Abstract
Background
Important roles of INHBB in various malignancies are increasingly identified. The underlying mechanisms in gastric cancer (GC) microenvironment are still greatly unexplored.
Methods
The clinical significance of INHBB and the correlation between INHBB and p-p65 in GC were assessed through analyzing publicly available databases and human paraffin embedded GC tissues. The biological crosstalk of INHBB between GC cells and fibroblasts was explored both in vitro and in vivo. RNA-seq analyses were performed to determine the mechanisms which regulating fibroblasts reprogramming. Luciferase reporter assay and chromatin immunoprecipitation (CHIP) assay were used to verify the binding relationship of p65 and INHBB in GC cells.
Results
Our study showed that INHBB level was significantly higher in GC, and that increased INHBB was associated with poor survival. INHBB positively regulates the proliferation, migration, and invasion of GC cells in vitro. Also, activin B promotes the occurrence of GC by reprogramming fibroblasts into cancer-associated fibroblasts (CAFs). The high expression of INHBB in GC cells activates the NF-κB pathway of normal gastric fibroblasts by secreting activin B, and promotes fibroblasts proliferation, migration, and invasion. In addition, activin B activates NF-κB pathway by controlling TRAF6 autoubiquitination to induce TAK1 phosphorylation in fibroblasts. Fibroblasts activated by activin B can induce the activation of p65 phosphorylation of GC cells by releasing pro-inflammatory factors IL-1β. p65 can directly bind to the INHBB promoter and increase the INHBB transcription of GC cells, thus establishing a positive regulatory feedback loop to promote the progression of GC.
Conclusions
GC cells p65/INHBB/activin B and fibroblasts p65/IL-1β signal loop led to the formation of a whole tumor-promoting inflammatory microenvironment, which might be a promising therapeutic target for GC.
Highlights
Tumor-secreted activin B promotes gastric cancer via fibroblast reprogramming.
Activin B activates the NF-κB pathway by inducing auto-ubiquitination of TRAF6 in fibroblasts.
Activin B stimulates the production of a pro-tumorigenic secretome and matrisome.
Activin B activates NF-κB pathway of fibroblasts to up-regulate IL-1β secretion.
IL-1β activates transcription factor p65 of gastric cancer to up-regulate INHBB expression.
Similar content being viewed by others
Introduction
Globally, gastric cancer (GC) ranks fifth among cancers in terms of diagnostic prevalence and third in terms of major causes of death [1, 2]. Exploring the important genes that affect cell survival in the genome regulatory network is crucial for transforming molecular characterization to useful clinical application. Because tumor tissue cells and their complex relationship with microenvironment affect all aspects of tumor generation and development, the role played by genes in regulating the phenotype and function of microenvironment is worth in-depth study [3, 4].
Activin B, a member of the TGF-β superfamily, is a dimer structure formed by the carboxyl terminal domain of the inhibin β subunit (INHBB) linked by a single disulfide bond [5]. Activin pathway is a well-studied pathway that is widely expressed in all stages of body growth, where it is recognized for its many essential roles in embryonic development, disease progression, and tissue homeostasis. Through series studies, Zhang et al. uncovered that activin B/Rho A/mDia1/Cdc42 axis plays a key function in bone marrow-derived mesenchymal stromal cells (BMSCs) migration by promoting membrane ruffling, microtubule morphology, and adhesion signaling dynamics. Inactivation of activin B-Cdc42 inhibits stimulation of Golgi polarization and adipose-derived mesenchymal stem cells-mediated skin wound healing [6,7,8]. Furthermore, overexpressing of activin B was identified in three different models of kidney fibrosis, as well as in human kidneys with fibrosis. Sox9-activin B provides a prospective entry point to surmount kidney fibrosis [9, 10]. Nevertheless, there is still room for improvement in understanding its specific value in tumor research [8A-B, S10A). Conversely, mice bearing INHBB-overexpressing cells had significantly larger tumors compared to mice bearing control cells (Fig. 8C-D, S10B). We verified the expression of INHBB in mice xenografts. (Fig. S10C). Furthermore, our results showed that in INHBB-derived overexpressing tumors, the areas of collagen-positive or α-SMA-positive fibroblasts increased significantly, indicating a profibrotic fibroblast phenotype. Together with prophase data, the number of Vimentin, IL-1β, and p-p65 positive GC cells was also higher in INHBB-overexpressing mice xenografts compared to that of the control groups (Fig. 8E). IHC statistical analyses were presented in Fig. S10D. We next evaluated the efficacy of IL-1b and JSH-23 in vivo. IL-1b (100 ng per mouse; in tumor; twice a week; for 2 weeks) significantly reversed the growth of subcutaneous INHBB-knockdown MGC-803 xenografts (Fig. 8F-G, S10E). Also, JSH-23 (1 mg/kg; orally administered; daily; for 2 weeks) significantly suppressed the growth of subcutaneous INHBB-overexpressing HGC-27 xenografts (Fig. 8H-I, S10F). IHC results showed that IL-1b promoted p65 phosphorylation and up-regulated INHBB expression in mice xenografts from INHBB-knockdown MGC-803 cells when compared to shNC cells. On the contrary, JSH-23 inhibition of p65 phosphorylation weakened the expression of INHBB in mice xenografts from INHBB-overexpressing HGC-27 cells (Fig. 8J). IHC statistical analyses were presented in Fig. S10G. The administration design is presented in Fig. S10H.
