Abstract
Pyrrolizidine alkaloids (PAs) that are plant toxin naturally produced for protection against herbivores in some plant families. They are associated with the potential hepatotoxic and carcinogenic diseases serious hepatic disease in humans and animals. As the concern of human health risk posed by exposure to PAs has been gradually increased, precise and reliable analysis is required for monitoring PAs. The present study developed a new and simple pretreatment using 50% MeOH (methanol) for quantification analysis of the PAs contained with high content in the herbal medicines. Another pretreatment method using cation-ion exchange solid-phase extraction (MCX-SPE) was employed for determining most of the PAs that are not contained in the herbal medicines. That is, the proposed LC–MS/MS method coupled with MCX-SPE extraction and 50% MeOH extraction method was developed. And to evaluate the reliability of its application for Farfarae Flos and Lithospermi Radix, a validation study was conducted. In addition, monitory study was performed with ten samples in each herbal medicine. As a result, the proposed method had good linearity with r2 ≥ 0.997. Also, the recoveries indicated to be in the ranges of 70.4–118.0% for the Farfarae Flos, 70.2–119.7% for the Lithospermi Radix. In two herbal medicines, the intra-day precision was revealed to satisfy the reference criteria in most of the PAs. In monitoring results, most of the PAs were not contained in two herbal medicines, whereas a part of PAs revealed to have high concentration in Farfarae Flos and Lithospermi Radix. The proposed method is considered as a simple and reliable method to quantify 28 PAs present in two herbal medicines. Especially, the simple MeOH extraction method seems to be available for quantification analysis of certain PAs in herbal medicines with high content.
Similar content being viewed by others
Introduction
As the use of traditional medicines in developed countries is exponentially growing, the concern associated with the human health risk by exposure of Pyrrolizidine alkaloids (PAs) has gradually increased because raw plant materials widely distributed in the world are used for medicinalpurposes such as dietary supplements and traditional medicines. PAs are secondary plant metabolites, produced naturally for protection against herbivores in some plant families [1]. Exposure of several PAs over longer periods has been known to cause hepatotoxicity, genotoxicity and carcinogenic potential in humans and animals for decades [2). However, unlike othr herbal medicines, there was difficulty in performance of the validation study of the two herbal medicines. That is, we found that Farfarae Flos contained 3 PAs including Senecionine, Senecionine N-oxide, Senkirkine, whereas Lithospermi Radix had 5 PAs such as Echimedine, Echimedine N-oxide, Intermedine, and Intermedine N-oxide, Lycopsamine N-oxide through the preliminary test.
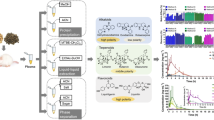
The blank samples that do not contain the analytes are required for measuring the precise recovery in the validation study. Due to their extremely high content, they occurred easily saturation at LC–MS/MS analysis that quantifies the trace amount in samples to deviate quantitative range so that their recoveries could not be quantified simultaneously with those of the most PAs. To overcome such problem, this study developed a new extraction method for these PAs contained with high content in the herbal medicine samples. The new extraction was to a strategy inducing dilution effect. That is, it was a simple extraction that small amount sample (0.1 g) was extracted with a large amount (20–200 mL) of 50% MeOH without the purification using cation-ion cartridge (Fig. 2B).
Lebada et al. [36] explain that the best method to extract senkirkine and senecionine from Tussilago farfara was a combination comprised of refluxing with 50% methanol acidified and purification using solid-phase extraction on diol‐phase cartridges. The content of senkirkine in coltsfoot leaves was measured using capillary zone electrophoresis and its concentration was 2.5–11.2 ppm. Such a large variation of the amount of PAs seems to be ascribed to a different origin of plant materials, its preparation (raw material or dried samples), extraction methods (with various solvents), and quantification analytical methods (capillary zone electrophoresis, gas, or liquid chromatography) [36, 44]. To quantify the PAs in herbal medicines, the selection of appropriate solvent for extraction of PAs and analytical method is considered to be an important work. In addition, as PAs contained with a large amount in herbal medicines can occur easily saturation, the analysis methods used in practice should be validated to ensure its reliability prior to quantification analysis.
