
-
SMT isoforms isolated after isoelectric focusing
References
Ainslie, G.R., Jr., Shill, J.P., Neet, K.E. (1972) Transients and cooperativity.A slow transition model for relating transients and cooperative kinetics of enzymes. J. Biol. Chem. 247, 7088–7096
Bradford, H. (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72, 248–254
Dahlbender, B., Strack, D. (1986) Purification and properties of 1-(hydroxycinnamoyl)-glucose: 1-(hydroxycinnamoyl)-glucose hydroxycinnamoyltransferase from radish seedlings. Phytochemistry 25, 1043–1046
Denzel, K., Gross, G.G. (1991) Biosynthesis of gallotannins. Enzymic “disproportionation” of 1,6-digalloylglucose to 1,2,6-trigalloylglucose and 6-galloylglucose by an acyltransferase from leaves of Rhus typhina L. Planta 184, 285–289
Dixon, M. (1953) The determination of enzyme inhibitor constants. Biochem. J. 55, 170–171
Ferdinand, W. (1966) The interpretation of non-hyperbolic rate curves for two-substrate enzymes.A possible mechanism for phosphofructokinase. Biochem. J. 98, 278–283
Görg, A., Postel, W., Westermeier, R., Righetti, P.G., Ek, K. (1981) One and two dimensional electrophoresis performed horizontally in ultrathin SDS pore-gradient gels. LKB Application Note 320, 1–12
Hanes, C.S. (1932) Studies on plant amylase.I. the effect of starch concentration upon the velocity of hydrolysis by the amylase of germinated barley. Biochem. J. 26, 1406–1421
Hill, A.V. (1910) The combination of hemoglobin with oxygen and with carbon monoxide. J. Physiol. 40, 4–7
Heukeshoven, J., Dernick, R. (1988a) Vereinfachte und universelle Methode zur Silber-Färbung von Proteinen in Polyacrylamidgelen: Bemerkungen zum Mechanismus der Silberfärbung. Pharmacia LKB-Sonderdrucke, RE-034, 92–97
Heukeshoven, J., Dernick, R. (1988b) Improved silver staining procedure for fast staining in PhastSystem development unit. I. Staining of sodium dodecyl sulfate gels. Electrophoresis 9, 28–32
McCoy, R.W., Aiken, R.L., Pauls, R.E., Ziegel, E.R., Wolf, T., Fritz, G.T., Marmion, D.M. (1984) Results of a cooperative study comparing the precision of peak height and area measurements in liquid chromatography. J. Chromatogr. Sci. 22, 425–431
Neville, D.M. Jr., Glossmann, H. (1974) Molecular weight determination of membrane protein and glycoprotein subunits by discontinuous gel electrophoresis in dodecyl sulfate. Methods Enzymol. 32, 92–102
Nurmann, G., Strack, D. (1979) Sinapine esterase. I. Characterization of sinapine esterase from cotyledons of Raphanus sativus. Z. Naturforsch. 34c, 715–720
Regenbrecht, J., Strack, D. (1985) Distribution of 1-sinapoylglucose: choline sinapoyltransferase in the Brassicaceae. Phytochemistry 24, 407–410
Ricard, J., Meunier, J.-C., Buc, J. (1965) Regulatory behavior of monomeric enzymes. 1. The mnemonical enzyme concept. Eur. J. Biochem. 49, 195–208
Sharma, V., Strack, D. (1985) Vacuolar localization of 1-sinapoylglucose: l-malate sinapoyltransferase in protoplasts from cotyledons of Raphanus sativus. Planta 163, 563–568
Strack, D. (1982) Development of 1-O-sinapoyl-β-glucose: l-malate sinapoyltransferase activity in cotyledons of red radish (Raphanus sativus L. var. sativus). Planta 155, 31–36
Strack, D., Mock, H.-P. (1992) Hydroxycinnamic acids and lignins. In: Methods in plant biochemistry, Dey, P.M., Harborne, J.B., eds., vol. 9: Enzymes of secondary metabolism, Lea, P.J., ed., in press. Academic Press, London
Strack, D., Sharma, V. (1985) Vacuolar localization of the enzymatic synthesis of hydroxycinnamic acid esters of malic acid in protoplasts of Raphanus sativus leaves. Physiol. Plant. 65, 45–50
Strack, D., Nurmann, G., Sachs, G. (1980) Sinapine esterase. II. Specificity and change of sinapine esterase activity during germination of Raphanus sativus. Z. Naturforsch. 35c, 963–966
Strack, D., Knogge, W., Dahlbender, B. (1983) Enzymatic synthesis of sinapine from 1-O-sinapoyl-β-d-glucose and choline by a cell-free system from develo** seeds of red radish (Raphanus sativus L. var. sativus). Z. Naturforsch. 38c, 21–27
Strack, D., Sharma, V., Felle, H. (1987) Vacuolar pH in radish cotyledonal mesophyll cells. Planta 172, 563–565
Strack, D., Ellis, B.E., Gräwe, W., Heilemann, J. (1990) Sinapoylglucose: malate sinapoyltransferase activity in seeds and seedlings of rape. Planta 180, 217–219
Tkotz, N., Strack, D. (1980) Enzymatic synthesis of sinapoyl-l-malate from 1-sinapoylglucose and l-malate by a protein preparation from Raphanus sativus cotyledons. Z. Naturforsch. 35c, 835–837
Weber, K., Osborne, M. (1969) The reliability of molecular weight determinations by dodecyl sulfate-polyacrylamide gel electrophoresis. J. Biol. Chem. 244, 4406–4412
Author information
Authors and Affiliations
Additional information
We thank H. Bisswanger (Physiologisch-chemisches Institut, Universität (Tübingen, FRG) for help on the interpretation of substrate kinetic data and B.E. Ellis (Department of Plant Science, The University of British Columbia, Vancouver, B.C., Canada) for linguistic advice. Support by the Deutsche Forschungsgemeinschaft (Bonn, FRG) and the Fonds der Chemischen Industrie (Frankfurt, FRG) is gratefully acknowledged.
Rights and permissions
About this article
Cite this article
Gräwe, W., Bachhuber, P., Mock, HP. et al. Purification and characterization of sinapoylglucose:malate sinapoyltransferase from Raphanus sativus L.. Planta 187, 236–241 (1992). https://doi.org/10.1007/BF00201945
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00201945