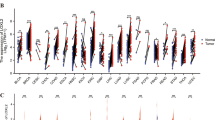
INHBB promotes GC progression in vivo. (A) The quantitative analysis of tumor weight from MGC-803 cells with stable INHBB knockdown or vehicle control (n = 4). (B) The growth curve of tumor volume from MGC-803 cells with stable INHBB knockdown or vehicle control (n = 4). (C) The quantitative analysis of tumor weight from HGC-27 cells with stable INHBB overexpressing or vehicle control (n = 5). (D) The growth curve of tumor volume from HGC-27 cells with stable INHBB overexpressing or vehicle control (n = 5). (E) The expression of collagen, a-SMA, Vimentin, IL-1B and p-p65 were evaluated by IHC in tissues of xenograft. (F) The quantitative analysis of tumor weight from MGC-803 shINHBB cells with IL-1b treatment or control treatment (n = 4). (G) The growth curve of tumor volume from MGC-803 shINHBB cells with IL-1b treatment or control treatment (n = 4). (H) The quantitative analysis of tumor weight from HGC-27 INHBB cells with JSH-23 treatment or control treatment (n = 5). (I) The growth curve of tumor volume from HGC-27 INHBB cells with JSH-23 treatment or control treatment (n = 5). (J) The expression of p-p65 and INHBB in tissues of xenograft. K. Diagrammatic presentation of the INHBB/NF-kB/IL-1b positive regulatory feedback loop between GC cells and fibroblasts that can promote GC progression. Activin B, the dimer structure formed by INHBB subunit, activates NF-kB pathway by controlling the autoubiquitination of TRAF6 and inducing TAK1 phosphorylation in fibroblasts. Activation of NF-kB releases IL-1b, which facilitates the phosphorylation of p65 and increases GC cells proliferation and invasion. Furthermore, p65 directly promotes INHBB transcription and induces INHBB expression, establishing a positive- feedback loop in GC microenvironment. *, P < 0.05; **, P < 0.01; ***, P < 0.001; ****, P < 0.0001
Discussion
Our study confirmed that INHBB was up-regulated in GC, which could promote GC cell proliferation, migration, and invasion. In addition, GC cells overexpressing INHBB increased the activity of NF-κB pathway in normal gastric fibroblasts by secreting activin B, and induced the reprogramming of fibroblasts. The activation of NF-κB leads to the release of inflammatory cytokine IL-1β, which leads to the structural activation of p65 in GC. Interestingly, we also found that p65 could cause an increase in INHBB transcriptional expression, which indicated that there is a positive feedback loop in GC. Our study showed that activin B is a major player in activating fibroblasts into pro-tumorigenic CAFs, and for the first time proposed an underlying molecular mechanism for INHBB in GC in combination with tumor microenvironment.
