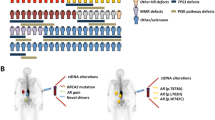
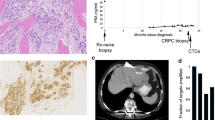
Abstract
Background
There are multiple existing and emerging therapeutic avenues for metastatic prostate cancer, with a common denominator, which is the need for predictive biomarkers. Circulating tumor DNA (ctDNA) has the potential to cost-efficiently accelerate precision medicine trials to improve clinical efficacy and diminish costs and toxicity. However, comprehensive ctDNA profiling in metastatic prostate cancer to date has been limited.
Methods
A combination of targeted and low-pass whole genome sequencing was performed on plasma cell-free DNA and matched white blood cell germline DNA in 364 blood samples from 217 metastatic prostate cancer patients.
Results
ctDNA was detected in 85.9% of baseline samples, correlated to line of therapy and was mirrored by circulating tumor cell enumeration of synchronous blood samples. Comprehensive profiling of the androgen receptor (AR) revealed a continuous increase in the fraction of patients with intra-AR structural variation, from 15.4% during first-line metastatic castration-resistant prostate cancer therapy to 45.2% in fourth line, indicating a continuous evolution of AR during the course of the disease. Patients displayed frequent alterations in DNA repair deficiency genes (18.0%). Additionally, the microsatellite instability phenotype was identified in 3.81% of eligible samples (≥ 0.1 ctDNA fraction). Sequencing of non-repetitive intronic and exonic regions of PTEN, RB1, and TP53 detected biallelic inactivation in 47.5%, 20.3%, and 44.1% of samples with ≥ 0.2 ctDNA fraction, respectively. Only one patient carried a clonal high-impact variant without a detectable second hit. Intronic high-impact structural variation was twice as common as exonic mutations in PTEN and RB1. Finally, 14.6% of patients presented false positive variants due to clonal hematopoiesis, commonly ignored in commercially available assays.
Conclusions
ctDNA profiles appear to mirror the genomic landscape of metastatic prostate cancer tissue and may cost-efficiently provide somatic information in clinical trials designed to identify predictive biomarkers. However, intronic sequencing of the interrogated tumor suppressors challenges the ubiquitous focus on coding regions and is vital, together with profiling of synchronous white blood cells, to minimize erroneous assignments which in turn may confound results and impede true associations in clinical trials.
Similar content being viewed by others
Background
Prostate cancer is the most commonly detected male cancer in Europe and the third major cause of cancer-related death among men [1]. Although the majority of metastatic hormone-naïve prostate cancers (mHNPCs) demonstrate a reliable response to initial androgen deprivation therapy which targets AR signaling, progression to a castration-resistant state is inevitable. However, the treatment landscape for metastatic castration-resistant prostate cancer (mCRPC) is evolving with the recent approval of several new drugs translating to an increased overall survival [2,3,4,5,6]. Multiple additional avenues exist as genomic profiling of metastatic tissue revealed that the majority of mCRPC patients harbor clinically relevant alterations beyond the AR signaling pathway [7].
The most promising non-approved treatment avenue in metastatic prostate cancers (mPCs) exploits synthetic lethality in treating homologous recombination-deficient cancers with poly (ADP-ribose) polymerase (PARP) inhibitors [8]. Approximately one fifth of mCRPC carry mutations in DNA repair genes [7]. However, the mutational signatures of biallelic inactivation are heterogeneous between different DNA repair genes [9], and future studies are therefore needed to determine which genes are associated with a response to PARP inhibition. Approximately 3% of mPC are driven by microsatellite instability (MSI) [7, 10]. Pembrolizumab recently became the first drug to be approved by the U.S. Food and Drug Administration based on the MSI phenotype, irrespective of tumor type [11]. Although checkpoint blockade did not confer a survival advantage as compared with placebo for chemotherapy-relapsed mCRPC [12], anecdotal cases have been reported to display partial or complete responses [10, 13,14,15].