Matrix effects
As most herbal medicines are natural plants that contain various chemical compounds, they may influence the quantification analysis of pyrrolizidine alkaloids. Therefore, this study evaluated the matrix effects of 28 PAs in Farfarae Flos and Lithospermi Radix. The matrix effects (ME) were evaluated by the following categories: (i) high signal suppression (− 50% > ME) and moderate signal suppression (− 50% < ME > − 20%); (ii) no matrix effect (− 20% < ME > 20%); (iii) moderate signal enhancement (20% < ME > 50%), and high signal enhancement (ME > 50%) [45]. As Farfarae Flow contained 3 PAs such as Senecionine, Senecionine N-oxide, and Senkirkine and Lithospermi Radix included Intermedine, and Intermedine N-oxide with high content. Matrix effects of these PAs could not be successfully evaluated because the coefficient deviations (r2) value of standard in these PAs curve could been not acquired as more than 0.99 so that their quantification analysis could not be accomplished.
As shown in Fig. 4 the matrix effects of the other PAs indicated as follows; In Farfarae Flow, 5 PAs such as Echimidine-N-oxide (EmN), Europine (Eu), Lasiocarpine (Lc), Lasiocarpine N-oxide (LcN), and Retrorsine N-oxide (ReN) indicated strong ion enhancement, whereas 4 PAs including Echimidine (Em), Erucifoline N-oxide (ErN), Jacobine (Jb), Lycopsamine N-oxide (LcN) occurred strong ion suppression. In Lithospermi Radix, 4 PAs such as Echimidine-N-oxide (EmN), Erucifoline N-oxide (ErN), Heliotrine N-oxide (HnN), Lycopsamine (La) showed strong ion enhancement, while 7 PAs like Erucifoline occurred ion suppression. Considering these results, quantification analysis against most of the analytes seemed to be influenced by various components originated from herbal medicine samples so that this study conducted the matrix-matched analysis to avoid or reduce the interfering of various materials contained in the herbal medicine samples.
Matrix effects of 28 pyrrolizidine alkaloids in Farfarae Flos and Lithospermi Radix; 1: Echimidine, 2: Echimidine-N-oxide, 3: Erucifoline, 4: Erucifoline-N-oxide, 5: Europine, 6: Europine-N-oxide, 7: Heliotrine, 8: Heliotrine-N-oxide, 9: Intermedine, 10: Intermedine-N-oxide, 11: Jacobine, 12: Jacobine-N-oxide, 13: Lasiocarpine, 14: Lasiocarpine-N-oxide, 15: Lycopsamine, 16: Lycopsamine-N-oxide, 17: Monocrotaline, 18: Monocrotaline-N-oxide, 19: Retrorsine, 20: Retrorsine-N-oxide, 21: Senecionine, 22: Senecionine-N-oxide, 23: Seneciphylline, 24: Seneciphylline-N-oxide, 25: Senecivernine, 26: Senecivernine-N-oxide, 27: Senkirkine, 28: Trichodesmine
Linearity and sensitivity
Because of matrix effects in herbal medicines, our validation study was performed by matrix-matched analysis. The results of linearity in most of the PAs revealed to have good linearity, indicating r2 ≥ 0.997 in Farfarae Flos or Lithospermi Radix (Tables 3 and 4). The linearity of 3 PAs (Senecionine, Senecionine N-oxide, Senkirkine) contained in Farfarae Flos and 2 PAs ( Intermedine and Intermedine N-oxide) included in Lithospermi Radix revealed to have good linearities, indicating r2 = 0.999. In Farfarae Flos, LOQs of 25 PAs included in the original samples were revealed to have the range of 0.1–24.2 μg/kg (Table 3). In Lithospermi Radix, LOQs of 24 PAs contained in Lithospermi Radix indicated to be the range 0.2–7.2 μg/kg. The LOQs of Seneciphylline and Retrorsin N-oxide indicated to have 23.0 μg/kg, and 17.4 μg/kg in Lithospermi Radix respectively, indicating comparatively higher LOQ than that of the other PAs (Table 4). The improved LC–MS/MS method indicated that Lycopsamine N-oxide had 1.1 μg/kg of LOQ in Farfarae Flos. Through such results, the matrix components seemed to influence LOQs of the PA.
Also, we assumed that certain components that contained Lithospermi Radix might occur strong suppression of Lycopasamine N-oxide. As 3 PAs such as Senecione, Senecione N-oxide, and Senkerkine in Farfarae Flos and 5 PAs intermedine, inter medine N-oxide, Echimidine, and Echimidine N-oxide, and Lycopsamine N-oxide in Lithospermi Radix were contained with a large amount in herbal medicines so that the saturation occurs, their LOD and LOQ could not be measured. BfR-PA-Tea method indicated to have 0.5–1.7 μg/kg of LOD and 1.7–6.4 μg/kg of LOQ for 28 PAs. The proposed method revealed to have LOQ values less than 10 μg/kg in most PAs, except of a few PAs. The LOQ values obtained by the proposed method was not largely different from those of BfR method.
