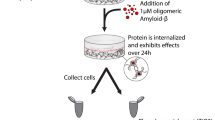
Abstract
Alzheimer’s disease (AD) and other tauopathies are characterized by the aggregation of tau into soluble and insoluble forms (including tangles and neuropil threads). In humans, a fraction of both phosphorylated and non-phosphorylated N-terminal to mid-domain tau species, are secreted into cerebrospinal fluid (CSF). Some of these CSF tau species can be measured as diagnostic and prognostic biomarkers, starting from early stages of disease. While in animal models of AD pathology, soluble tau aggregates have been shown to disrupt neuronal function, it is unclear whether the tau species present in CSF will modulate neural activity. Here, we have developed and applied a novel approach to examine the electrophysiological effects of CSF from patients with a tau-positive biomarker profile. The method involves incubation of acutely-isolated wild-type mouse hippocampal brain slices with small volumes of diluted human CSF, followed by a suite of electrophysiological recording methods to evaluate their effects on neuronal function, from single cells through to the network level. Comparison of the toxicity profiles of the same CSF samples, with and without immuno-depletion for tau, has enabled a pioneering demonstration that CSF-tau potently modulates neuronal function. We demonstrate that CSF-tau mediates an increase in neuronal excitability in single cells. We then observed, at the network level, increased input–output responses and enhanced paired-pulse facilitation as well as an increase in long-term potentiation. Finally, we show that CSF-tau modifies the generation and maintenance of hippocampal theta oscillations, which have important roles in learning and memory and are known to be altered in AD patients. Together, we describe a novel method for screening human CSF-tau to understand functional effects on neuron and network activity, which could have far-reaching benefits in understanding tau pathology, thus allowing for the development of better targeted treatments for tauopathies in the future.
Graphical Abstract

Similar content being viewed by others
Introduction
Tau plays a central role in the neuropathology of Alzheimer's disease (AD) and other tauopathies [69, 113]. Whilst insoluble high-molecular weight tau aggregates, including neurofibrillary tangles are more prominent in late phases of these diseases and can help to stage their pathological and clinical severity [21, 30, 95], the soluble forms of tau, including low-molecular weight aggregated and phosphorylated forms, are thought to be the most toxic species that modulate changes in neuronal function in the early stages of disease. In animal models of AD pathology, soluble tau aggregates have been shown to disrupt neuronal function, alter synaptic plasticity and impair cognitive function [20, 45, 81, 105, 108].
In AD, N-terminal to mid-domain tau fragments, both phosphorylated and non-phosphorylated variants, are released into the cerebrospinal fluid (CSF) early in disease progression, which has enabled the development of biomarkers for disease prognosis, diagnosis, and staging [19, 32, 57, 67, 110, 111]. While the presence of different tau forms in the CSF of AD patients is associated with neurofibrillary tangle pathology and cognitive decline [98, 99, 104, 109, 118, 119].
This leads to the question of what incubation with CSF-tau in this study might be doing to hippocampal neurons to mediate the elevation in the power of theta oscillations. A combination of experimental and simulated data has indicated that the early-stage AD hyperactivity underpinning oscillatory slowing could be due to pyramidal neuron hyperexcitability [25, 27, 49, 97, 117, 138] and/or reduced excitability of GABAergic PV interneurons and thus pyramidal cell disinhibition [4, 31, 59, 89, 97, 103, 121]. Support for the latter hypothesis of disinhibition also comes from the loss of inhibitory synapses in AD along with the pronounced GABAergic dysfunction observed in AD mouse models, including the model of APOE4, the principal genetic disease risk factor [24, 97]. Such findings provide a potential mechanism for the oscillatory disruption induced by CSF-tau incubation in this study and imply such network alterations to represent an element of the core, initial neuropathology of AD. Indeed, recent studies are now evaluating the therapeutic implementation of theta entrainment for AD patients [40, 84, 133, 134].
It has also been demonstrated that changes to theta oscillatory power—similar to what we observed herein—can be used to distinguish between prodromal AD and non-AD cases with cognitive decline [97]. The fact that our effects on oscillations agree with clinical human electrophysiological studies gives confidence that this is a method that could, in the future, be clinically useful in compliment to EEG testing.
Taking all of these changes together, it would be helpful to know whether the changes in membrane potential (4 mV depolarisation), increase in input resistance (~ 1/3) and reduction in rheobase are sufficient to account for the increases in the other parameters measured (e.g., baseline synaptic strength, PPF, LTP and theta oscillations). This question is difficult to answer without several additional experiments. In this proof of principle study, we only recorded from CA1 pyramidal cells. Thus, we do not know if similar effects on excitability also occur with CA3 pyramidal cells and with inhibitory interneurons. With this additional information it would be possible to construct computational models and for example determine whether these changes could account for the increase in theta oscillations and this will be explored further in future studies.
Conclusion
In this study, we have demonstrated that incubation of acutely-isolated wild-type mouse hippocampal brain slices with small volumes of diluted human CSF-tau allowed us to evaluate effects on neuronal function from single cells through to network level effects. Comparison of the toxicity profiles of the same CSF samples, with and without immuno-depletion for tau, enabled a pioneering demonstration that CSF-tau potently modulates neuronal function. We demonstrate that CSF-tau mediates an increase in neuronal excitability in single cells. We observed, at the network level, increased input–output responses and enhanced paired-pulse facilitation as well as an increase in long-term potentiation. Finally, we show that CSF-tau modifies the generation and maintenance of hippocampal theta oscillations, which have important roles in learning and memory and are known to be altered in AD patients. Together, we describe a novel method for screening human CSF to understand functional effects on neuron and network activity, which could have far-reaching benefits in understanding pathological mechanisms of tauopathies, thus allowing the development of better targeted treatments.
Availability of data and materials
The raw data generated and analysed for this manuscript are available from the corresponding author upon reasonable request.