The emergence of additional drugs, both towards common and rare mPC phenotypes such as PTEN-deficient [16, 17] and neuroendocrine cancers [18], raises questions of how to efficiently translate the multitude of treatment options to improved patient outcomes. The genomic heterogeneity of mCRPC [7] and, in turn, the low response rates of currently approved drugs [2,3,4,5, 19, 20] argue for the urgent need of predictive biomarkers. Ineffective trial-and-error decisions inevitably lead to unnecessary side effects and unsustainable costs [21]. The AR splice variant 7 (AR-V7) [22] demonstrated promising results as a negative response biomarker for androgen receptor signaling inhibitors. However, follow-up studies have been unable to validate the initial clear-cut finding [23, 24], and although AR-V7 is clearly prognostic, clinical implementation remains debated due to (1) lack of treatment options for AR-V7-positive patients and (2) lack of data from a prospective clinical trial demonstrating the predictive power of AR-V7 for treatment selection [25]. Recently, Scher and colleagues demonstrated increased survival for AR-V7-positive patients receiving taxanes in a blinded retrospective multicenter study [26]. However, AR-V7 assay positivity and taxane therapy were both correlated to tumor burden, obscuring interpretation and further highlighting the need for a prospective randomized clinical trial to determine if AR-V7 can be applied as a predictive biomarker [27].
The lack of predictive biomarkers is in part due to the difficulty of obtaining temporally matched metastatic tissue as the majority of mPCs metastasize to the bone. Multiple studies on the acquisition of tumor tissue with or without direct image guidance report a range of success rates [28,29,30,31]. A recent effort, focusing on methodological improvements, obtained > 20% cell content in the majority of bone biopsies [32]. Circulating tumor DNA is a viable alternative to metastatic tissue with demonstrated high fractions of ctDNA [33,34,35,36] enabling sensitive detection of somatic variation, and direct comparisons to metastatic tissue have revealed high concordance [33, 37, 38]. Circulating tumor DNA has several advantages as sampling through simple blood draws is fast, cost efficient, and without side effects and allows for longitudinal monitoring and the detection of multiple resistance alleles during therapy [38, 39].
Although ctDNA has the potential to accelerate biomarker-driven trials in mPC, several questions remain unanswered, e.g. if it is possible to detect MSI directly from liquid biopsies and how ctDNA fractions correlate to line of therapy. The ctDNA fraction determines the sensitivity to detect somatic variation which in turn has consequences for the design of prospective biomarker studies relying on liquid biopsies. Here, we present a retrospective analysis of 217 cases and 364 blood samples covering the entire spectrum of mPC. The purpose of this study was to gather information relevant for future liquid biopsy-driven biomarker studies with a focus on (1) how ctDNA fractions vary from mHNPC to end stage castration-resistant disease; (2) a rationale for how to treat samples with low ctDNA fraction; (3) the relative impact of different types of somatic variation, affecting the sequencing strategy; (4) the detection of potentially predictive biomarkers; (5) and finally, how clonal expansions in the hematopoietic stem cells [66]. A large fraction of the mutations was detected in the WBC DNA, corroborating our conclusions that to avoid false positive variant calls, sequencing of WBC DNA should be undertaken to the same or higher depth as the cell-free DNA. We therefore discourage the use of commercial assays that only analyze cfDNA from plasma.
Although multiple potentially predictive biomarkers have been reported for metastatic prostate cancer, no level 1 evidence currently exists from prospective randomized clinical trials. Based on our experience to date, we consider ctDNA profiling to be at Technology Readiness Level 7 according to the definition applied in the Horizon 2020 calls [67]. We have therefore initiated a prospective outcome-adaptive, multi-arm, open-label, multiple-assignment randomized biomarker-driven trial in patients with mCRPC where ctDNA profiling will be applied to identify somatic alterations (ProBio, EudraCT Number 2018-002350-78). The goal of the trial is to determine whether treatment choice based on a biomarker signature can improve progression-free survival compared to standard of care in patients with mCRPC and to evaluate the predictive capability of the investigated biomarker signatures.
