Abstract
Background
Leptin is important in physiological and pathological functions in various cancers, however, the significance and mechanisms of leptin in nasopharyngeal carcinoma remain ambiguous.
Methods
Leptin expression was analyzed by QPCR, immunohistochemistry, Western blotting, and TCGA database. The impact of gain- or loss-of-function of leptin were determined by MTT, colony formation, wound healing, and Transwell assays in NPC cells, and by a xenograft tumor model. Leptin-modulated glucose consumption and lactate production were assessed by ELISA. Furthermore, leptin-regulated signaling pathways were examined by QPCR and Western blotting assays. The immunoprecipitation assay was conducted to determine interaction between leptin and EGFR. In addition, miR-874-3p-regulated leptin expression was evaluated using bioinformatics, QPCR, luciferase assay, AGO2-RIP assay, and Western blotting.
Results
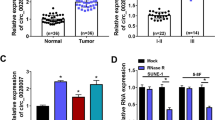
In this study, we found that leptin was highly expressed in the sera and tumor tissues of patients with NPC, and elevated leptin expression was associated with advanced clinical features and poor prognosis. Functional assays demonstrated that leptin remarkably promoted NPC cell growth, motility, and glycolysis in vitro and in vivo. Mechanistically, leptin associated with EGFR, resulting in enhanced cell growth through the regulation of cell-cycle related markers, glycolysis-related genes, and EGFR/AKT/c-Myc signaling. Moreover, leptin potentiated the invasive capacity of NPC cells by promoting EMT. We further explored that miR-874-3p influenced leptin-mediated NPC progression. Overexpression of miR-874-3p prevented cell growth, motility, glucose consumption, and lactate production in NPC cells, whereas miR-874-3p inhibition had the opposite effects. AGO-RIP assays confirmed that Argonaute 2 (AGO2), a protein associated with miR-874-3p, regulated leptin expression in NPC cells. The rescue assays indicated that inhibition of leptin suppressed the effects of miR-874-3p inhibitor. In clinical specimens, miR-874-3p was negatively correlated with leptin.
Conclusions
Leptin may serve as a novel prognostic factor and potential therapeutic target for patients with NPC. In addition, a newly discovered regulatory axis of leptin/EGFR/AKT/c-Myc can provide a novel therapeutic strategy for NPC.
Similar content being viewed by others
Background
Nasopharyngeal carcinoma (NPC) is a malignant tumor involving the head and neck areas. Unlike other head and neck cancers, NPC is characterized by increased invasiveness and metastasis [1]. Survival outcomes are significantly better in patients with early-stage NPC than in those with late-stage NPC [1, 2]. Although NPC is rare in most parts of the world, it has a high regional incidence in Asia, with that in Southern China and Southeast Asia ranking among the highest in the world [2]. Generally, there are three main types of NPC according to the World Health Organization (WHO), which include keratinizing squamous cell carcinoma, non-keratinizing carcinoma and undifferentiated carcinoma [3]. Epstein-Barr virus (EBV) has been shown to be a risk factor for NPC, especially for non-keratinizing and undifferentiated carcinoma [3,4,5]. The earliest evidence of NPC associated with EBV was identified in 1973 [6]. Although EBV is detected in most patients with NPC, cases of EBV-negative NPC have also been reported [4, 7]. Other lifestyle-associated risk factors and/or host genetic variants may interact with EBV to play roles in the carcinogenesis of NPC [5, 8, 9]. Obesity can occur due to unhealthy lifestyle habits and has been associated with an increased risk for various cancers, including breast, esophageal, pancreatic, and colorectal cancers [10,11,12,13]. Interestingly, several epidemiological studies have shown that patients with NPC have higher BMI, implying that overweight or obese people are tended to have a greater risk of NPC [14, In summary, we conducted a study to understand the effects of leptin on NPC by combining a small-scale clinical analysis, in vitro, in vivo and in silico methods. Our small-scale clinical findings indicated that elevated intra-tumoral leptin was associated with more advanced tumors and poor prognosis of NPC. Our data also suggested that leptin was involved in alteration of diverse molecules related to pathways that regulate EMT and cell proliferation. These results supported the idea that leptin facilitates nasopharyngeal cell survival, proliferation and invasion by influencing the expression levels of EMT-, cell proliferation- and glycolysis-related proteins, which are key to tumor invasiveness and metastasis and the shortened survival of patients with cancer. Similarly, these results may also explain the findings of our clinical research, as well as our mouse model of tumor growth, considering the cancer-promoting effects of leptin in our assays. Furthermore, we utilized in silico miRNA-target prediction to identify miR-874-3p as a novel regulator of leptin and rigorously validated this prediction experimentally. Our data suggests that miR-874-3p targets leptin mRNA to reduce cell survival and proliferation in NPC cells. Overall, this study provides insights into how leptin is involved in the carcinogenesis of NPC with a focus on potential pathological effects of leptin and therapeutic targeting of leptin expression, which may potentially be applied in future clinical settings.Conclusions
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Abbreviations
- NPC:
-
Nasopharyngeal carcinoma
- EBV:
-
Epstein-Barr virus
- AJCC:
-
American Joint Committee on Cancer
- EMT:
-
Epithelial-mesenchymal transition
- QPCR:
-
Quantitative polymerase chain reaction
- TCGA:
-
The Cancer Genome Atlas
References
Luo SD, Chen WC, Wu CN, Yang YH, Li SH, Fang FM, Huang TL, Wang YM, Chiu TJ, Wu SC: Low-dose aspirin use significantly improves the survival of late-stage NPC: a propensity score-matched cohort study in taiwan. Cancers (Basel). 2020;12(6):1551.