References
Acquarone E, Argyrousi EK, van den Berg M, Gulisano W, Fà M, Staniszewski A, Calcagno E, Zuccarello E, D’Adamio L, Deng S-X, Puzzo D, Arancio O, Fiorito J (2019) Synaptic and memory dysfunction induced by tau oligomers is rescued by up-regulation of the nitric oxide cascade. Mol Neurodegener 14:26. https://doi.org/10.1186/s13024-019-0326-4
Ai H, Yang W, Ye M, Lu W, Yao L, Luo J (2011) Differential regulation of AMPA receptor GluA1 phosphorylation at serine 831 and 845 associated with activation of NMDA receptor subpopulations. Neurosci Lett 497:94–98. https://doi.org/10.1016/j.neulet.2011.04.038
Akay M, Wang K, Akay Y, Dragomir A, Wu J (2009) Nonlinear dynamical analysis of carbachol induced hippocampal oscillations in mice. Acta Pharmacol Sin
Ambrad Giovannetti E, Fuhrmann M (2019) Unsupervised excitation: GABAergic dysfunctions in Alzheimer’s disease. Brain Res 1707:216–226. https://doi.org/10.1016/j.brainres.2018.11.042
Amilhon B, Huh CYL, Manseau F, Ducharme G, Nichol H, Adamantidis A, Williams S (2015) Parvalbumin interneurons of hippocampus tune population activity at theta frequency. Neuron 86:1277–1289. https://doi.org/10.1016/j.neuron.2015.05.027
Andorfer C, Kress Y, Espinoza M, de Silva R, Tucker KL, Barde Y-A, Duff K, Davies P (2003) Hyperphosphorylation and aggregation of tau in mice expressing normal human tau isoforms. J Neurochem 86:582–590. https://doi.org/10.1046/j.1471-4159.2003.01879.x
Apartis E, Poindessous-Jazat FR, Lamour YA, Bassant MH (1998) Loss of rhythmically bursting neurons in rat medial septum following selective lesion of septohippocampal cholinergic system. J Neurophysiol 79:1633–1642. https://doi.org/10.1152/jn.1998.79.4.1633
Ashton NJ, Pascoal TA, Karikari TK, Benedet AL, Lantero-Rodriguez J, Brinkmalm G, Snellman A, Schöll M, Troakes C, Hye A, Gauthier S, Vanmechelen E, Zetterberg H, Rosa-Neto P, Blennow K (2021) Plasma p-tau231: a new biomarker for incipient Alzheimer’s disease pathology. Acta Neuropathol (Berl) 141:709–724. https://doi.org/10.1007/s00401-021-02275-6
Auger C, Marty A (2000) Quantal currents at single-site central synapses. J Physiol 526:3–11. https://doi.org/10.1111/j.1469-7793.2000.t01-3-00003.x
Badel L, Lefort S, Berger TK, Petersen CCH, Gerstner W, Richardson MJE (2008) Extracting non-linear integrate-and-fire models from experimental data using dynamic I-V curves. Biol Cybern 99:361–370. https://doi.org/10.1007/s00422-008-0259-4
Badel L, Lefort S, Brette R, Petersen CCH, Gerstner W, Richardson MJE (2008) Dynamic I-V curves are reliable predictors of naturalistic pyramidal-neuron voltage traces. J Neurophysiol 99:656–666. https://doi.org/10.1152/jn.01107.2007
Bassett SS, Yousem DM, Cristinzio C, Kusevic I, Yassa MA, Caffo BS, Zeger SL (2006) Familial risk for Alzheimer’s disease alters fMRI activation patterns. Brain J Neurol 129:1229–1239. https://doi.org/10.1093/brain/awl089
Begus K, Bonawitz E (2020) The rhythm of learning: Theta oscillations as an index of active learning in infancy. Dev Cogn Neurosci 45:100810. https://doi.org/10.1016/j.dcn.2020.100810
Bennys K, Rondouin G, Vergnes C, Touchon J (2001) Diagnostic value of quantitative EEG in Alzheimer’s disease. Neurophysiol Clin Neurophysiol 31:153–160. https://doi.org/10.1016/S0987-7053(01)00254-4
Berry SD, Thompson RF (1978) Prediction of learning rate from the hippocampal electroencephalogram. Science 200:1298–1300. https://doi.org/10.1126/science.663612
Bezanson J, Edelman A, Karpinski S, Shah VB (2017) Julia: a fresh approach to numerical computing. SIAM Rev 59:65–98. https://doi.org/10.1137/141000671
Bjorefeldt A, Andreasson U, Daborg J, Riebe I, Wasling P, Zetterberg H, Hanse E (2015) Human cerebrospinal fluid increases the excitability of pyramidal neurons in the in vitro brain slice. J Physiol 593:231–243. https://doi.org/10.1113/jphysiol.2014.284711
Blennow K, Chen C, Cicognola C, Wildsmith KR, Manser PT, Bohorquez SMS, Zhang Z, **e B, Peng J, Hansson O, Kvartsberg H, Portelius E, Zetterberg H, Lashley T, Brinkmalm G, Kerchner GA, Weimer RM, Ye K, Höglund K (2020) Cerebrospinal fluid tau fragment correlates with tau PET: a candidate biomarker for tangle pathology. Brain 143:650–660. https://doi.org/10.1093/brain/awz346
Blennow K, Zetterberg H (2018) Biomarkers for Alzheimer’s disease: current status and prospects for the future. J Intern Med 284:643–663. https://doi.org/10.1111/joim.12816
Booth CA, Witton J, Nowacki J, Tsaneva-Atanasova K, Jones MW, Randall AD, Brown JT (2016) Altered intrinsic pyramidal neuron properties and pathway-specific synaptic dysfunction underlie aberrant hippocampal network function in a mouse model of tauopathy. J Neurosci 36:350–363. https://doi.org/10.1523/JNEUROSCI.2151-15.2016
Braak H, Braak E (1991) Neuropathological stageing of Alzheimer-related changes. Acta Neuropathol (Berl) 82:239–259. https://doi.org/10.1007/BF00308809
Brzezicka A, Kamiński J, Reed CM, Chung JM, Mamelak AN, Rutishauser U (2019) Working memory load-related theta power decreases in dorsolateral prefrontal cortex predict individual differences in performance. J Cogn Neurosci 31:1290–1307. https://doi.org/10.1162/jocn_a_01417
Busche MA (2019) Tau suppresses neuronal activity in vivo, even before tangles form. Brain J Neurol 142:843–846. https://doi.org/10.1093/brain/awz060
Busche MA, Eichhoff G, Adelsberger H, Abramowski D, Wiederhold K-H, Haass C, Staufenbiel M, Konnerth A, Garaschuk O (2008) Clusters of hyperactive neurons near amyloid plaques in a mouse model of Alzheimer’s disease. Science 321:1686–1689. https://doi.org/10.1126/science.1162844
Busche MA, Konnerth A (2015) Neuronal hyperactivity–A key defect in Alzheimer’s disease? BioEssays News Rev Mol Cell Dev Biol 37:624–632. https://doi.org/10.1002/bies.201500004