Conclusions
This study strengthens the accumulating evidence that ctDNA profiling mirrors the somatic alteration landscape from metastatic tissue by demonstrating, for the first time, that the MSI phenotype may be detected directly from cell-free DNA. To enable acceleration of clinical trials through ctDNA analysis, intronic sequencing of tumor suppressors in combination with synchronous profiling of white blood cells must be applied to prevent inaccurate somatic variant calls, which in turn may reduce the power to identify predictive biomarkers.
Abbreviations
- AR:
-
Androgen receptor
- AR-V7:
-
AR splice variant 7
- cfDNA:
-
Cell-free DNA
- CNA:
-
Copy-number alteration
- CTC:
-
Circulating tumor cell
- ctDNA:
-
Circulating tumor DNA
- mCRPC:
-
Metastatic castration-resistant prostate cancer
- mHNPCs:
-
Metastatic hormone-naïve prostate cancers
- mPC:
-
Metastatic prostate cancer
- MSI:
-
Microsatellite instability
- PARP:
-
Poly (ADP-ribose) polymerase
- WBC:
-
White blood cell
References
Ferlay J, Steliarova-Foucher E, Lortet-Tieulent J, Rosso S, Coebergh JWW, Comber H, et al. Cancer incidence and mortality patterns in Europe: estimates for 40 countries in 2012. Eur J Cancer. 2013;49:1374–403.
Beer TM, Armstrong AJ, Rathkopf DE, Loriot Y, Sternberg CN, Higano CS, et al. Enzalutamide in metastatic prostate cancer before chemotherapy. N Engl J Med. 2014;371:424–33.
de Bono JS, Logothetis CJ, Molina A, Fizazi K, North S, Chu L, et al. Abiraterone and increased survival in metastatic prostate cancer. N Engl J Med. 2011;364:1995–2005.
Scher HI, Fizazi K, Saad F, Taplin M-E, Sternberg CN, Miller K, et al. Increased survival with enzalutamide in prostate cancer after chemotherapy. N Engl J Med. 2012;367:1187–97.
Ryan CJ, Smith MR, de Bono JS, Molina A, Logothetis CJ, de Souza P, et al. Abiraterone in metastatic prostate cancer without previous chemotherapy. N Engl J Med. 2013;368:138–48.
Parker C, Nilsson S, Heinrich D, Helle SI, O’Sullivan JM, Fosså SD, et al. Alpha emitter radium-223 and survival in metastatic prostate cancer. N Engl J Med. 2013;369:213–23.
Robinson D, Van Allen EM, Wu Y-M, Schultz N, Lonigro RJ, Mosquera J-M, et al. Integrative clinical genomics of advanced prostate cancer. Cell. 2015;161:1215–28.
Mateo J, Carreira S, Sandhu S, Miranda S, Mossop H, Perez-Lopez R, et al. DNA-repair defects and olaparib in metastatic prostate cancer. N Engl J Med. 2015;373:1697–708.
Polak P, Kim J, Braunstein LZ, Karlic R, Haradhavala NJ, Tiao G, et al. A mutational signature reveals alterations underlying deficient homologous recombination repair in breast cancer. Nat Genet. 2017;49:1476–86.
Zehir A, Benayed R, Shah RH, Syed A, Middha S, Kim HR, et al. Mutational landscape of metastatic cancer revealed from prospective clinical sequencing of 10,000 patients. Nat Med. 2017;23:703–13.
Commissioner O of T. Press Announcements - FDA approves first cancer treatment for any solid tumor with a specific genetic feature. Office of the Commissioner; [cited 2018 Jan 5]; Available from: https://www.fda.gov/newsevents/newsroom/pressannouncements/ucm560167.htm
Kwon ED, Drake CG, Scher HI, Fizazi K, Bossi A, van den Eertwegh AJM, et al. Ipilimumab versus placebo after radiotherapy in patients with metastatic castration-resistant prostate cancer that had progressed after docetaxel chemotherapy (CA184-043): a multicentre, randomised, double-blind, phase 3 trial. Lancet Oncol. 2014;15:700–12.