Accuracy and precision
The recoveries and relative standard deviations (RSDs) of PAs were evaluated through the matrix-matched analysis. The test solutions were prepared as follows; after the mixed standard solutions were spiked into the samples to be three different concentration levels and then pretreated by the method described above. However, These PAs that contained a large amount in Farfarae Flos or Lithospermi Radix, the recoveries and RSDs were measured by quantification method using standard solution instead of matrix-matched analysis. In test method validation, AOAC guideline requires the reference criteria such as recovery of 70–125%, repeatability precision (intra-day RSD) < 15%, and the reproducibility precision (inter-day RSD) < 32%.
In Farfarae Flos, most PAs that were not contained in Farfarae Flos showed satisfactory recovery as the ranges of 70.4–118.0%. In precision evaluation of Farfarae Flos, most analytes indicated satisfactory intra-day RSD values, however, a few analytes showed to be beyond the criteria in intra-day RSD as follows; Echimidine(18.8%), Retrorsine (20.1%), Seneciphylline (17.6%), Seneciphylline N-oxide (16.9%), Seneciverine (17.8%). In inter-day RSDs, 25 PAs indicated to be less than the criteria recommended at AOAC guideline (Table 3).
In Lithospermi Radix, most of the PAs that were not contained in the Lithospermi Radix indicted the ranges of 70.2–116.5%. Their intra-day RSDs of all analytes except 2 PAs, Europine N-oxide (24.2%) and Jacobine (18.5%), showed to be less than 15% and the inter-day RSDs of 24 PAs indicated to have values less than 32% (Table 3).
Three PAs including senecionine, senecionine N-oxide and senkirkine showed satisfactory recovery in Farfarae Flos, indicating the ranges of 92.8–119.0% and their intra-and inter-day RSDs also indicated to satisfy the recommended criteria. Also, in Lithospermi Radix, 5 PAs considered to have high content in Lithospermi Radix revealed recovery ranges of 108.5–130.3%, even though only Echimidine N-oxide did not satisfy the criteria, indicating 130.2% of recovery. Their intra-and inter-day RSDs had satisfactory values in in Lithospermi Radix (Table 4). On the basis of these results, the proposed method was considered as a great quantification analysis that can determine some PAs with high content such as 3 PAs in Farfare Flos and 5 PAs in Lithospermi Radix as well as most of the PAs used in this study.
Monitoring of PAs
The developed LC–MS/MS method was applied to determine the content of 28 PAs in 20 herbal medicine samples purchased from Korean herbal medicine markets imported from China. Ten herbal medicine samples per matrix were used for the monitoring study. Most PAs among 28 PAs were not detected or indicated to have contents of less than the LOQ in two herbal medicine samples, Farfarae Flos and Lithospermi Radix. However, they revealed to possess a part of PAs with high concentration in. In Farfarae Flos, all samples contained three PAs such as senecionine (6.9–9.5 μg/g), senecionine N-oxide (6.2–13.7 μg/g), and senkirkine (97.4–175.1 μg/g), whereas in Lithospermi Radix, all samples showed to have four PAs such as intermedine (2.3–30.9 μg/g), intermedine N-oxide (27.1–78.5 μg/g), echimedine (0.4–1.6 µg/g), and echimedine N-oxide (0.7–5.5 µg/g) and only one sample contained Lycopsamine (1.73 µg/g) and Lycopsamine N-oxide (7.82 µg/g).
According to the literature, Adamczak et al. [35] quantified the alkaloid content (Senkirkine and Senecionine) in T. farfara using HPLC analysis method. The method employs two-time extraction by using 50% (v/v) methanol acidified with citric acid and dichloromethane and diethyl ether. After evaporating the solvent, the dissolved residual solution was purified by SPE extraction. As a result, their content indicated 0.02–0.58 of Senecionine and 0.02–0.47 μg/g of Senkirkine. Also, Tussilago farfara are reported to contain toxic PAs with various ranges from 0.1 to 368 μg/g [36, 44, 46,47,48]. Also, some researchers claim that dry drugs of Tussilago farfara (Farfarae Flos) contain senkirkine from 0.1 to 150 ppm [49, 50] and senecionine ranging from 0.1 to 10 ppm [50]. In comparing these results, the contents of senkirkine and senecionine obtained by the proposed method seemed to be not largely different from their content reported previously. Therefore, the proposed method was considered to determine well such PAs in herbal medicinal samples. In addition, this study suggests that it is are necessary to monitor their residual amount in the final herbal medicinal products used Farfarae Flos or Lithospermi Radix as well as the herbal medicines through a further study.