Bush D, Bisby JA, Bird CM, Gollwitzer S, Rodionov R, Diehl B, McEvoy AW, Walker MC, Burgess N (2017) Human hippocampal theta power indicates movement onset and distance travelled. Proc Natl Acad Sci U S A 114:12297–12302. https://doi.org/10.1073/pnas.1708716114
Cardin JA, Carlén M, Meletis K, Knoblich U, Zhang F, Deisseroth K, Tsai L-H, Moore CI (2009) Driving fast-spiking cells induces gamma rhythm and controls sensory responses. Nature 459:663–667. https://doi.org/10.1038/nature08002
Celone KA, Calhoun VD, Dickerson BC, Atri A, Chua EF, Miller SL, DePeau K, Rentz DM, Selkoe DJ, Blacker D, Albert MS, Sperling RA (2006) Alterations in memory networks in mild cognitive impairment and Alzheimer’s disease: an independent component analysis. J Neurosci Off J Soc Neurosci 26:10222–10231. https://doi.org/10.1523/JNEUROSCI.2250-06.2006
Chen Z, Mengel D, Keshavan A, Rissman RA, Billinton A, Perkinton M, Percival-Alwyn J, Schultz A, Properzi M, Johnson K, Selkoe DJ, Sperling RA, Patel P, Zetterberg H, Galasko D, Schott JM, Walsh DM (2019) Learnings about the complexity of extracellular tau aid development of a blood-based screen for Alzheimer’s disease. Alzheimers Dement J Alzheimers Assoc 15:487–496. https://doi.org/10.1016/j.jalz.2018.09.010
Chung DC, Roemer S, Petrucelli L, Dickson DW (2021) Cellular and pathological heterogeneity of primary tauopathies. Mol Neurodegener 16:57. https://doi.org/10.1186/s13024-021-00476-x
Chung H, Park K, Jang HJ, Kohl MM, Kwag J (2020) Dissociation of somatostatin and parvalbumin interneurons circuit dysfunctions underlying hippocampal theta and gamma oscillations impaired by amyloid β oligomers in vivo. Brain Struct Funct 225:935–954. https://doi.org/10.1007/s00429-020-02044-3
Cicognola C, Brinkmalm G, Wahlgren J, Portelius E, Gobom J, Cullen NC, Hansson O, Parnetti L, Constantinescu R, Wildsmith K, Chen H-H, Beach TG, Lashley T, Zetterberg H, Blennow K, Höglund K (2019) Novel tau fragments in cerebrospinal fluid: relation to tangle pathology and cognitive decline in Alzheimer’s disease. Acta Neuropathol (Berl) 137:279–296. https://doi.org/10.1007/s00401-018-1948-2
Coben LA, Chi D, Snyder AZ, Storandt M (1990) Replication of a study of frequency analysis of the resting awake EEG in mild probable Alzheimer’s disease. Electroencephalogr Clin Neurophysiol 75:148–154. https://doi.org/10.1016/0013-4694(90)90168-j
Cowan CM, Chee F, Shepherd D, Mudher A (2010) Disruption of neuronal function by soluble hyperphosphorylated tau in a Drosophila model of tauopathy. Biochem Soc Trans 38:564–570. https://doi.org/10.1042/BST0380564
Crespo-García M, Zeiller M, Leupold C, Kreiselmeyer G, Rampp S, Hamer HM, Dalal SS (2016) Slow-theta power decreases during item-place encoding predict spatial accuracy of subsequent context recall. Neuroimage 142:533
Crimins JL, Rocher AB, Peters A, Shultz P, Lewis J, Luebke JI (2011) Homeostatic responses by surviving cortical pyramidal cells in neurodegenerative tauopathy. Acta Neuropathol (Berl) 122:551–564. https://doi.org/10.1007/s00401-011-0877-0
Czigler B, Csikós D, Hidasi Z, Anna Gaál Z, Csibri E, Kiss E, Salacz P, Molnár M (2008) Quantitative EEG in early Alzheimer’s disease patients - power spectrum and complexity features. Int J Psychophysiol Off J Int Organ Psychophysiol 68:75–80. https://doi.org/10.1016/j.ijpsycho.2007.11.002
DaSilva LLP, Wall MJ, de Almeida L, Wauters SC, Januário YC, Müller J, Corrêa SAL (2016) Activity-regulated cytoskeleton-associated protein controls ampar endocytosis through a direct interaction with clathrin-adaptor protein 2. eNeuro. https://doi.org/10.1523/ENEURO.0144-15.2016
Datta D, Leslie SN, Wang M, Morozov YM, Yang S, Mentone S, Zeiss C, Duque A, Rakic P, Horvath TL, van Dyck CH, Nairn AC, Arnsten AFT (2021) Age-related calcium dysregulation linked with tau pathology and impaired cognition in non-human primates. Alzheimers Dement 17:920–932. https://doi.org/10.1002/alz.12325
Di Lorenzo F, Bonnì S, Picazio S, Motta C, Caltagirone C, Martorana A, Koch G (2020) Effects of cerebellar theta burst stimulation on contralateral motor cortex excitability in patients with Alzheimer’s disease. Brain Topogr 33:613–617. https://doi.org/10.1007/s10548-020-00781-6
Dickerson BC, Salat DH, Greve DN, Chua EF, Rand-Giovannetti E, Rentz DM, Bertram L, Mullin K, Tanzi RE, Blacker D, Albert MS, Sperling RA (2005) Increased hippocampal activation in mild cognitive impairment compared to normal aging and AD. Neurology 65:404–411. https://doi.org/10.1212/01.wnl.0000171450.97464.49
Ekstrom AD, Caplan JB, Ho E, Shattuck K, Fried I, Kahana MJ (2005) Human hippocampal theta activity during virtual navigation. Hippocampus 15:881–889. https://doi.org/10.1002/hipo.20109
Engels MMA, van der Flier WM, Stam CJ, Hillebrand A, Scheltens P, van Straaten ECW (2017) Alzheimer’s disease: The state of the art in resting-state magnetoencephalography. Clin Neurophysiol Off J Int Fed Clin Neurophysiol 128:1426–1437. https://doi.org/10.1016/j.clinph.2017.05.012
Engels MMA, Hillebrand A, van der Flier WM, Stam CJ, Scheltens P, van Straaten ECW (2016) Slowing of hippocampal activity correlates with cognitive decline in early onset Alzheimer’s disease. An MEG study with virtual electrodes. Front Hum Neurosci 10:238. https://doi.org/10.3389/fnhum.2016.00238
Fá M, Puzzo D, Piacentini R, Staniszewski A, Zhang H, Baltrons MA, Li Puma DD, Chatterjee I, Li J, Saeed F, Berman HL, Ripoli C, Gulisano W, Gonzalez J, Tian H, Costa JA, Lopez P, Davidowitz E, Yu WH, Haroutunian V, Brown LM, Palmeri A, Sigurdsson EM, Duff KE, Teich AF, Honig LS, Sierks M, Moe JG, D’Adamio L, Grassi C, Kanaan NM, Fraser PE, Arancio O (2016) Extracellular tau oligomers produce an immediate impairment of LTP and memory. Sci Rep 6:19393. https://doi.org/10.1038/srep19393