Cabel L, Loir E, Gravis G, Lavaud P, Massard C, Albiges L, et al. Long-term complete remission with Ipilimumab in metastatic castrate-resistant prostate cancer: case report of two patients. J Immunother Cancer. 2017;5:31.
Le DT, Durham JN, Smith KN, Wang H, Bartlett BR, Aulakh LK, et al. Mismatch repair deficiency predicts response of solid tumors to PD-1 blockade. Science. 2017;357:409–13.
Graff JN, Alumkal JJ, Drake CG, Thomas GV, Redmond WL, Farhad M, et al. Early evidence of anti-PD-1 activity in enzalutamide-resistant prostate cancer. Oncotarget. 2016;7:52810–7.
de Bono JS, De Giorgi U, Massard C, Bracarda S, Nava Rodrigues D, Kocak I, et al. PTEN loss as a predictive biomarker for the Akt inhibitor ipatasertib combined with abiraterone acetate in patients with metastatic castration-resistant prostate cancer (mCRPC). Ann Oncol [Internet]. Oxford University Press; 2016 [cited 2018 Jan 16];27. Available from: https://academic.oup.com/annonc/article/27/suppl_6/718O/2799456
Sumanasuriya S, De Bono J. Treatment of advanced prostate cancer - a review of current therapies and future promise. Cold Spring Harb Perspect Med [Internet]. 2017; Available from: https://doi.org/10.1101/cshperspect.a030635
Barbieri CE, Chinnaiyan AM, Lerner SP, Swanton C, Rubin MA. The emergence of precision urologic oncology: a collaborative review on biomarker-driven therapeutics. Eur Urol. 2017;71:237–46.
de Bono JS, Oudard S, Ozguroglu M, Hansen S, Machiels J-P, Kocak I, et al. Prednisone plus cabazitaxel or mitoxantrone for metastatic castration-resistant prostate cancer progressing after docetaxel treatment: a randomised open-label trial. Lancet. 2010;376:1147–54.
Petrylak DP, Tangen CM, Hussain MHA, Lara PN Jr, Jones JA, Taplin ME, et al. Docetaxel and estramustine compared with mitoxantrone and prednisone for advanced refractory prostate cancer. N Engl J Med. 2004;351:1513–20.
Mailankody S, Prasad V. Five years of cancer drug approvals: innovation, efficacy, and costs. JAMA Oncol. 2015;1:539–40.
Antonarakis ES, Lu C, Wang H, Luber B, Nakazawa M, Roeser JC, et al. AR-V7 and resistance to enzalutamide and abiraterone in prostate cancer. N Engl J Med. 2014;371:1028–38.
Antonarakis ES, Lu C, Luber B, Wang H, Chen Y, Zhu Y, et al. Clinical significance of androgen receptor splice variant-7 mRNA detection in circulating tumor cells of men with metastatic castration-resistant prostate cancer treated with first- and second-line abiraterone and enzalutamide. J Clin Oncol. 2017;35:2149–56.
De Laere B, Oeyen S, Mayrhofer M, Whitington T, van Dam P-J, Van Oyen P, et al. TP53 outperforms other androgen receptor biomarkers to predict abiraterone or enzalutamide outcome in metastatic castration-resistant prostate cancer. Clin Cancer Res [Internet]. 2018; Available from: https://doi.org/10.1158/1078-0432.CCR-18-1943
Steinestel J, Bernemann C, Schrader AJ, Lennerz JK. Re: Emmanuel S. Antonarakis, Changxue Lu, Brandon Luber, et al. Clinical significance of androgen receptor splice variant-7 mRNA detection in circulating tumor cells of men with metastatic castration-resistant prostate cancer treated with first- and second-line abiraterone and enzalutamide. J Clin Oncol 2017;35:2149-56: AR-V7 Testing: What’s in it for the Patient? Eur Urol. 2017;72:e168–e169.