References
Koleva II, van Beek TA, Soffers AE, Dusemund B, Rietjens IM (2012) Alkaloids in the human food chain-natural occurrence and possible adverse effects. Mol Nutr Food Res 56(1):30–52
Pp FU, Yang YC, **a Q, Chou MW, Cui YY, Lin G (2002) Pyrrolizidine alkaloids—tumorigenic components in Chinese herbal medicines and dietary supplements. J Food Drug Anal 10(4):198–211
EMA (European Medicines Agency) (2014) Public statement on the use of herbal medicinal products containing toxic, unsaturated pyrrolizidine alkaloids. Ema.europa.eu. Retrieved 10 March 2021, from https://www.ema.europa.eu/en/documents/public-statement/draft-public-statement-use-herbal-medicinal-products-containing-toxic-unsaturated-pyrrolizidine_en-0.pdf
Kakar F, Akbarian Z, Leslie T, Mustafa ML, Watson J, VanEgmond HP, Mofleh J (2010) An outbreak of hepatic veno-occlusive disease in Western Afghanistan associated with exposure to wheat flour contaminated with pyrrolizidine alkaloids. J Toxicol 2010(7980):313280. https://doi.org/10.1144/2010/313280
Roeder E, Wiedenfeld H, Edgar JA (2015) Pyrrolizidine alkaloids in medicinal plants from North America. Pharmazie 70(6):357–367
IARC (1987) IARC monographs on the evaluation of carcinogenic risks to humans, Supplement 6, genetic and related effects: an updating of selected IARC Monographs from Volumes 1 to 42, Lyon
IARC (International Agency for Research on Cancer) (2002) Working Group on the Evaluation of Carcinogenic Risks to Humans. Some traditional herbal medicines, some mycotoxins, naphthalene and styrene (Vol. 82)
Cong L, Hongwei W, Lixia W, Hanyan L, Yaqi L, Qiong Z, Liying T, Zhuju W (2020) Farfarae Flos: a review of botany, traditional uses, phytochemistry, pharmacology, and toxicology. J Ethnopharmacol 260:113038
Tobyn G, Denham A, Whitelegg M (2011) Tussilago Farfara, Coltsfoot. Medical Herbs. pp. 317–326
Xue SY, Li ZY, Zhi HJ, Sun HF, Zhang LZ, Guo XQ, Qin XM (2012) Metabolic fingerprinting investigation of Tussilago farfara L. by GC–MS and multivariate data analysis. Biochem Syst Ecol 41:6–12
Cheon HJ, Nam SH, Kim JK (2018) Tussilagone, a major active component in Tussilago farfara, ameliorates inflammatory responses in dextran sulphate sodium-induced murine colitis. Chem Biol Interact 294:74–80
Qin K, Liu C, Qi Y, Li K (2014) Evaluation of antioxidant activity of polysaccharides from Tussilago farfara L. by flow injection analysis. Asian J Chem 26(10):3073
Uysal S, Senkardes I, Mollica A, Zengin G, Bulut G, Dogan A, Glamoclija J, Sokovic M, Lobine D, Mahomoodally FM (2019) Biologically active compounds from two members of the Asteraceae family: Tragopogon dubius Scop. and Tussilago farfara L. J Biomol Struct Dyn 37(12):3269–3281
Gao H, Huang YN, Gao B, Xu PY, Inagaki C, Kawabata J (2008) α-Glucosidase inhibitory effect by the flower buds of Tussilago farfara L. Food Chem 106(3):1195–1201
Lee J, Song K, Huh E, Oh MS, Kim YS (2018) Neuroprotection against 6-OHDA toxicity in PC12 cells and mice through the Nrf2 pathway by a sesquiterpenoid from Tussilago farfara. Redox Biol 18:6–15
Hwang SB, Chang MN, Garcia ML, Han QQ, Huang L, King VF, Winquist RJ (1987) L-652,469-a dual receptor antagonist of platelet activating factor and dihydropyridines from Tussilago farfara L. Eur J Pharmacol 141(2):269–281
Li H, Lee HJ, Ahn YH, Kwon HJ, Jang CY, Kim WY, Ryu JH (2014) Tussilagone suppresses colon cancer cell proliferation by promoting the degradation of β-catenin. Biochem Biophys Res Commun 443(1):132–137
Kopp T, Abdel-Tawab M, Mizaikoff B (2020) Extracting and analyzing pyrrolizidine alkaloids in medicinal plants: a review. Toxins 12(5):320