Fellous J-M, Sejnowski TJ (2000) Cholinergic induction of oscillations in the hippocampal slice in the slow (0.5–2 Hz), theta (5–12 Hz), and gamma (35–70 Hz) bands. Hippocampus 10:187–197. https://doi.org/10.1002/(SICI)1098-1063(2000)10:2%3c187::AID-HIPO8%3e3.0.CO;2-M
Fernández A, Maestú F, Amo C, Gil P, Fehr T, Wienbruch C, Rockstroh B, Elbert T, Ortiz T (2002) Focal temporoparietal slow activity in Alzheimer’s disease revealed by magnetoencephalography. Biol Psychiatry 52:764–770. https://doi.org/10.1016/S0006-3223(02)01366-5
Filippini N, MacIntosh BJ, Hough MG, Goodwin GM, Frisoni GB, Smith SM, Matthews PM, Beckmann CF, Mackay CE (2009) Distinct patterns of brain activity in young carriers of the APOE-epsilon4 allele. Proc Natl Acad Sci U S A 106:7209–7214. https://doi.org/10.1073/pnas.0811879106
Ghatak S, Dolatabadi N, Trudler D, Zhang X, Wu Y, Mohata M, Ambasudhan R, Talantova M, Lipton SA (2019) Mechanisms of hyperexcitability in Alzheimer’s disease hiPSC-derived neurons and cerebral organoids vs isogenic controls. Elife 8:e50333. https://doi.org/10.7554/eLife.50333
Gobom J, Benedet AL, Mattsson-Carlgren N, Montoliu-Gaya L, Schultz N, Ashton NJ, Janelidze S, Servaes S, Sauer M, Pascoal TA, Karikari TK, Lantero-Rodriguez J, Brinkmalm G, Zetterberg H, Hansson O, Rosa-Neto P, Blennow K (2022) Antibody-free measurement of cerebrospinal fluid tau phosphorylation across the Alzheimer’s disease continuum. Mol Neurodegener 17:81. https://doi.org/10.1186/s13024-022-00586-0
Gobom J, Parnetti L, Rosa-Neto P, Vyhnalek M, Gauthier S, Cataldi S, Lerch O, Laczo J, Cechova K, Clarin M, Benet AL, Pascoal TA, Rahmouni N, Vandijck M, Huyck E, Bastard NL, Stevenson J, Chamoun M, Alcolea D, Lleó A, Andreasson U, Verbeek MM, Bellomo G, Rinaldi R, Ashton NJ, Zetterberg H, Sheardova K, Hort J, Blennow K (2022) Validation of the LUMIPULSE automated immunoassay for the measurement of core AD biomarkers in cerebrospinal fluid. Clin Chem Lab Med CCLM 60:207–219. https://doi.org/10.1515/cclm-2021-0651
Goutagny R, Jackson J, Williams S (2009) Self-generated theta oscillations in the hippocampus. Nat Neurosci 12:1491–1493. https://doi.org/10.1038/nn.2440
Goutagny R, Krantic S (2013) Hippocampal oscillatory activity in Alzheimer’s disease: toward the identification of early biomarkers? Aging Dis 4:134–140
de Haan W, Mott K, van Straaten ECW, Scheltens P, Stam CJ (2012) Activity dependent degeneration explains hub vulnerability in Alzheimer’s disease. PLOS Comput Biol 8:1002582. https://doi.org/10.1371/journal.pcbi.1002582
de Haan W, Stam CJ, Jones BF, Zuiderwijk IM, van Dijk BW, Scheltens P (2008) Resting-state oscillatory brain dynamics in Alzheimer disease. J Clin Neurophysiol Off Publ Am Electroencephalogr Soc 25:187–193. https://doi.org/10.1097/WNP.0b013e31817da184
Hämäläinen A, Pihlajamäki M, Tanila H, Hänninen T, Niskanen E, Tervo S, Karjalainen PA, Vanninen RL, Soininen H (2007) Increased fMRI responses during encoding in mild cognitive impairment. Neurobiol Aging 28:1889–1903. https://doi.org/10.1016/j.neurobiolaging.2006.08.008
Han P, Serrano G, Beach TG, Caselli RJ, Yin J, Zhuang N, Shi J (2017) A quantitative analysis of brain soluble tau and the tau secretion factor. J Neuropathol Exp Neurol 76:44–51. https://doi.org/10.1093/jnen/nlw105
Harrison PM, Badel L, Wall MJ, Richardson MJE (2015) Experimentally verified parameter sets for modelling heterogeneous neocortical pyramidal-cell populations. PLOS Comput Biol 11:e1004165. https://doi.org/10.1371/journal.pcbi.1004165
Hijazi S, Heistek TS, Scheltens P, Neumann U, Shimshek DR, Mansvelder HD, Smit AB, van Kesteren RE (2020) Early restoration of parvalbumin interneuron activity prevents memory loss and network hyperexcitability in a mouse model of Alzheimer’s disease. Mol Psychiatry 25:3380–3398. https://doi.org/10.1038/s41380-019-0483-4
Hill E, Karikari TK, Lantero-Rodriguez J, Zetterberg H, Blennow K, Richardson MJ, Wall MJ (2021) Truncating tau reveals different pathophysiological actions of oligomers in single neurons. Commun Biol 4:1265. https://doi.org/10.1038/s42003-021-02791-x
Hill E, Karikari TK, Moffat KG, Richardson MJE, Wall MJ (2019) Introduction of tau oligomers into cortical neurons alters action potential dynamics and disrupts synaptic transmission and plasticity. Eneuro 6:ENEURO.0166-19.2019. https://doi.org/10.1523/ENEURO.0166-19.2019
Hill E, Wall MJ, Moffat KG, Karikari TK (2020) Understanding the pathophysiological actions of tau oligomers: a critical review of current electrophysiological approaches. Front Mol Neurosci. https://doi.org/10.3389/fnmol.2020.00155
Hölscher C, McGlinchey L, Anwyl R, Rowan MJ (1997) HFS-induced long-term potentiation and LFS-induced depotentiation in area CA1 of the hippocampus are not good models for learning. Psychopharmacology 130:174–182. https://doi.org/10.1007/s002130050226
Horie K, Barthélemy NR, Sato C, Bateman RJ (2020) CSF tau microtubule binding region identifies tau tangle and clinical stages of Alzheimer’s disease. Brain 144:515–527. https://doi.org/10.1093/brain/awaa373
Horie K, Barthélemy NR, Spina S, VandeVrede L, He Y, Paterson RW, Wright BA, Day GS, Davis AA, Karch CM, Seeley WW, Perrin RJ, Koppisetti RK, Shaikh F, Lago AL, Heuer HW, Ghoshal N, Gabelle A, Miller BL, Boxer AL, Bateman RJ, Sato C (2022) CSF tau microtubule-binding region identifies pathological changes in primary tauopathies. Nat Med 28:2547–2554. https://doi.org/10.1038/s41591-022-02075-9
Hsiao F-J, Wang Y-J, Yan S-H, Chen W-T, Lin Y-Y (2013) Altered oscillation and synchronization of default-mode network activity in mild Alzheimer’s disease compared to mild cognitive impairment: an electrophysiological study. PLoS ONE 8:e68792. https://doi.org/10.1371/journal.pone.0068792
