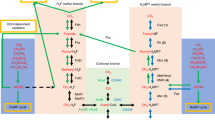
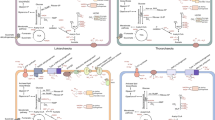
Abstract
When the first genome sequence of a Planctomycete became available (in 2003), it revealed the presence of genes encoding a pathway for a tetrahydromethanopterin-mediated transfer of C1 units between the oxidation levels of formaldehyde and formate, resembling a pathway for methanogenesis being carried out by a specialized group of Archaea and a pathway for formaldehyde oxidation employed by some methylotrophic bacteria, the latter pathway acting in reverse to methanogenesis. This discovery was of importance as the presence of the genes in question in the Planctomycetes has challenged the assumption of a limited distribution of these genes/pathways in the microbial world, at the same time suggesting novel scenarios for the evolution of C1 transfer pathways in microbes and providing support for the potential antiquity of these pathways. In this chapter, I review the early work on the discovery and analysis of the genetic determinants of C1 functions in Planctomycetes and the significance of these discoveries in interpreting the emergence and the evolution of C1 metabolism in Prokaryotes. This is followed by a review of the continuously emerging new genomic data suggesting a much wider distribution of the tetrahydromethanopterin-linked functions in Prokaryotes, further supporting the hypothesis of the long evolution for these functions. While the Planctomycetes provide these important insights into the evolution of specific biochemical pathways as well as the evolution of Prokaryotes in general, the exact function of the tetrahydromethanopterin-linked C1 transfer pathway in Planctomycetes and in many other phyla remains unknown.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
Abbreviations
- H4MPT:
-
Tetrahydromethanopterin
- C1:
-
Single carbon atom as in C1 compound, organic compound containing a single carbon atom
- MtdB:
-
NAD(P)-linked methylene-H4MPT dehydrogenase
- MFR:
-
Methanofuran
- H4MPT:
-
Tetrahydromethanopterin
- F420 :
-
Coenzyme F420
- CoM:
-
Coenzyme M
- CoB:
-
Coenzyme B. Fae formaldehyde-activating enzyme
- MtdB MtdC:
-
Methylene-H4MPT dehydrogenases
- Mch:
-
Methenyl-H4MPT cyclohydrolase
- FhcABCD:
-
Formyltransferase/hydrolase complex
- FdwD:
-
Is homologous to the D subunit of formyl-MFR dehydrogenase
- LUCA:
-
Last universal common ancestor
References
Arrhenius T, Arrhenius G, Paplawsky W (1994) Archean geochemistry of formaldehyde and cyanide and the oligomerization of cyanohydrin. Orig Life Evol Biosph 24:1–17
Bauer M, Lombardot T, Teeling H, Ward NL, Amann RI, Glöckner FO (2004) Archaea-like genes for C1-transfer enzymes in Planctomycetes: phylogenetic implications of their unexpected presence in this phylum. J Mol Evol 59:571–586
Boucher Y, Douady CJ, Papke RT, Walsh DA, Boudreau MER, Nesbø C, Case RJ, Doolitle WF (2003) Lateral gene transfer and the origins of prokaryotic groups. Annu Rev Genet 37:283–328
Brochier C, Philippe H (2002) Phylogeny: a non-hyperthermophilic ancestor for bacteria. Nature 417:244
Brown SD, Begemann MB, Mormile MR, Wall JD, Han CS, Goodwin LA, Pitluck S, Land ML, Hauser LJ, Elias DA (2011) Complete genome sequence of the haloalkaliphilic, hydrogen-producing bacterium Halanaerobium hydrogeniformans. J Bacteriol 193:3682–3683
Cavalier-Smith T (2002) The neomuran origin of archaebacteria, the negibacterial root of the universal tree and bacterial megaclassification. Int J Syst Evol Microbiol 52:7–76