Mahdavifar N, Towhidi F, Makhsosi BR, Pakzad R, Moini A, Ahmadi A, Lotfi S, Salehiniya H. Incidence and mortality of nasopharynx cancer and its relationship with human development index in the world in 2012. World J Oncol. 2016;7:109–18.
Lo EJ, Bell D, Woo J, Li G, Hanna EY, El-Naggar AK, Sturgis EM. Human papillomavirus & WHO type I nasopharyngeal carcinoma. Laryngoscope. 2010;120(Suppl 4):S185.
Niedobitek G. Epstein-Barr virus infection in the pathogenesis of nasopharyngeal carcinoma. Mol Pathol. 2000;53:248–54.
Tsao SW, Tsang CM, Lo KW: Epstein-Barr virus infection and nasopharyngeal carcinoma. Philos Trans R Soc Lond B Biol Sci. 2017;372(1732):20160270.
Wolf H, zur Hausen H, Becker V. EB viral genomes in epithelial nasopharyngeal carcinoma cells. Nat New Biol. 1973;244:245–7.
Lee HS, Chang MS, Yang HK, Lee BL, Kim WH. Epstein-barr virus-positive gastric carcinoma has a distinct protein expression profile in comparison with epstein-barr virus-negative carcinoma. Clin Cancer Res. 2004;10:1698–705.
Okekpa SI, Rabiatul Basria S M N Mydin, Mangantig E, Azmi NSA, Zahari SNS, Kaur G, Musa Y. Nasopharyngeal Carcinoma (NPC) risk factors: a systematic review and meta-analysis of the association with lifestyle, diets, socioeconomic and sociodemographic in Asian region. Asian Pac J Cancer Prev. 2019;20:3505–14.
Yu G, Hsu WL, Coghill AE, Yu KJ, Wang CP, Lou PJ, Liu Z, Jones K, Vogt A, Wang M, et al. Whole-exome sequencing of nasopharyngeal carcinoma families reveals novel variants potentially involved in nasopharyngeal carcinoma. Sci Rep. 2019;9:9916.
Renehan AG, Tyson M, Egger M, Heller RF, Zwahlen M. Body-mass index and incidence of cancer: a systematic review and meta-analysis of prospective observational studies. Lancet. 2008;371:569–78.
Hoyo C, Cook MB, Kamangar F, Freedman ND, Whiteman DC, Bernstein L, Brown LM, Risch HA, Ye W, Sharp L, et al. Body mass index in relation to oesophageal and oesophagogastric junction adenocarcinomas: a pooled analysis from the International BEACON Consortium. Int J Epidemiol. 2012;41:1706–18.
Genkinger JM, Spiegelman D, Anderson KE, Bernstein L, van den Brandt PA, Calle EE, English DR, Folsom AR, Freudenheim JL, Fuchs CS, et al. A pooled analysis of 14 cohort studies of anthropometric factors and pancreatic cancer risk. Int J Cancer. 2011;129:1708–17.
Ma Y, Yang Y, Wang F, Zhang P, Shi C, Zou Y, Qin H. Obesity and risk of colorectal cancer: a systematic review of prospective studies. PLoS One. 2013;8: e53916.