Ishida K, Yamada K, Nishiyama R, Hashimoto T, Nishida I, Abe Y, Yasui M, Iwatsubo T (2022) Glymphatic system clears extracellular tau and protects from tau aggregation and neurodegeneration. J Exp Med 219:e10211275. https://doi.org/10.1084/jem.20211275
Ishikawa T, Sahara Y, Takahashi T (2002) A single packet of transmitter does not saturate postsynaptic glutamate receptors. Neuron 34:613–621. https://doi.org/10.1016/S0896-6273(02)00692-X
Jack CR, Bennett DA, Blennow K, Carrillo MC, Dunn B, Haeberlein SB, Holtzman DM, Jagust W, Jessen F, Karlawish J, Liu E, Molinuevo JL, Montine T, Phelps C, Rankin KP, Rowe CC, Scheltens P, Siemers E, Snyder HM, Sperling R, Contributors, (2018) NIA-AA Research Framework: Toward a biological definition of Alzheimer’s disease. Alzheimers Dement J Alzheimers Assoc 14:535–562. https://doi.org/10.1016/j.jalz.2018.02.018
Jelic V, Johansson SE, Almkvist O, Shigeta M, Julin P, Nordberg A, Winblad B, Wahlund LO (2000) Quantitative electroencephalography in mild cognitive impairment: longitudinal changes and possible prediction of Alzheimer’s disease. Neurobiol Aging 21:533–540. https://doi.org/10.1016/s0197-4580(00)00153-6
Jeong J (2004) EEG dynamics in patients with Alzheimer’s disease. Clin Neurophysiol Off J Int Fed Clin Neurophysiol 115:1490–1505. https://doi.org/10.1016/j.clinph.2004.01.001
** Y, Su Q-X, Shen J-X, Marks MJ, Wu J (2013) Impaired hippocampal theta oscillations in the mice null alpha7 nicotinic acetylcholine receptors. CNS Neurosci Ther 19:721–723. https://doi.org/10.1111/cns.12138
Kanmert D, Cantlon A, Muratore CR, ** M, O’Malley TT, Lee G, Young-Pearse TL, Selkoe DJ, Walsh DM (2015) C-terminally truncated forms of tau, but not full-length tau or its c-terminal fragments, are released from neurons independently of cell death. J Neurosci 35:10851–10865. https://doi.org/10.1523/JNEUROSCI.0387-15.2015
Karikari TK, Ashton NJ, Brinkmalm G, Brum WS, Benedet AL, Montoliu-Gaya L, Lantero-Rodriguez J, Pascoal TA, Suárez-Calvet M, Rosa-Neto P, Blennow K, Zetterberg H (2022) Blood phospho-tau in Alzheimer disease: analysis, interpretation, and clinical utility. Nat Rev Neurol 18:400–418. https://doi.org/10.1038/s41582-022-00665-2
Karikari TK, Benedet AL, Ashton NJ, Lantero Rodriguez J, Snellman A, Suárez-Calvet M, Saha-Chaudhuri P, Lussier F, Kvartsberg H, Rial AM, Pascoal TA, Andreasson U, Schöll M, Weiner MW, Rosa-Neto P, Trojanowski JQ, Shaw LM, Blennow K, Zetterberg H (2021) Alzheimer’s Disease Neuroimaging Initiative. Diagnostic performance and prediction of clinical progression of plasma phospho-tau181 in the Alzheimer’s Disease Neuroimaging Initiative. Mol Psychiatry 26:429–442. https://doi.org/10.1038/s41380-020-00923-z
Karikari TK, Pascoal TA, Ashton NJ, Janelidze S, Benedet AL, Rodriguez JL, Chamoun M, Savard M, Kang MS, Therriault J, Schöll M, Massarweh G, Soucy J-P, Höglund K, Brinkmalm G, Mattsson N, Palmqvist S, Gauthier S, Stomrud E, Zetterberg H, Hansson O, Rosa-Neto P, Blennow K (2020) Blood phosphorylated tau 181 as a biomarker for Alzheimer’s disease: a diagnostic performance and prediction modelling study using data from four prospective cohorts. Lancet Neurol 19:422–433. https://doi.org/10.1016/S1474-4422(20)30071-5
Kim S-P, Kang J-H, Choe S-H, Jeong JW, Kim HT, Yun K, Jeong J, Lee S-H (2012) Modulation of theta phase synchronization in the human electroencephalogram during a recognition memory task. NeuroReport 23:637–641. https://doi.org/10.1097/WNR.0b013e328354afed
Koelewijn L, Lancaster TM, Linden D, Dima DC, Routley BC, Magazzini L, Barawi K, Brindley L, Adams R, Tansey KE, Bompas A, Tales A, Bayer A, Singh K (2019) Oscillatory hyperactivity and hyperconnectivity in young APOE-ɛ4 carriers and hypoconnectivity in Alzheimer’s disease. Elife 8:e36011. https://doi.org/10.7554/eLife.36011
Lantero-Rodriguez J, Snellman A, Benedet AL, Milà-Alomà M, Camporesi E, Montoliu-Gaya L, Ashton NJ, Vrillon A, Karikari TK, Gispert JD, Salvadó G, Shekari M, Toomey CE, Lashley TL, Zetterberg H, Suárez-Calvet M, Brinkmalm G, Rosa Neto P, Blennow K (2021) P-tau235: a novel biomarker for staging preclinical Alzheimer’s disease. EMBO Mol Med 13:e15098. https://doi.org/10.15252/emmm.202115098
Lasagna-Reeves CA, Castillo-Carranza DL, Sengupta U, Guerrero-Munoz MJ, Kiritoshi T, Neugebauer V, Jackson GR, Kayed R (2012) Alzheimer brain-derived tau oligomers propagate pathology from endogenous tau. Sci Rep 2:700. https://doi.org/10.1038/srep00700
Lasagna-Reeves CA, Castillo-Carranza DL, Sengupta U, Sarmiento J, Troncoso J, Jackson GR, Kayed R (2012) Identification of oligomers at early stages of tau aggregation in Alzheimer’s disease. FASEB J Off Publ Fed Am Soc Exp Biol 26:1946–1959. https://doi.org/10.1096/fj.11-199851
Leitão MJ, Silva-Spínola A, Santana I, Olmedo V, Nadal A, Le Bastard N, Baldeiras I (2019) Clinical validation of the Lumipulse G cerebrospinal fluid assays for routine diagnosis of Alzheimer’s disease. Alzheimers Res Ther 11:91. https://doi.org/10.1186/s13195-019-0550-8
Leung LS, Fu XW (1994) Factors affecting paired-pulse facilitation in hippocampal CA1 neurons in vitro. Brain Res 650:75–84. https://doi.org/10.1016/0006-8993(94)90209-7
Li G, Bien-Ly N, Andrews-Zwilling Y, Xu Q, Bernardo A, Ring K, Halabisky B, Deng C, Mahley RW, Huang Y (2009) GABAergic interneuron dysfunction impairs hippocampal neurogenesis in adult apolipoprotein E4 knockin mice. Cell Stem Cell 5:634–645. https://doi.org/10.1016/j.stem.2009.10.015
Lisman JE, Jensen O (2013) The θ-γ neural code. Neuron 77:1002–1016. https://doi.org/10.1016/j.neuron.2013.03.007