Yazaki K (2017) Lithospermum erythrorhizon cell cultures: present and future aspects. Plant Biotechnol 34(3):131–142
Andujar I, Recio MC, Giner RM, Rios JL (2013) Traditional chinese medicine remedy to jury: the pharmacological basis for the use of shikonin as an anticancer therapy. Curr Med Chem 20(23):2892–2898
Sun H, Xu H, Zhang A, Wang P, Han Y, Wang X (2014) In vitro anticancer activity of acetylshikonin action against cervical cancer. Plant Science Today 1(2):39–45
Sasaki K, Abe H, Yoshizaki F (2002) In vitro antifungal activity of naphthoquinone derivatives. Biol Pharm Bull 25(5):669–670
Tabata M, Tsukada M, Fukui H (1982) Antimicrobial activity of quinone derivatives from Echium lycopsis callus cultures. Planta Med 44:234–236
Hisa T, Kimura Y, Takada K, Suzuki F, Takigawa M (1998) Shikonin, an ingredient of Lithospermum erythrorhizon, inhibits angiogenesis in vivo and in vitro. Anticancer Res 18(2A):783–790
Ahn BZ, Baik KU, Kweon GR, Lim K, Hwang BD (1995) Acylshikonin analogs: synthesis and inhibition of DNA topoisomerase-I. J Med Chem 38(6):1044–1047
Arseculeratne SN, Gunatilaka AL, Panabokke RG (1981) Studies on medicinal plants of Sri Lanka: occurrence of pyrrolizidine alkaloids and hepatotoxic properties in some traditional medicinal herbs. J Ethnopharmacol 4(2):159–177
Manteiga R, Park DL, Ali SS (1997) Risks associated with consumption of herbal teas. In: Ware GW, editors. Reviews of environmental contamination and toxicology. Reviews of environmental contamination and toxicology vol. 150, Springer, New York, NY. https://doi.org/10.1007/978-1-4612-2278-1_1
BfR (German Federal Institute for Risk Assessment) (2013) Pyrrolizidine alkaloids in herbal teas and teas. Opinion No. 018/2013 of 5 July 2013. Berlin 1032 (Germany): Bundesinstitut für Risikobewertung. Available at: 1033. https://www.bfr.bund.de/cm/349/pyrrolizidine-alkaloids-in-herbal-teas-and-teas.pdf. Accessed 1034 Jun 2020
EMA. (2016) Public statement on contamination of herbal medicinal products/traditional herbal medicinal products with pyrrolizidine alkaloids. Ema.europa.eu. Retrieved 20 June 2020, from https://www.ema.europa.eu/en/documents/public-statement/public-statement-contamination-herbal-medicinal-products/traditional-herbal-medicinal-products-pyrrolizidine-alkaloids_en.pdf
EMA (2019) Assessment report. Referral under Article 31 of Directive 2001/83/EC angiotensin-II receptor antagonists (sartans) containing a tetrazole group. Ema.europa.eu. Retrieved 10 March 2021, from https://www.ema.europa.eu/en/documents/variation-report/angiotensin-ii-receptor-antagonists-sartans-article-31-referral-chmp-assessment-report_en.pdf
Steinhoff B (2019) Pyrrolizidine alkaloid contamination in herbal medicinal products: limits and occurrence. Food Chem Toxicol 130:262–266
BfArM (The Federal Institute for Drugs and Medical Devices, Germany) (2016) Available at: https://www.bfarm.de/DE/Arzneimittel/Arzneimittelzulassung/Zulassungsarten/BesondereTherapierichtungen/Allgemeines/_node.html. Assessed Jun 2020
EDQM (European Directorate for the Quality of Medicines & Healthcare) (2021) European Pharmacopoeia Commission adopts new general chapter Contaminant pyrrolizidine alkaloids (2.8.26). (2021, January 05). Retrieved from https://www.edqm.eu/en/news/european-pharmacopoeia-commission-adopts-new-general-chapter-contaminant-pyrrolizidine
Smyrska-Wieleba N, Wojtanowski KK, Mroczek T (2017) Comparative HILIC/ESI-QTOF-MS and HPTLC studies of pyrrolizidine alkaloids in flowers of Tussilago farfara and roots of Arnebia euchroma. Phytochem Lett 20:339–349
Adamczak A, Opala B, Gryszczynska A, Buchwald W (2013) Content of pyrrolizidine alkaloids in the leaves of coltsfoot (Tussilago farfara L.) in Poland. Acta Soc Botanicorum Poloniae 82(4)