Chistoserdova L (2011) Modularity of methylotrophy, revisited. Environ Microbiol 13:2603–2622
Chistoserdova L, Vorholt JA, Thauer RK, Lidstrom ME (1998) C1 transfer enzymes and coenzymes linking methylotrophic bacteria and methanogenic archaea. Science 281:99–102
Chistoserdova L, Jenkins C, Kalyuzhnaya MG, Marx CJ, Lapidus A, Vorholt JA, Staley JT, Lidstrom ME (2004) The enigmatic planctomycetes may hold a key to the origins of methanogenesis and methylotrophy. Mol Biol Evol 21:1234–1241
Chistoserdova L, Lapidus A, Han C, Goodwin L, Saunders L, Brettin T, Tapia R, Gilna P, Lucas S, Richardson PM, Lidstrom ME (2007) The genome of Methylobacillus flagellatus, the molecular basis for obligate methylotrophy, and the polyphyletic origin of methylotrophy. J Bacteriol 189:4020–4027
Clum A, Tindall BJ, Sikorski J, Ivanova N, Mavrommatis K, Lucas S, Glavina Del Rio T, Nolan M, Chen F, Tice H, Pitluck S, Cheng JF, Chertkov O, Brettin T, Han C, Detter JC, Kuske C, Bruce D, Goodwin L, Ovchinikova G, Pati A, Mikhailova N, Chen A, Palaniappan K, Land M, Hauser L, Chang YJ, Jeffries CD, Chain P, Rohde M, Göker M, Bristow J, Eisen JA, Markowitz V, Hugenholtz P, Kyrpides NC, Klenk HP, Lapidus A (2009) Complete genome sequence of Pirellula staleyi type strain (ATCC 27377). Stand Genomic Sci 1:308–316
DeLong EF (2000) Resolving a methane mystery. Nature 407:577
Di Giulio M (2003) The ancestor of the Bacteria domain was a hyperthermophile. J Theor Biol 224:277–283
Elshahed MS, Youssef NH, Luo Q, Najar FZ, Roe BA, Sisk TM, Bühring SI, Hinrichs KU, Krumholz LR (2007) Phylogenetic and metabolic diversity of Planctomycetes from anaerobic, sulfide- and sulfur-rich Zodletone Spring, Oklahoma. Appl Environ Microbiol 73:4707–4716
Ettwig KF, Butler MK, Le Paslier D, Pelletier E, Mangenot S, Kuypers MM, Schreiber F, Dutilh BE, Zedelius J, de Beer D, Gloerich J, Wessels HJ, van Alen T, Luesken F, Wu ML, van de Pas-Schoonen KT, Op den Camp HJ, Janssen-Megens EM, Francoijs KJ, Stunnenberg H, Weissenbach J, Jetten MS, Strous M (2010) Nitrite-driven anaerobic methane oxidation by oxygenic bacteria. Nature 464:543–548
Fuerst JA, Sagulenko E (2011) Beyond the bacterium: planctomycetes challenge our concepts of microbial structure and function. Nat Rev Microbiol 9:403–413
Glöckner FO, Kube M, Bauer M, Teeling H, Lombardot T, Ludwig W, Gade D, Beck A, Borzym K, Heitmann K, Rabus R, Schlesner H, Amann R, Reinhardt R (2003) Complete genome sequence of the marine planctomycete Pirellula sp. strain 1. Proc Natl Acad Sci U S A 100:8298–8303
Gogarten JP, Doolittle WF, Lawrence JG (2002) Prokaryotic evolution in light of gene transfer. Mol Biol Evol 19:2226–2238
Göker M, Cleland D, Saunders E, Lapidus A, Nolan M, Lucas S, Hammon N, Deshpande S, Cheng JF, Tapia R, Han C, Goodwin L, Pitluck S, Liolios K, Pagani I, Ivanova N, Mavromatis K, Pati A, Chen A, Palaniappan K, Land M, Hauser L, Chang YJ, Jeffries CD, Detter JC, Beck B, Woyke T, Bristow J, Eisen JA, Markowitz V, Hugenholtz P, Kyrpides NC, Klenk HP (2011) Complete genome sequence of Isosphaera pallida type strain (IS1B). Stand Genomic Sci 4:63–71
Hagemeier CH, Chistoserdova L, Lidstrom ME, Thauer RK, Vorholt JA (2000) Characterization of a second methylene tetrahydromethanopterin dehydrogenase from Methylobacterium extorquens AM1. Eur J Biochem 267:3762–3769
Kalyuzhnaya MG, Chistoserdova L (2005) Community-level analysis: genes encoding methanopterin-dependent enzymes. Meth Enzymol 397:443–454