Scher HI, Graf RP, Schreiber NA, Jayaram A, Winquist E, McLaughlin B, et al. Assessment of the validity of nuclear-localized androgen receptor splice variant 7 in circulating tumor cells as a predictive biomarker for castration-resistant prostate cancer. JAMA Oncol [Internet]. 2018; Available from: https://doi.org/10.1001/jamaoncol.2018.1621
Plymate SR, Sharp A, de Bono JS. Nuclear circulating tumor cell androgen receptor variant 7 in castration-resistant prostate cancer: the devil is in the detail. JAMA Oncol [Internet]. 2018; Available from: https://doi.org/10.1001/jamaoncol.2018.1615
Spritzer CE, Afonso PD, Vinson EN, Turnbull JD, Morris KK, Foye A, et al. Bone marrow biopsy: RNA isolation with expression profiling in men with metastatic castration-resistant prostate cancer--factors affecting diagnostic success. Radiology. 2013;269:816–23.
Ross RW, Halabi S, Ou S-S, Rajeshkumar BR, Woda BA, Vogelzang NJ, et al. Predictors of prostate cancer tissue acquisition by an undirected core bone marrow biopsy in metastatic castration-resistant prostate cancer--a Cancer and Leukemia Group B study. Clin Cancer Res. 2005;11:8109–13.
Lorente D, Omlin A, Zafeiriou Z, Nava-Rodrigues D, Pérez-López R, Pezaro C, et al. Castration-resistant prostate cancer tissue acquisition from bone metastases for molecular analyses. Clin Genitourin Cancer. 2016;14:485–93.
Holmes MG, Foss E, Joseph G, Foye A, Beckett B, Motamedi D, et al. CT-guided bone biopsies in metastatic castration-resistant prostate cancer: factors predictive of maximum tumor yield. J Vasc Interv Radiol. 2017;28:1073–81 e1.
Sailer V, Schiffman MH, Kossai M, Cyrta J, Beg S, Sullivan B, et al. Bone biopsy protocol for advanced prostate cancer in the era of precision medicine. Cancer [Internet]. 2017; Available from: https://doi.org/10.1002/cncr.31173
Ulz P, Belic J, Graf R, Auer M, Lafer I, Fischereder K, et al. Whole-genome plasma sequencing reveals focal amplifications as a driving force in metastatic prostate cancer. Nat Commun. 2016;7:12008.
Romanel A, Gasi Tandefelt D, Conteduca V, Jayaram A, Casiraghi N, Wetterskog D, et al. Plasma AR and abiraterone-resistant prostate cancer. Sci Transl Med. 2015;7:312re10.
Wyatt AW, Azad AA, Volik SV, Annala M, Beja K, McConeghy B, et al. Genomic alterations in cell-free DNA and enzalutamide resistance in castration-resistant prostate cancer. JAMA Oncol. 2016;2:1598–606.
De Laere B, van Dam P-J, Whitington T, Mayrhofer M, Diaz EH, Van den Eynden G, et al. Comprehensive profiling of the androgen receptor in liquid biopsies from castration-resistant prostate cancer reveals novel intra-AR structural variation and splice variant expression patterns. Eur Urol. 2017;72:192–200.
Wyatt AW, Annala M, Aggarwal R, Beja K, Feng F, Youngren J, et al. Concordance of circulating tumor DNA and matched metastatic tissue biopsy in prostate cancer. J Natl Cancer Inst [Internet]. 2017;109. Available from: https://doi.org/10.1093/jnci/djx118
Goodall J, Mateo J, Yuan W, Mossop H, Porta N, Miranda S, et al. Circulating cell-free DNA to guide prostate cancer treatment with PARP inhibition. Cancer Discov. 2017;7:1006–17.
Quigley D, Alumkal JJ, Wyatt AW, Kothari V, Foye A, Lloyd P, et al. Analysis of circulating cell-free DNA identifies multiclonal heterogeneity of BRCA2 reversion mutations associated with resistance to PARP inhibitors. Cancer Discov. 2017;7:999–1005.
**e M, Lu C, Wang J, McLellan MD, Johnson KJ, Wendl MC, et al. Age-related mutations associated with clonal hematopoietic expansion and malignancies. Nat Med. 2014;20:1472–8.