Changes in the function of cell components in the tumor microenvironment led to phenotypic variation and ultimately affect the tumor outcome. Fibrosis and inflammation are both microenvironment phenotypes that are closely related to tumor progression, thus promoting the continuous growth of the tumor [3]. The activation of resident normal fibroblasts mediates the transformation of cells to CAFs. An increase in proliferation and infiltration also involves the excessive deposition of ECM, thus providing a physical scaffold for the migration of tumor cells [34]. In the present study, we found that activin B can activate the NF-κB pathway of fibroblasts, promote the activation of fibroblasts, induce their proliferation, and up-regulate the migration and invasion phenotype. Simultaneously, it also induced the expression of CAF markers, including cytokines and ECM components. The activation of NF-κB plays an important role in the process of disease fibrosis. In chronic lung diseases, NF-κB can induce the process of pulmonary fibrosis by mediating the up-regulation of NLRP3 or NOX4 [35, 36]. In a model of renal interstitial fibrosis, NF-κB can induce EMT through the secretion of inflammatory cytokines [37]. In the present study, we found that the pro-inflammatory cytokine IL-1β showed a significantly increased production in normal gastric fibroblasts activated by activin B. Previous studies have shown that the inflammatory microenvironment is closely related to the occurrence and development of GC and the mutation of carcinogenic genes, where some inflammatory factors have been proved to be independent risk factors for GC [38]. Among these, IL-1β has been shown to increase the proliferation of GC cells through receptor-mediated tyrosine kinase pathway [39]. In the clinical setting, IL-1β polymorphisms were found to be closely associated with an increased risk of GC [40]. Therefore, the increase of IL-1β secretion of fibroblasts induced by NF-κB activation may promote the progression of GC.
As an important component of NF-κB transcription factor, p65 plays an important role in inflammation, immunity, cell proliferation and apoptosis. In addition, previous studies have shown that p65 is highly expressed in tumor tissues of patients with GC, which is positively correlated with higher degrees of tumor invasion, lymph node metastasis and distant metastasis [41]. In addition, overexpression of p-p65 in tumor tissue has been found to be associated with poor prognosis of GC patients [42]. As a part of the inflammatory tumor microenvironment, IL-1β can participate in the activation of NF-κB canonical pathway either directly (via p65) or in combination with other inflammatory factors, induce the positive feedback expression of various chemokines and inflammatory factors, and build a complex dynamic pro-tumor cycle [43]. In the present study, we found that IL-1β derived from gastric fibroblasts activated by activin B could activate the phosphorylation of p65 in GC cells under co-culture conditions. As a part of NF-κB transcription factor, p65 can directly bind to the INHBB promoter and promote the transcription of INHBB. Accordingly, we believe that the positive feedback loop between GC cell p65/INHBB/activin B and fibroblast p65/IL-1β controls the progression of GC. Furthermore, previous studies have shown that INHBA, which is homologous to INHBB in tumor cells, can partially mediate the activation of CAF in breast and ovarian tumor models. This activation is accomplished by INHBA-induced DNA damage or adrenergic signaling in tumor cells. [44,45,46]. Thus, our results brought corroborative evidence in the important role of INHBB as a CAF inducer.
In recent years, the microenvironment-targeted treatment of GC has begun to rise, including using various new drugs targeting FGFR [47], VEGFR [48, 49], MMP9 [50] and TGF-β1 [51] for resha** the matrix microenvironment [52]. Despite being a preliminary examination on showing the possible mechanism of INHBB’s impact on the microenvironment of GC, our study indicates that targeting fibroblast-cancer cell crosstalk is a promising alternative in GC clinical treatment by providing a well-explained possibility for the mechanism in place. GC progression is a huge and extremely complex interactive network, and there is still a long way to go to translate experimental research to clinical applications.
In conclusion, our research revealed that INHBB creates a local tumor-promoting inflammatory environment between GC cells and fibroblasts, thus providing new evidence for the role of INHBB in the occurrence and development of GC (Fig. 8K). We believe these findings have practical significance, which may deepen our understanding regarding the effects of INHBB in GC progression and treatment.
Data Availability
The datasets used/or analyzed during the study are available from the corresponding author on reasonable request.
Abbreviations
- GC:
-
gastric cancer
- CAFs:
-
cancer-associated fibroblasts
- INHBB:
-
inhibin β subunit
- ECM:
-
extracellular matrix
- IHC:
-
immunohistochemistry
- CHIP:
-
chromatin immunoprecipitation
- IP:
-
immunoprecipitation
- OS:
-
overall survival
- TCGA:
-
The Cancer Genome Atlas Database
- DFS:
-
disease-free survival
- EMT:
-
epithelial–mesenchymal transition
- MMP2:
-
matrix metalloproteinases 2
- α-SMA:
-
alpha-smooth muscle actin
- FAP:
-
fibroblast activation protein
- KEGG:
-
the Kyoto Encyclopedia of Genes and Genomes
- GO:
-
the gene ontology
- DEGs:
-
differential expression genes
- TRAF6:
-
tumor necrosis factor receptor-associated factor 6
- TAK1:
-
transforming growth factor β activated kinase 1
- MAPKKK:
-
mitogen-activated protein kinase kinase kinase
References
Ajani JA, et al. Gastric Cancer, Version 2.2022, NCCN Clinical Practice guidelines in Oncology. J Natl Compr Canc Netw. 2022;20:167–92. https://doi.org/10.6004/jnccn.2022.0008.