Liu G, Choi S, Tsien RW (1999) Variability of neurotransmitter concentration and nonsaturation of postsynaptic AMPA receptors at synapses in hippocampal cultures and slices. Neuron 22:395–409. https://doi.org/10.1016/S0896-6273(00)81099-5
Luo Y, Huang L, Liao P, Jiang R (2021) Contribution of neuronal and glial two-pore-domain potassium channels in health and neurological disorders. Neural Plast 2021:8643129. https://doi.org/10.1155/2021/8643129
Maeda S, Sahara N, Saito Y, Murayama S, Ikai A, Takashima A (2006) Increased levels of granular tau oligomers: an early sign of brain aging and Alzheimer’s disease. Neurosci Res 54:197–201. https://doi.org/10.1016/j.neures.2005.11.009
Martinez-Losa M, Tracy TE, Ma K, Verret L, Clemente-Perez A, Khan AS, Cobos I, Ho K, Gan L, Mucke L, Alvarez-Dolado M, Palop JJ (2018) Nav1.1-overexpressing interneuron transplants restore brain rhythms and cognition in a mouse model of Alzheimer’s disease. Neuron 98:75-89.e5. https://doi.org/10.1016/j.neuron.2018.02.029
Meredith JE Jr, Sankaranarayanan S, Guss V, Lanzetti AJ, Berisha F, Neely RJ, Slemmon JR, Portelius E, Zetterberg H, Blennow K, Soares H, Ahlijanian M, Albright CF (2013) Characterization of novel CSF tau and ptau biomarkers for Alzheimer’s disease. PLoS ONE 8:e76523. https://doi.org/10.1371/journal.pone.0076523
Mondragón-Rodríguez S, Salas-Gallardo A, González-Pereyra P, Macías M, Ordaz B, Peña-Ortega F, Aguilar-Vázquez A, Orta-Salazar E, Díaz-Cintra S, Perry G, Williams S (2018) Phosphorylation of Tau protein correlates with changes in hippocampal theta oscillations and reduces hippocampal excitability in Alzheimer’s model. J Biol Chem 293:8462–8472. https://doi.org/10.1074/jbc.RA117.001187
Moretti DV, Babiloni C, Binetti G, Cassetta E, Dal Forno G, Ferreric F, Ferri R, Lanuzza B, Miniussi C, Nobili F, Rodriguez G, Salinari S, Rossini PM (2004) Individual analysis of EEG frequency and band power in mild Alzheimer’s disease. Clin Neurophysiol 115:299–308. https://doi.org/10.1016/S1388-2457(03)00345-6
Mormino E, Brandel M, Madison C, Marks S, Baker S, Jagust WJ (2011) Aβ deposition in aging is associated with increases in brain activation during successful memory encoding. Cerebral Cortex. Oxford Academic
Musaeus CS, Engedal K, Høgh P, Jelic V, Mørup M, Naik M, Oeksengaard A-R, Snaedal J, Wahlund L-O, Waldemar G, Andersen BB (2018) EEG theta power is an early marker of cognitive decline in dementia due to Alzheimer’s disease. J Alzheimers Dis JAD 64:1359–1371. https://doi.org/10.3233/JAD-180300
Nelson PT, Alafuzoff I, Bigio EH, Bouras C, Braak H, Cairns NJ, Castellani RJ, Crain BJ, Davies P, Del Tredici K, Duyckaerts C, Frosch MP, Haroutunian V, Hof PR, Hulette CM, Hyman BT, Iwatsubo T, Jellinger KA, Jicha GA, Kövari E, Kukull WA, Leverenz JB, Love S, Mackenzie IR, Mann DM, Masliah E, McKee AC, Montine TJ, Morris JC, Schneider JA, Sonnen JA, Thal DR, Trojanowski JQ, Troncoso JC, Wisniewski T, Woltjer RL, Beach TG (2012) Correlation of Alzheimer disease neuropathologic changes with cognitive status: a review of the literature. J Neuropathol Exp Neurol 71:362–381. https://doi.org/10.1097/NEN.0b013e31825018f7
Niewiadomska G, Niewiadomski W, Steczkowska M, Gasiorowska A (2021) Tau Oligomers. Neurotoxicity Life 11:28. https://doi.org/10.3390/life11010028
van Nifterick AM, Gouw AA, van Kesteren RE, Scheltens P, Stam CJ, de Haan W (2022) A multiscale brain network model links Alzheimer’s disease-mediated neuronal hyperactivity to large-scale oscillatory slowing. Alzheimers Res Ther 14:101. https://doi.org/10.1186/s13195-022-01041-4
Ochoa JF, Alonso JF, Duque JE, Tobón CA, Baena A, Lopera F, Mañanas MA, Hernández AM (2017) Precuneus failures in subjects of the PSEN1 E280A family at risk of develo** Alzheimer’s disease detected using quantitative electroencephalography. J Alzheimers Dis 58:1229
Ochoa JF, Alonso JF, Duque JE, Tobón CA, Mañanas MA, Lopera F, Hernández AM (2017) Successful object encoding induces increased directed connectivity in presymptomatic early-onset Alzheimer’s disease. J Alzheimers Dis 55:1195–1205. https://doi.org/10.3233/JAD-160803
Oertner TG, Sabatini BL, Nimchinsky EA, Svoboda K (2002) Facilitation at single synapses probed with optical quantal analysis. Nat Neurosci 5:657–664. https://doi.org/10.1038/nn867
Osipova D, Ahveninen J, Jensen O, Ylikoski A, Pekkonen E (2005) Altered generation of spontaneous oscillations in Alzheimer’s disease. Neuroimage 27:835–841. https://doi.org/10.1016/j.neuroimage.2005.05.011
Palop JJ, Chin J, Roberson ED, Wang J, Thwin MT, Bien-Ly N, Yoo J, Ho KO, Yu G-Q, Kreitzer A, Finkbeiner S, Noebels JL, Mucke L (2007) Aberrant excitatory neuronal activity and compensatory remodeling of inhibitory hippocampal circuits in mouse models of Alzheimer’s disease. Neuron 55:697–711. https://doi.org/10.1016/j.neuron.2007.07.025
Palop JJ, Mucke L (2016) Network abnormalities and interneuron dysfunction in Alzheimer disease. Nat Rev Neurosci 17:777–792. https://doi.org/10.1038/nrn.2016.141
Pusil S, López ME, Cuesta P, Bruña R, Pereda E, Maestú F (2019) Hypersynchronization in mild cognitive impairment: the “X” model. Brain J Neurol 142:3936–3950. https://doi.org/10.1093/brain/awz320
Puzzo D, Piacentini R, Fá M, Gulisano W, Li Puma DD, Staniszewski A, Zhang H, Tropea MR, Cocco S, Palmeri A, Fraser P, D’Adamio L, Grassi C, Arancio O (2017) LTP and memory impairment caused by extracellular Aβ and Tau oligomers is APP-dependent. Elife. https://doi.org/10.7554/eLife.26991
Raghavachari S, Kahana MJ, Rizzuto DS, Caplan JB, Kirschen MP, Bourgeois B, Madsen JR, Lisman JE (2001) Gating of human theta oscillations by a working memory task. J Neurosci Off J Soc Neurosci 21:3175–3183. https://doi.org/10.1523/JNEUROSCI.21-09-03175.2001