Genovese G, Kähler AK, Handsaker RE, Lindberg J, Rose SA, Bakhoum SF, et al. Clonal hematopoiesis and blood-cancer risk inferred from blood DNA sequence. N Engl J Med. 2014;371:2477–87.
Coombs CC, Zehir A, Devlin SM, Kishtagari A, Syed A, Jonsson P, et al. Therapy-related clonal hematopoiesis in patients with non-hematologic cancers is common and associated with adverse clinical outcomes. Cell Stem Cell. 2017;21:374–82 e4.
Phallen J, Sausen M, Adleff V, Leal A, Hruban C, White J, et al. Direct detection of early-stage cancers using circulating tumor DNA. Sci Transl Med [Internet]. 2017;9. Available from: https://doi.org/10.1126/scitranslmed.aan2415
Henao Diaz E, Yachnin J, Grönberg H, Lindberg J. The in vitro stability of circulating tumour DNA. PLoS One. 2016;11:e0168153.
De Laere B, Oeyen S, Van Oyen P, Ghysel C, Ampe J, Ost P, et al. Circulating tumor cells and survival in abiraterone- and enzalutamide-treated patients with castration-resistant prostate cancer. Prostate. 2018;78:435–45.
Team RC. R: a language and environment for statistical computing. Vienna: R Foundation for Statistical Computing; 2014. p. 2016.
Salipante SJ, Scroggins SM, Hampel HL, Turner EH, Pritchard CC. Microsatellite instability detection by next generation sequencing. Clin Chem. 2014;60:1192–9.
Baca SC, Prandi D, Lawrence MS, Mosquera JM, Romanel A, Drier Y, et al. Punctuated evolution of prostate cancer genomes. Cell. 2013;153:666–77.
Annala M, Vandekerkhove G, Khalaf D, Taavitsainen S, Beja K, Warner EW, et al. Circulating tumor DNA genomics correlate with resistance to abiraterone and enzalutamide in prostate cancer. Cancer Discov [Internet]. 2018; Available from: https://doi.org/10.1158/2159-8290.CD-17-0937
Na R, Zheng SL, Han M, Yu H, Jiang D, Shah S, et al. Germline mutations in ATM and BRCA1/2 distinguish risk for lethal and indolent prostate cancer and are associated with early age at death. Eur Urol. 2017;71:740–7.
Pritchard CC, Mateo J, Walsh MF, De Sarkar N, Abida W, Beltran H, et al. Inherited DNA-repair gene mutations in men with metastatic prostate cancer. N Engl J Med. 2016;375:443–53.
Mandelker D, Zhang L, Kemel Y, Stadler ZK, Joseph V, Zehir A, et al. Mutation detection in patients with advanced cancer by universal sequencing of cancer-related genes in tumor and normal DNA vs guideline-based germline testing. JAMA. 2017;318:825–35.
Robinson JT, Thorvaldsdóttir H, Winckler W, Guttman M, Lander ES, Getz G, et al. Integrative genomics viewer. Nat Biotechnol. 2011;29:24–6.
Bozic I, Reiter JG, Allen B, Antal T, Chatterjee K, Shah P, et al. Evolutionary dynamics of cancer in response to targeted combination therapy. Elife. 2013;2:e00747.
Le Tourneau C, Delord J-P, Gonçalves A, Gavoille C, Dubot C, Isambert N, et al. Molecularly targeted therapy based on tumour molecular profiling versus conventional therapy for advanced cancer (SHIVA): a multicentre, open-label, proof-of-concept, randomised, controlled phase 2 trial. Lancet Oncol. 2015;16:1324–34.
Heller G, McCormack R, Kheoh T, Molina A, Smith MR, Dreicer R, et al. Circulating tumor cell number as a response measure of prolonged survival for metastatic castration-resistant prostate cancer: a comparison with prostate-specific antigen across five randomized phase III clinical trials. J Clin Oncol. 2018;36:572–80.