Lebada R, Schreier A, Scherz S, Resch C, Krenn L, Kopp B (2000) Quantitative analysis of the pyrrolizidine alkaloids senkirkine and senecionine in Tussilago farfara L. by capillary electrophoresis. Phytochem Anal 11(6):366–369
Nedelcheva A, Kostova N, Sidjimov A (2015) Pyrrolizidine alkaloids in Tussilago farfara from Bulgaria. Biotechnol Biotechnol Equip 29(Sup1):S1–S7
Seremet OC, Olaru OT, Gutu CM, Nitulescu GM, Ilie M, Negres S, Margina DM (2018) Toxicity of plant extracts containing pyrrolizidine alkaloids using alternative invertebrate models. Mol Med Rep 17(6):7757–7763
Cheng X, Liao M, Diao X, Sun Y, Zhang L (2018) Screening and identification of metabolites of two kinds of main active ingredients and hepatotoxic pyrrolizidine alkaloids in rat after lavage Farfarae Flos extract by UHPLC-Q-TOF–MS mass spectrometry. Biomed Chromatogr 32(2):e4047
Wissenschaftliche Prüfungsarbeit (2016) Pyrrolizidine alkaloids – Impact of the public statements made by EMA and national health authorities on the pharmaceutical industry. Master of Drug Regulatory Affairs. Universität Bonn, Katrin Lambrecht aus Berlin, Germany
Roeder E, Rengel B (1990) Pyrrolizidine alkaloids from Lithospermum erythrorhizon. Phytochemistry 29(2):690–693
BfR (2014) Determination of pyrrolizidine alkaloids (PA) in plant material by SPE-LC-MS/MS. Method Protocol BfR-PA-Tea-2.0. https://www.bfr.bund.de/cm/349/determination-of-pyrrolizidine-alkaloids-pa-in-plant-material.pdf
AOAC (2012) AOAC international guidelines for validation of botanical identification methods. J AOAC Int 96(1):269–272
Wawrosh C, Kopp B, Wiedenfeld H (2000) Permanent monitoring of pyrrolizidine alkaloid content in micropropagated Tussilago farfara L.: a tool to fulfill statutory demands for the quality of coltsfoot in Austria and Germany. Acta Hort 530:469–472
Hrynko I, Łozowicka B, Kaczyński P (2018) Liquid chromatographic MS/MS analysis of a large group of insecticides in honey by modified QuEChERS. Food Anal Methods 11(8):2307–2319
Mroczek T, Glowniak K, Wlaszczyk A (2002) Simultaneous determination of N-oxides and free bases of pyrrolizidine alkaloids by cation-exchange solid-phase extraction and ion-pair high-performance liquid chromatography. J Chromatogr A 949(1–2):249–262
Roeder E (1995) Medicinal plants in Europe containing pyrrolizidine alkaloids. Pharmazie 50(2):83–98
Jiang Z, Liu F, Goh JJL, Yu L, Li SFY, Ong ES, Ong CN (2009) Determination of senkirkine and senecionine in Tussilago farfara using microwave-assisted extraction and pressurized hot water extraction with liquid chromatography tandem mass spectrometry. Talanta 79(2):539–546
Crout DHG, Whitehous D (1977) Absolute configuration of 2,3-dihydroxy-3-methylpentanoic acid, an intermediate in the biosynthesis of isoleucine, and its identity with the esterifying acid of the pyrrolizidine alkaloid strigosine. J Chem Soc Perkin Trans 1:544–549
Roeder E (2000) Medicinal plants in China containing pyrrolizidine alkaloids. Pharmazie 55(10):711–726
Acknowledgements
This research was supported by a Grant (19171MFDS193) from the Ministry of Food and Drug Safety in 2019.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
All authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ko, K.Y., Jeong, S.H., Choi, E.Y. et al. A LC–ESI–MS/MS analysis procedure coupled with solid phase extraction and MeOH extraction method for determination of pyrrolizidine alkaloids in Tussilago farfara and Lithospermi erythrorhzion. Appl Biol Chem 64, 53 (2021). https://doi.org/10.1186/s13765-021-00621-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13765-021-00621-6