Kalyuzhnaya MG, Korotkova N, Crowther G, Marx CJ, Lidstrom ME, Chistoserdova L (2005) Analysis of gene islands involved in methanopterin-linked C1 transfer reactions reveals new functions and provides evolutionary insights. J Bacteriol 187:4607–4614
Kalyuzhnaya MG, Lapidus A, Ivanova N, McHardy A, Copeland AC, Suciu D, Salamov A, McHardy A, Szeto E, Levine SR, Barry K, Green-Tringe S, Grigoriev I, Markowitz V, Rigoutsos I, Richardson PM, Lidstrom ME, Chistoserdova L (2008) High-resolution metagenomics targets major functional types in complex microbial communities. Nat Biotechnol 26:1029–1034
Kane SR, Chakicherla AY, Chain PS, Schmidt R, Shin MW, Legler TC, Scow KM, Larimer FW, Lucas SM, Richardson PM, Hristova KR (2007) Whole-genome analysis of the methyl tert-butyl ether-degrading beta-proteobacterium Methylibium petroleiphilum PM1. J Bacteriol 189(5):1931–1945
Kasting J, Siefert JL (2002) Life and the evolution of Earth’s atmosphere. Science 296:1066–1068
Knittel K, Boetius A (2009) Anaerobic oxidation of methane: progress with an unknown process. Ann Rev Microbiol 63:31–334
LaButti K, Sikorski J, Schneider S, Nolan M, Lucas S, Glavina Del Rio T, Tice H, Cheng JF, Goodwin L, Pitluck S, Liolios K, Ivanova N, Mavromatis K, Mikhailova N, Pati A, Chen A, Palaniappan K, Land M, Hauser L, Chang YJ, Jeffries CD, Tindall BJ, Rohde M, Göker M, Woyke T, Bristow J, Eisen JA, Markowitz V, Hugenholtz P, Kyrpides NC, Klenk HP, Lapidus A (2010) Complete genome sequence of Planctomyces limnophilus type strain (Mü 290). Stand Genomic Sci 3:47–56
Lösekann TK, Knittel T, Nadalig B, Fuchs H, Niemann AB, Amann R (2007) Diversity and abundance of aerobic and anaerobic methane oxidizers at the Haakon Mosby mud volcano, Barents Sea. Appl Environ Microbiol 73:3348–3362
Martin W, Russell MJ (2003) On the origins of cells: a hypothesis for the evolutionary transitions from abiotic geochemistry to chemoautotrophic prokaryotes, and from prokaryotes to nucleated cells. Phil Trans R Soc Lond 358:59–83
Marx CJ, O'Brien BN, Breezee J, Lidstrom ME (2003) Novel methylotrophy genes of Methylobacterium extorquens AM1 identified by using transposon mutagenesis including a putative dihydromethanopterin reductase. J Bacteriol 185:669–673
Pagani I, Liolios K, Jansson J, Chen IM, Smirnova T, Nosrat B, Markowitz VM, Kyrpides NC (2012) The Genomes OnLine Database (GOLD) v. 4: status of genomic and metagenomic projects and their associated metadata. Nucleic Acids Res 40:D571–D579
Reeve JN, Nölling J, Morgan RM, Smith DR (1997) Methanogenesis: genes, genomes, and who’s on first? J Bacteriol 179:5975–5986
Sauter LM, Latypova E, Smalley NE, Lidstrom ME, Hallam S, Kalyuzhnaya MG (2012) Methanotrophic communities of Saanich Inlet: A microcosm perspective. Syst Appl Microbiol 35(3):198–203
Schlesner H, Rensmann C, Tindall BJ, Gade D, Rabus R, Pfeiffer S, Hirsch P (2004) Taxonomic heterogeneity within the Planctomycetales as derived by DNA–DNA hybridization, description of Rhodopirellula baltica gen. nov., sp. nov., transfer of Pirellula marina to the genus Blastopirellula gen. nov. as Blastopirellula marina comb. nov. and emended description of the genus Pirellula. Int J Syst Evol Microbiol 54:1567–1580