Mehra N, Dolling D, Sumanasuriya S, Christova R, Pope L, Carreira S, et al. Plasma cell-free DNA concentration and outcomes from taxane therapy in metastatic castration-resistant prostate cancer from two phase III trials (FIRSTANA and PROSELICA). Eur Urol. 2018;74:283–91.
Seed G, Yuan W, Mateo J, Carreira S, Bertan C, Lambros M, et al. Gene copy number estimation from targeted next-generation sequencing of prostate cancer biopsies: analytic validation and clinical qualification. Clin Cancer Res. 2017;23:6070–7.
Jiang R, Lu Y-T, Ho H, Li B, Chen J-F, Lin M, et al. A comparison of isolated circulating tumor cells and tissue biopsies using whole-genome sequencing in prostate cancer. Oncotarget. 2015;6:44781–93.
Lohr JG, Adalsteinsson VA, Cibulskis K, Choudhury AD, Rosenberg M, Cruz-Gordillo P, et al. Whole-exome sequencing of circulating tumor cells provides a window into metastatic prostate cancer. Nat Biotechnol. 2014;32:479–84.
FoundationACT [Internet]. Foundation Medicine. [cited 2018 Aug 8]. Available from: https://www.foundationmedicine.com/genomic-testing/foundation-act
Personal Genome Diagnostics | Pharma-Biotech Liquid Biopsy [Internet]. Personal Genome Diagnostics. [cited 2018 Aug 8]. Available from: http://www.personalgenome.com/pharma-biotech-companies/liquid-biopsy/
Guardant360 | Guardant Health [Internet]. Guardant Health. [cited 2018 Aug 8]. Available from: https://www.guardanthealth.com/guardant360/
Torga G, Pienta KJ. Patient-paired sample congruence between 2 commercial liquid biopsy tests. JAMA Oncol [Internet]. 2017; Available from: https://doi.org/10.1001/jamaoncol.2017.4027
Jones S, Anagnostou V, Lytle K, Parpart-Li S, Nesselbush M, Riley DR, et al. Personalized genomic analyses for cancer mutation discovery and interpretation. Sci Transl Med. 2015;7:283ra53.
Hu Y, Ulrich BC, Supplee J, Kuang Y, Lizotte PH, Feeney NB, et al. False-positive plasma genoty** due to clonal hematopoiesis. Clin Cancer Res. 2018;24:4437–43.
European Commission. FAQ - Research Participant Portal [Internet]. [cited 2018 Aug 9]. Available from: https://ec.europa.eu/research/participants/portal/desktop/en/support/faqs/faq-2890.html
de Bono JS, Scher HI, Montgomery RB, Parker C, Miller MC, Tissing H, et al. Circulating tumor cells predict survival benefit from treatment in metastatic castration-resistant prostate cancer. Clin Cancer Res. 2008;14:6302–9.
Peeters DJE, De Laere B, Van den Eynden GG, Van Laere SJ, Rothé F, Ignatiadis M, et al. Semiautomated isolation and molecular characterisation of single or highly purified tumour cells from CellSearch enriched blood samples using dielectrophoretic cell sorting. Br J Cancer. 2013;108:1358–67.
Acknowledgements
The authors thank all patients for their willingness to participate in this study. We thank Luc De Laere, Thijs Develter, Sophie Vantieghem, Sofie Herman, Gwen Colfs, Veerle Lamotte, Anita Boumans, Abdelbari Baitar, Roos Haeck, Goele Wallays, Hanna Emanuelsson, Per Gustafsson, Jenny Wängberg, Maria Bergh, and Claudia Maes for their assistance with patient inclusion, sampling management, and data collection. We also thank Rebecka Bergström for excellent bioinformatic support.
Funding
The Belgian Foundation Against Cancer (grant number C/2014/227); Kom op tegen Kanker (Stand up to Cancer), the Flemish Cancer Society (grant number 00000000116000000206); Royal College of Surgeons/Cancer Research UK (C19198/A1533); The Cancer Research Funds of Radiumhemmet, through the PCM program at KI (grant number 163012); The Erling-Persson family foundation (grant number 4-2689-2016); the Swedish Research Council (grant number K2010-70X-20430-04-3), and the Swedish Cancer Foundation (grant number 09-0677). These funding bodies had no role in the design, execution, or interpretation of the data in this study and did not influence the decision to submit results for publication.