Regan P, Piers T, Yi J-H, Kim D-H, Huh S, Park SJ, Ryu JH, Whitcomb DJ, Cho K (2015) Tau phosphorylation at serine 396 residue is required for hippocampal LTD. J Neurosci Off J Soc Neurosci 35:4804–4812. https://doi.org/10.1523/JNEUROSCI.2842-14.2015
Rocher AB, Crimins JL, Amatrudo JM, Kinson MS, Todd-Brown MA, Lewis J, Luebke JI (2010) Structural and functional changes in tau mutant mice neurons are not linked to the presence of NFTs. Exp Neurol 223:385–393. https://doi.org/10.1016/j.expneurol.2009.07.029
Rodriguez GA, Barrett GM, Duff KE, Hussaini SA (2020) Chemogenetic attenuation of neuronal activity in the entorhinal cortex reduces Aβ and tau pathology in the hippocampus. PLOS Biol 18:e3000851. https://doi.org/10.1371/journal.pbio.3000851
Sato C, Barthélemy NR, Mawuenyega KG, Patterson BW, Gordon BA, Jockel-Balsarotti J, Sullivan M, Crisp MJ, Kasten T, Kirmess KM, Kanaan NM, Yarasheski KE, Baker-Nigh A, Benzinger TLS, Miller TM, Karch CM, Bateman RJ (2018) Tau kinetics in neurons and the human central nervous system. Neuron 97:1284-1298.e7. https://doi.org/10.1016/j.neuron.2018.02.015
Scheltens P, Blennow K, Breteler MMB, de Strooper B, Frisoni GB, Salloway S, Van der Flier WM (2016) Alzheimer’s disease. Lancet Lond Engl 388:505–517. https://doi.org/10.1016/S0140-6736(15)01124-1
Schreiter-Gasser U, Gasser T, Ziegler P (1994) Quantitative EEG analysis in early onset Alzheimer’s disease: correlations with severity, clinical characteristics, visual EEG and CCT. Electroencephalogr Clin Neurophysiol 90:267–272. https://doi.org/10.1016/0013-4694(94)90144-9
Serrano-Pozo A, Frosch MP, Masliah E, Hyman BT (2011) Neuropathological alterations in Alzheimer disease. Cold Spring Harb Perspect Med 1:a006189. https://doi.org/10.1101/cshperspect.a006189
Setti SE, Hunsberger HC, Reed MN (2017) Alterations in hippocampal activity and Alzheimer’s disease. Transl Issues Psychol Sci 3:348–356. https://doi.org/10.1037/tps0000124
Shafiei SS, Guerrero-Muñoz MJ, Castillo-Carranza DL (2017) Tau oligomers: cytotoxicity, propagation, and mitochondrial damage. Front Aging Neurosci 9
Simrén J, Brum WS, Ashton NJ, Benedet AL, Karikari TK, Kvartsberg H, Sjons E, Lussier FZ, Chamoun M, Stevenson J, Hopewell R, Pallen V, Ye K, Pascoal TA, Zetterberg H, Rosa-Neto P, Blennow K (2022) CSF tau368/total-tau ratio reflects cognitive performance and neocortical tau better compared to p-tau181 and p-tau217 in cognitively impaired individuals. Alzheimers Res Ther 14:192. https://doi.org/10.1186/s13195-022-01142-0
Sohal VS, Zhang F, Yizhar O, Deisseroth K (2009) Parvalbumin neurons and gamma rhythms enhance cortical circuit performance. Nature 459:698–702. https://doi.org/10.1038/nature07991
Sperling RA, Dickerson BC, Pihlajamaki M, Vannini P, LaViolette PS, Vitolo OV, Hedden T, Becker JA, Rentz DM, Selkoe DJ, Johnson KA (2010) Functional alterations in memory networks in early Alzheimer’s disease. Neuromolecular Med 12:27–43. https://doi.org/10.1007/s12017-009-8109-7
Stam CJ, de Haan W, Daffertshofer A, Jones BF, Manshanden I, van Walsum AM, Montez T, Verbunt JPA, de Munck JC, van Dijk BW, Berendse HW, Scheltens P (2009) Graph theoretical analysis of magnetoencephalographic functional connectivity in Alzheimer’s disease. Brain J Neurol 132:213–224. https://doi.org/10.1093/brain/awn262
Stan GF, Church TW, Randall E, Harvey JRM, Brown JT, Wilkinson KA, Hanley JG, Marrion NV (2022) Tau isoform-specific enhancement of L-type calcium current and augmentation of afterhyperpolarization in rat hippocampal neurons. Sci Rep 12:15231. https://doi.org/10.1038/s41598-022-18648-0
Stefanovski L, Triebkorn P, Spiegler A, Diaz-Cortes M-A, Solodkin A, Jirsa V, McIntosh AR, Ritter P (2019) Alzheimer’s Disease Neuroimaging Initiative. Linking molecular pathways and large-scale computational modeling to assess candidate disease mechanisms and pharmacodynamics in Alzheimer’s disease. Front Comput Neurosci 13:54. https://doi.org/10.3389/fncom.2019.00054
Targa Dias Anastacio H, Matosin N, Ooi L (2022) Neuronal hyperexcitability in Alzheimer’s disease: what are the drivers behind this aberrant phenotype? Transl Psychiatry 12:1–14. https://doi.org/10.1038/s41398-022-02024-7
Teles-Grilo Ruivo LM, Mellor JR (2013) Cholinergic modulation of hippocampal network function. Front Synaptic Neurosci 5:2. https://doi.org/10.3389/fnsyn.2013.00002
Tracy TE, Gan L (2018) Tau-mediated synaptic and neuronal dysfunction in neurodegenerative disease. Curr Opin Neurobiol 51:134–138. https://doi.org/10.1016/j.conb.2018.04.027
Uhlenbeck GE, Ornstein LS (1930) On the theory of the brownian motion. Phys Rev 36:823–841. https://doi.org/10.1103/PhysRev.36.823
Vardalaki D, Chung K, Harnett MT (2022) Filopodia are a structural substrate for silent synapses in adult neocortex. Nature 612:323–327. https://doi.org/10.1038/s41586-022-05483-6
Verret L, Mann EO, Hang GB, Barth AMI, Cobos I, Ho K, Devidze N, Masliah E, Kreitzer AC, Mody I, Mucke L, Palop JJ (2012) Inhibitory interneuron deficit links altered network activity and cognitive dysfunction in Alzheimer model. Cell 149:708–721. https://doi.org/10.1016/j.cell.2012.02.046
Vertes RP, Hoover WB, Viana Di Prisco G (2004) Theta rhythm of the hippocampus: subcortical control and functional significance. Behav Cogn Neurosci Rev 3:173–200. https://doi.org/10.1177/1534582304273594
Ward SM, Himmelstein DS, Lancia JK, Binder LI (2012) Tau oligomers and tau toxicity in neurodegenerative disease. Biochem Soc Trans 40:667–671. https://doi.org/10.1042/BST20120134
Williams JH, Kauer JA (1997) Properties of carbachol-induced oscillatory activity in rat hippocampus. J Neurophysiol 78:2631–2640. https://doi.org/10.1152/jn.1997.78.5.2631
Winson J (1978) Loss of hippocampal theta rhythm results in spatial memory deficit in the rat. Science 201:160–163. https://doi.org/10.1126/science.663646