Harold KM, et al. Advances in imaging of inflammation, fibrosis, and Cancer in the gastrointestinal tract. Int J Mol Sci. 2022;23. https://doi.org/10.3390/ijms232416109.
Hanahan D, Coussens LM. Accessories to the crime: functions of cells recruited to the Tumor microenvironment. Cancer Cell. 2012;21:309–22. https://doi.org/10.1016/j.ccr.2012.02.022.
Gwee YX, et al. Integration of genomic Biology Into therapeutic strategies of gastric Cancer peritoneal Metastasis. J Clin Oncol. 2022;40:2830. https://doi.org/10.1200/JCO.21.02745.
Lodberg A. Principles of the activin receptor signaling pathway and its inhibition. Cytokine Growth Factor Rev. 2021;60:1–17. https://doi.org/10.1016/j.cytogfr.2021.04.001.
Wang X, et al. mDia1 and Cdc42 regulate activin B-Induced Migration of Bone Marrow-derived mesenchymal stromal cells. Stem Cells. 2019;37:150–62. https://doi.org/10.1002/stem.2924.
Wang X, et al. RhoA regulates activin B-induced stress fiber formation and migration of bone marrow-derived mesenchymal stromal cell through distinct signaling. Biochim Biophys Acta Gen Subj. 2017;1861:3011–8. https://doi.org/10.1016/j.bbagen.2016.09.027.
Huang S, et al. Activin B-activated Cdc42 signaling plays a key role in regulating adipose-derived mesenchymal stem cells-mediated skin wound healing. Stem Cell Res Ther. 2022;13:248. https://doi.org/10.1186/s13287-022-02918-9.
Krepinsky JC, Activin B. A new player in kidney fibrosis?(dagger). J Pathol. 2022;256:363–5. https://doi.org/10.1002/path.5847.
Sun Y, et al. Tubule-derived INHBB promotes interstitial fibroblast activation and renal fibrosis. J Pathol. 2022;256:25–37. https://doi.org/10.1002/path.5798.
**a Y, Schneyer AL. The biology of activin: recent advances in structure, regulation and function. J Endocrinol. 2009;202:1–12. https://doi.org/10.1677/JOE-08-0549.
Chen Y, et al. Sox9/INHBB axis-mediated crosstalk between the hepatoma and hepatic stellate cells promotes the Metastasis of hepatocellular carcinoma. Cancer Lett. 2021;499:243–54. https://doi.org/10.1016/j.canlet.2020.11.025.
Qian L, Lai X, Gu B, Sun X. An Immune-related gene signature for Predicting Neoadjuvant Chemoradiotherapy efficacy in rectal carcinoma. Front Immunol. 2022;13:784479. https://doi.org/10.3389/fimmu.2022.784479.
Reader KL, et al. Activin B and activin C have Opposing effects on Prostate Cancer progression and cell growth. Cancers (Basel). 2022;15. https://doi.org/10.3390/cancers15010147.
Yu W, et al. INHBB is a novel prognostic biomarker and correlated with immune infiltrates in gastric cancer. Front Genet. 2022;13:933862. https://doi.org/10.3389/fgene.2022.933862.
Kalluri R. The biology and function of fibroblasts in cancer. Nat Rev Cancer. 2016;16:582–98. https://doi.org/10.1038/nrc.2016.73.
Madar S, Goldstein I, Rotter V. Cancer associated fibroblasts’--more than meets the eye. Trends Mol Med. 2013;19:447–53. https://doi.org/10.1016/j.molmed.2013.05.004.
Gascard P, Tlsty TD. Carcinoma-associated fibroblasts: orchestrating the composition of malignancy. Genes Dev. 2016;30:1002–19. https://doi.org/10.1101/gad.279737.116.
Wang Y, et al. Activin B promotes the initiation and progression of liver fibrosis. Hepatol Commun. 2022;6:2812–26. https://doi.org/10.1002/hep4.2037.
Hedger MP, de Kretser DM. The activins and their binding protein, follistatin-diagnostic and therapeutic targets in inflammatory Disease and fibrosis. Cytokine Growth Factor Rev. 2013;24:285–95. https://doi.org/10.1016/j.cytogfr.2013.03.003.
Shao X, et al. MatrisomeDB 2.0: 2023 updates to the ECM-protein knowledge database. Nucleic Acids Res. 2023;51:D1519–30. https://doi.org/10.1093/nar/gkac1009.