Availability of data and materials
All data relevant for the interpretation of our findings is provided in the main manuscript or the supplemental information except for the raw sequence data. Any data providing genotype information is considered to be a personal registry by the Swedish law (Personal Data Act), thereby prohibiting the submission to a public repository. The raw sequence data is instead available upon request from the authors (contact johan.lindberg@ki.se) if approval has been obtained from the Regional Ethical Vetting Board in Stockholm.
Author information
Authors and Affiliations
Contributions
BDL, PR, JL, and HG conceived the study and designed the methodologies and experiments. BDL, SO, JL, and GVdE performed experiments and acquired data. BDL, MM, TW, JL, and MR analysed and interpreted the data. PVO, CG, JA, PO, WD, LH, DS, BB, WL, EE, DDM, MS, KF, AR, AU, JY, HG, TN, MA, PR, and LD recruited patients and acquired blood samples for analysis. BDL, PR, JL, MM, and HG prepared the manuscript. BDL, PR, SO, MM, LE, TW, PJvD, PVO, CG, JA, PO, WD, LH, DS, BB, WL, EE, DDM, MS, KF, AB, DG, LH, GVdE, AR, MR, AU, JY, HG, SVL, JL, and LD performed critical revision of the manuscript for important intellectual content. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Ethical approval was obtained from ethical committees in Belgium (Antwerp University Hospital, registration number: B300201524217) and Sweden (Stockholm Regional Ethical Vetting Board registration numbers: 2016/101-32, amendment 2017/252-32; 2009/780-31/4, amendment 2014/1564-32). All patients provided a written informed consent document. The study was conducted in accordance with the Declaration of Helsinki.
Consent for publication
All patients additionally consented to have their data published in an anonymous format. Each patient and blood draw were assigned an untraceable number. The sample donor ID represents a unique patient and the sample ID a specific sampling occasion.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Additional files
Additional file 1:
Supplemental methods. (DOCX 21 kb)
Additional file 2:
Table S1. Detailed overview of the capture designs and targeted regions. (XLSX 974 kb)
Additional file 3:
Table S2. Summary of bioinformatic tools and settings. (XLSX 13 kb)
Additional file 4:
Table S3. Overview of which samples that were sequenced with which capture design. (XLSX 123 kb)
Additional file 5:
Table S4. Overview of the number of samples per described analysis. (XLSX 12 kb)
Additional file 6:
Figure S1. Circulating tumor DNA fraction in baseline samples. Figure S2. Tumor burden at different lines of therapy. Figure S3. Correlation between circulating tumor cell count and circulating tumor DNA fraction. Figure S4. Microsatellite instability by targeted sequencing of microsatellites. Figure S5. Chained structural event. Figure S6. Gene body panel design. Figure S7. Subclonal dynamics. Figure S8. Correlation between circulating tumor DNA fraction and cell-free DNA concentration. Figure S9. Baseline circulating tumor DNA fraction and circulating tumor cell counts at first- and second-line mCRPC treatment [68, 69]. Figure S10. False positive rate evaluation for point mutation variant calling. Figure S11. Allele frequencies of structural variants and mutations. (DOCX 3335 kb)
Additional file 7:
Table S5. CTC count, ctDNA fraction and line of therapy. (XLSX 27 kb)
Additional file 8:
Table S6. Somatic and germline alterations. (XLSX 141 kb)
Additional file 9:
Table S7. Table comparing relevant germline alterations, presented here, with other publications. (XLSX 26 kb)
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
About this article
Cite this article
Mayrhofer, M., De Laere, B., Whitington, T. et al. Cell-free DNA profiling of metastatic prostate cancer reveals microsatellite instability, structural rearrangements and clonal hematopoiesis. Genome Med 10, 85 (2018). https://doi.org/10.1186/s13073-018-0595-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13073-018-0595-5