Witton J, Staniaszek LE, Bartsch U, Randall AD, Jones MW, Brown JT (2016) Disrupted hippocampal sharp-wave ripple-associated spike dynamics in a transgenic mouse model of dementia. J Physiol 594:4615–4630. https://doi.org/10.1113/jphysiol.2014.282889
Wu X, Ji G-J, Geng Z, Wang L, Yan Y, Wu Y, **ao G, Gao L, Wei Q, Zhou S, Wei L, Tian Y, Wang K (2022) Accelerated intermittent theta-burst stimulation broadly ameliorates symptoms and cognition in Alzheimer’s disease: a randomized controlled trial. Brain Stimulat 15:35–45. https://doi.org/10.1016/j.brs.2021.11.007
Wu X, Ji G-J, Geng Z, Zhou S, Yan Y, Wei L, Qiu B, Tian Y, Wang K (2020) Strengthened theta-burst transcranial magnetic stimulation as an adjunctive treatment for Alzheimer’s disease: an open-label pilot study. Brain Stimulat 13:484–486. https://doi.org/10.1016/j.brs.2019.12.020
Zhang J, Yang Y, Li H, Cao J, Xu L (2005) Amplitude/frequency of spontaneous mEPSC correlates to the degree of long-term depression in the CA1 region of the hippocampal slice. Brain Res 1050:110–117. https://doi.org/10.1016/j.brainres.2005.05.032
Zhang W, Falcon B, Murzin AG, Fan J, Crowther RA, Goedert M, Scheres SH (2019) Heparin-induced tau filaments are polymorphic and differ from those in Alzheimer’s and Pick’s diseases. Elife 8:e43584. https://doi.org/10.7554/eLife.43584
Zhu G, Liu Y, Wang Y, Bi X, Baudry M (2015) Different patterns of electrical activity lead to long-term potentiation by activating different intracellular pathways. J Neurosci 35:621–633. https://doi.org/10.1523/JNEUROSCI.2193-14.2015
Zott B, Simon MM, Hong W, Unger F, Chen-Engerer H-J, Frosch MP, Sakmann B, Walsh DM, Konnerth A (2019) A vicious cycle of β amyloid-dependent neuronal hyperactivation. Science 365:559–565. https://doi.org/10.1126/science.aay0198
Acknowledgements
We would like to thank Professor Bruno Frenguelli for providing an interface chamber for the recording of theta oscillations.
Funding
EH holds a Race Against Dementia Fellowship funded by the Barbara Naylor Foundation, in collaboration with ARUK. JB is funded by the Medical Research Council doctoral training partnership at The University of Manchester. AW is funded by the Medical Research Council doctoral training partnership at the University of Warwick. TKK is funded by the Swedish Research Council (Vetenskapsrådet #2021-03244), the Alzheimer’s Association Research Fellowship (#AARF-21-850325), the Aina (Ann) Wallströms and Mary-Ann Sjöbloms stiftelsen, and the Emil och Wera Cornells stiftelsen. HZ is a Wallenberg Scholar supported by grants from the Swedish Research Council (#2022-01018), the European Union’s Horizon Europe research and innovation programme under grant agreement No 101053962, Swedish State Support for Clinical Research (#ALFGBG-71320), the Alzheimer Drug Discovery Foundation (ADDF), USA (#201809-2016862), the AD Strategic Fund and the Alzheimer's Association (#ADSF-21-831376-C, #ADSF-21-831381-C, and #ADSF-21-831377-C), the Bluefield Project, the Olav Thon Foundation, the Erling-Persson Family Foundation, Stiftelsen för Gamla Tjänarinnor, Hjärnfonden, Sweden (#FO2022-0270), the European Union’s Horizon 2020 research and innovation programme under the Marie Skłodowska-Curie grant agreement No 860197 (MIRIADE), the European Union Joint Programme—Neurodegenerative Disease Research (JPND2021-00694), and the UK Dementia Research Institute at UCL (UKDRI-1003). KB is supported by the Swedish Research Council (#2017-00915 and #2022-00732), the Swedish Alzheimer Foundation (#AF-930351, #AF-939721 and #AF-968270), Hjärnfonden, Sweden (#FO2017-0243 and #ALZ2022-0006), the Swedish state under the agreement between the Swedish government and the County Councils, the ALF-agreement (#ALFGBG-715986 and #ALFGBG-965240), the European Union Joint Program for Neurodegenerative Disorders (JPND2019-466-236), the Alzheimer’s Association 2021 Zenith Award (ZEN-21-848495), and the Alzheimer’s Association 2022-2025 Grant (SG-23-1038904 QC).
Author information
Authors and Affiliations
Contributions
Conceptualization: EH, MJW, TKK, JB, Methodology: EH, MJW, TKK, JB, Formal analysis, and Investigation: EH, MJW, JB, EC, JLR, MO, AW, BM, Writing—original draft preparation: EH, Writing—review and editing: EH, MJW, TKK, JB, EC, JLR, MO, AW, BM, HZ, KB, Resources: EH, MJW, TKK, EC, JLR, MO, HZ, KB, Supervision: EH, MJW, TKK.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
CSF collection and processing were performed at the University of Gothenburg, Sweden, with local ethical approval, and the samples sent to the University of Warwick, UK, where all experiments were done as approved by the local Human Tissue Authority and Biomedical & Scientific Research Ethics Committees. All animal care and experimental procedures were reviewed and approved by the institutional animal welfare and ethical review body (AWERB) at the University of Warwick.
Consent for publication
All authors have approved the manuscript for submission and that the contents have not been published or submitted elsewhere.
Competing interests
The authors have no conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
40478_2023_1562_MOESM1_ESM.docx
Additional file 1. Figure 1. Dose response for the effect of CSF-tau on neuronal function. Figure 2. Protocol for analysis of hippocampal CA3 theta oscillations. Figure 3. Validation of tau effects using CSF-mock-depleted.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Brown, J., Camporesi, E., Lantero-Rodriguez, J. et al. Tau in cerebrospinal fluid induces neuronal hyperexcitability and alters hippocampal theta oscillations. acta neuropathol commun 11, 67 (2023). https://doi.org/10.1186/s40478-023-01562-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s40478-023-01562-5