Landstrom M. The TAK1-TRAF6 signalling pathway. Int J Biochem Cell Biol. 2010;42:585–9. https://doi.org/10.1016/j.biocel.2009.12.023.
Min Y, Kim MJ, Lee S, Chun E, Lee KY. Inhibition of TRAF6 ubiquitin-ligase activity by PRDX1 leads to inhibition of NFKB activation and autophagy activation. Autophagy. 2018;14:1347–58. https://doi.org/10.1080/15548627.2018.1474995.
Ji YX, et al. The ubiquitin E3 ligase TRAF6 exacerbates pathological cardiac hypertrophy via TAK1-dependent signalling. Nat Commun. 2016;7:11267. https://doi.org/10.1038/ncomms11267.
Singh AK, et al. Suppression of monosodium urate crystal-induced inflammation by inhibiting TGF-beta-activated kinase 1-dependent signaling: role of the ubiquitin proteasome system. Cell Mol Immunol. 2021;18:162–70. https://doi.org/10.1038/s41423-019-0284-3.
Inman GJ, et al. SB-431542 is a potent and specific inhibitor of transforming growth factor-beta superfamily type I activin receptor-like kinase (ALK) receptors ALK4, ALK5, and ALK7. Mol Pharmacol. 2002;62:65–74. https://doi.org/10.1124/mol.62.1.65.
Luo H, et al. Pan-cancer single-cell analysis reveals the heterogeneity and plasticity of cancer-associated fibroblasts in the Tumor microenvironment. Nat Commun. 2022;13:6619. https://doi.org/10.1038/s41467-022-34395-2.
Tu S, et al. Overexpression of interleukin-1beta induces gastric inflammation and cancer and mobilizes myeloid-derived suppressor cells in mice. Cancer Cell. 2008;14:408–19. https://doi.org/10.1016/j.ccr.2008.10.011.
Cao L, et al. Helicobacter pylori-induced RASAL2 through activation of Nuclear factor-kappab promotes gastric tumorigenesis via beta-catenin Signaling Axis. Gastroenterology. 2022;162:1716–1731e1717. https://doi.org/10.1053/j.gastro.2022.01.046.
Zhang P, et al. Intratumoral IL-1R1 expression delineates a distinctive molecular subset with therapeutic resistance in patients with gastric cancer. J Immunother Cancer. 2022;10. https://doi.org/10.1136/jitc-2021-004047.
Yin S, Lan C, Pei H, Zhu Z. Expression of interleukin 1beta in gastric cancer tissue and its effects on gastric cancer. Onco Targets Ther. 2016;9:31–5. https://doi.org/10.2147/OTT.S94277.
Diep S, Maddukuri M, Yamauchi S, Geshow G, Delk NA. Interleukin-1 and nuclear factor Kappa B Signaling promote Breast Cancer Progression and Treatment Resistance. Cells. 2022;11. https://doi.org/10.3390/cells11101673.
Sen R, Smale ST. Selectivity of the NF-kappaB response. Cold Spring Harb Perspect Biol. 2010;2:a000257. https://doi.org/10.1101/cshperspect.a000257.
Kalluri R, Zeisberg M. Fibroblasts in cancer. Nat Rev Cancer. 2006;6:392–401. https://doi.org/10.1038/nrc1877.
Peng L, et al. Scutellarin ameliorates pulmonary fibrosis through inhibiting NF-kappaB/NLRP3-mediated epithelial-mesenchymal transition and inflammation. Cell Death Dis. 2020;11:978. https://doi.org/10.1038/s41419-020-03178-2.
Park SH, et al. PM014 attenuates radiation-induced pulmonary fibrosis via regulating NF-kB and TGF-b1/NOX4 pathways. Sci Rep. 2020;10:16112. https://doi.org/10.1038/s41598-020-72629-9.
Li R, et al. Salidroside ameliorates renal interstitial fibrosis by inhibiting the TLR4/NF-kappaB and MAPK signaling pathways. Int J Mol Sci. 2019;20. https://doi.org/10.3390/ijms20051103.
Tsujimoto H, et al. Roles of inflammatory cytokines in the progression of gastric cancer: friends or foes? Gastric Cancer. 2010;13:212–21. https://doi.org/10.1007/s10120-010-0568-x.
Beales IL. Effect of interlukin-1beta on proliferation of gastric epithelial cells in culture. BMC Gastroenterol. 2002;2. https://doi.org/10.1186/1471-230x-2-7.