Strous M, Pelletier E, Mangenot S, Rattei T, Lehner A, Taylor MW, Horn M, Daims H, Bartol-Mavel D, Wincker P, Barbe V, Fonknechten N, Vallenet D, Segurens B, Schenowitz-Truong C, Médigue C, Collingro A, Snel B, Dutilh BE, Op den Camp HJ, van der Drift C, Cirpus I, van de Pas-Schoonen KT, Harhangi HR, van Niftrik L, Schmid M, Keltjens J, van de Vossenberg J, Kartal B, Meier H, Frishman D, Huynen MA, Mewes HW, Weissenbach J, Jetten MS, Wagner M, Le Paslier D (2006) Deciphering the evolution and metabolism of an anammox bacterium from a community genome. Nature 440:790–794
Vorholt JA, Chistoserdova L, Lidstrom ME, Thauer RK (1998) The NADP-dependent methylene tetrahydromethanopterin dehydrogenase in Methylobacterium extorquens AM1. J Bacteriol 180:5351–5356
Vorholt JA, Chistoserdova L, Stolyar SM, Lidstrom ME, Thauer RK (1999) Distribution of tetrahydromethanopterin-dependent enzymes in methylotrophic bacteria and phylogeny of methenyl tetrahydromethanopterin cyclohydrolases. J Bacteriol 181:5750–5757
Vorholt JA, Marx CJ, Lidstrom ME, Thauer RK (2000) Novel formaldehyde-activating enzyme in Methylobacterium extorquens AM1 required for growth on methanol. J Bacteriol 182(23):6645–6650
Vorholt JA, Kalyuzhnaya MG, Hagemeier CH, Lidstrom ME, Chistoserdova L (2005) MtdC, a novel class of methylene tetrahydromethanopterin dehydrogenases. J Bacteriol 187:6069–6074
Vuilleumier S, Chistoserdova L, Lee MC, Bringel F, Lajus A, Zhou Y, Gourion B, Barbe V, Chang J, Cruveiller S, Dossat C, Gillett W, Gruffaz C, Haugen E, Hourcade E, Levy R, Mangenot S, Muller E, Nadalig T, Pagni M, Penny C, Peyraud R, Robinson DG, Roche D, Rouy Z, Saenampechek C, Salvignol G, Vallenet D, Wu Z, Marx CJ, Vorholt JA, Olson MV, Kaul R, Weissenbach J, Médigue C, Lidstrom ME (2009) Methylobacterium genome sequences: a reference blueprint to investigate microbial metabolism of C1 compounds from natural and industrial sources. PLoS One 4:e5584
Webster G, Sass H, Cragg BA, Gorra R, Knab NJ, Green CJ, Mathes F, Fry JC, Weightman AJ, Parkes RJ (2011) Enrichment and cultivation of prokaryotes associated with the sulphate-methane transition zone of diffusion-controlled sediments of Aarhus Bay, Denmark, under heterotrophic conditions. FEMS Microbiol Ecol 77:248–263
Woebken D, Teeling H, Wecker P, Dumitriu A, Kostadinov I, Delong EF, Amann R, Glöckner FO (2007) Fosmids of novel marine Planctomycetes from the Namibian and Oregon coast upwelling systems and their cross-comparison with planctomycete genomes. ISME J 1:419–435
Wu D, Hugenholtz P, Mavromatis K, Pukall R, Dalin E, Ivanova NN, Kunin V, Goodwin L, Wu M, Tindall BJ, Hooper SD, Pati A, Lykidis A, Spring S, Anderson IJ, D'haeseleer P, Zemla A, Singer M, Lapidus A, Nolan M, Copeland A, Han C, Chen F, Cheng JF, Lucas S, Kerfeld C, Lang E, Gronow S, Chain P, Bruce D, Rubin EM, Kyrpides NC, Klenk HP, Eisen JA (2009) A phylogeny-driven genomic encyclopaedia of Bacteria and Archaea. Nature 462:1056–1060
Acknowledgements
The author acknowledges support from the National Science Foundation (Grants MCB-0604269 and MCB-0950183).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer Science+Business Media New York
About this chapter
Cite this chapter
Chistoserdova, L. (2013). The Distribution and Evolution of C1 Transfer Enzymes and Evolution of the Planctomycetes. In: Fuerst, J. (eds) Planctomycetes: Cell Structure, Origins and Biology. Humana Press, Totowa, NJ. https://doi.org/10.1007/978-1-62703-502-6_8
Download citation
DOI: https://doi.org/10.1007/978-1-62703-502-6_8
Published:
Publisher Name: Humana Press, Totowa, NJ
Print ISBN: 978-1-62703-501-9
Online ISBN: 978-1-62703-502-6
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)