El-Omar EM, et al. Interleukin-1 polymorphisms associated with increased risk of gastric cancer. Nature. 2000;404:398–402. https://doi.org/10.1038/35006081.
Balcerczak E, et al. Expression of the P65 gene in gastric cancer and in tissues with or without Helicobacter pylori Infection. Neoplasma. 2005;52:464–8.
Fu ZH, et al. NIK– and IKKbeta–binding protein contributes to gastric cancer chemoresistance by promoting epithelial–mesenchymal transition through the NF–kappaB signaling pathway. Oncol Rep. 2018;39:2721–30. https://doi.org/10.3892/or.2018.6348.
Ben-Baruch A. Partners in crime: TNFalpha-based networks promoting cancer progression. Cancer Immunol Immunother. 2020;69:263–73. https://doi.org/10.1007/s00262-019-02435-4.
Fordyce CA, et al. Cell-extrinsic consequences of epithelial stress: activation of protumorigenic tissue phenotypes. Breast Cancer Res. 2012;14:R155. https://doi.org/10.1186/bcr3368.
Nagaraja AS, et al. Adrenergic-mediated increases in INHBA drive CAF phenotype and collagens. JCI Insight. 2017;2. https://doi.org/10.1172/jci.insight.93076.
Cangkrama M, et al. A paracrine activin A-mDia2 axis promotes squamous carcinogenesis via fibroblast reprogramming. EMBO Mol Med. 2020;12:e11466. https://doi.org/10.15252/emmm.201911466.
Kim SB, et al. First-in-human phase I study of Aprutumab Ixadotin, a fibroblast growth factor receptor 2 antibody-drug Conjugate (BAY 1187982) in patients with Advanced Cancer. Target Oncol. 2019;14:591–601. https://doi.org/10.1007/s11523-019-00670-4.
Kobayshi K, et al. A phase II multicenter trial assessing the efficacy and safety of first-line S-1 + ramucirumab in elderly patients with advanced/recurrent gastric cancer: KSCC1701. Eur J Cancer. 2022;166:279–86. https://doi.org/10.1016/j.ejca.2022.02.028.
Bang YJ, et al. Ramucirumab and durvalumab for previously treated, advanced non-small-cell Lung cancer, gastric/gastro-oesophageal junction adenocarcinoma, or hepatocellular carcinoma: an open-label, phase Ia/b study (JVDJ). Eur J Cancer. 2020;137:272–84. https://doi.org/10.1016/j.ejca.2020.06.007.
Shah MA, et al. Phase III study to Evaluate Efficacy and Safety of Andecaliximab with mFOLFOX6 as first-line treatment in patients with Advanced gastric or GEJ Adenocarcinoma (GAMMA-1). J Clin Oncol. 2021;39:990–1000. https://doi.org/10.1200/JCO.20.02755.
Lan Y, et al. Enhanced preclinical antitumor activity of M7824, a bifunctional fusion protein simultaneously targeting PD-L1 and TGF-beta. Sci Transl Med. 2018;10. https://doi.org/10.1126/scitranslmed.aan5488.
Li K, Zhang A, Li X, Zhang H, Zhao L. Advances in clinical immunotherapy for gastric cancer. Biochim Biophys Acta Rev Cancer. 2021;1876:188615. https://doi.org/10.1016/j.bbcan.2021.188615.
Acknowledgements
Not applicable.
Funding
The study was supported by National Science Foundation of China (82273126 and 81972707) and the Collaborative Innovation Cluster Project of Shanghai Municipal Health Commission (2020CXJQ03) and Innovative research team of high-level local universities in Shanghai.
Author information
Authors and Affiliations
Contributions
YJ, BY and JZ, designed and performed the experiments; LW, provided the human samples; YJ, QC and LW, wrote the manuscript; JJ, CW, JW and FQ, conceived the project and critically revised the manuscript; YS, JJ, BZ and LZ, analyzed the data. All the authors approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Ethics approval
The study was approved by the Ethics Committee of the Rui** Hospital, Shanghai Jiao Tong University School of Medicine.
Consent for publication
The authors declare that they agree to submit the article for publication.
Competing interests
The authors have declared that no competing interest exists.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
**, Y., Cai, Q., Wang, L. et al. Paracrine activin B-NF-κB signaling shapes an inflammatory tumor microenvironment in gastric cancer via fibroblast reprogramming. J Exp Clin Cancer Res 42, 269 (2023). https://doi.org/10.1186/s13046-023-02861-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13046-023-02861-4