Liu YT, Fan YY, Xu CH, Lin XL, Lu YK, Zhang XL, Zhang CX, Chen YM. Habitual consumption of soy products and risk of nasopharyngeal carcinoma in Chinese adults: a case-control study. PLoS One. 2013;8: e77822.
Feng R, Chang ET, Liu Z, Liu Q, Cai Y, Zhang Z, Chen G, Huang QH, **e SH, Cao SM, et al. Body mass index, body shape, and risk of nasopharyngeal carcinoma: a population-based case-control study in Southern China. Cancer Med. 2019;8:1835–44.
Liu YT, Dai JJ, Xu CH, Lu YK, Fan YY, Zhang XL, Zhang CX, Chen YM. Greater intake of fruit and vegetables is associated with lower risk of nasopharyngeal carcinoma in Chinese adults: a case-control study. Cancer Causes Control. 2012;23:589–99.
Considine RV, Sinha MK, Heiman ML, Kriauciunas A, Stephens TW, Nyce MR, Ohannesian JP, Marco CC, McKee LJ, Bauer TL, et al. Serum immunoreactive-leptin concentrations in normal-weight and obese humans. N Engl J Med. 1996;334:292–5.
Wauman J, Zabeau L, Tavernier J. The leptin receptor complex: heavier than expected? Front Endocrinol (Lausanne). 2017;8:30.
Ando S, Barone I, Giordano C, Bonofiglio D, Catalano S. The multifaceted mechanism of leptin signaling within tumor microenvironment in driving breast cancer growth and progression. Front Oncol. 2014;4:340.
da Rocha RG, Santos EMS, Santos EM, Gomes ESB, Ramos GV, Aguiar KM, Goncalves BR, Santos SHS, De Paula AMB, Guimaraes ALS, Farias LC. Leptin impairs the therapeutic effect of ionizing radiation in oral squamous cell carcinoma cells. J Oral Pathol Med. 2019;48:17–23.
Harbuzariu A, Rampoldi A, Daley-Brown DS, Candelaria P, Harmon TL, Lipsey CC, Beech DJ, Quarshie A, Ilies GO, Gonzalez-Perez RR. Leptin-Notch signaling axis is involved in pancreatic cancer progression. Oncotarget. 2017;8:7740–52.
Sengupta S, den Boon JA, Chen IH, Newton MA, Dahl DB, Chen M, Cheng YJ, Westra WH, Chen CJ, Hildesheim A, et al. Genome-wide expression profiling reveals EBV-associated inhibition of MHC class I expression in nasopharyngeal carcinoma. Cancer Res. 2006;66:7999–8006.
Peng Y, Croce CM. The role of MicroRNAs in human cancer. Signal Transduct Target Ther. 2016;1:15004.
Si W, Shen J, Zheng H, Fan W. The role and mechanisms of action of microRNAs in cancer drug resistance. Clin Epigenetics. 2019;11:25.
Tan G, Tang X, Tang F. The role of microRNAs in nasopharyngeal carcinoma. Tumour Biol. 2015;36:69–79.
Liu X, Lv XB, Wang XP, Sang Y, Xu S, Hu K, Wu M, Liang Y, Liu P, Tang J, et al. MiR-138 suppressed nasopharyngeal carcinoma growth and tumorigenesis by targeting the CCND1 oncogene. Cell Cycle. 2012;11:2495–506.
Zhu HM, Jiang XS, Li HZ, Qian LX, Du MY, Lu ZW, Wu J, Tian XK, Fei Q, He X, Yin L. miR-184 inhibits tumor invasion, migration and metastasis in nasopharyngeal carcinoma by targeting notch2. Cell Physiol Biochem. 2018;49:1564–76.
Li M, Huang H, Cheng F, Hu X, Liu J. miR-141-3p promotes proliferation and metastasis of nasopharyngeal carcinoma by targeting NME1. Adv Med Sci. 2020;65:252–8.
**a B, Lin M, Dong W, Chen H, Li B, Zhang X, Hou Y, Lou G. Upregulation of miR-874-3p and miR-874-5p inhibits epithelial ovarian cancer malignancy via SIK2. J Biochem Mol Toxicol. 2018;32: e22168.
Yuan RB, Zhang SH, He Y, Zhang XY, Zhang YB. MiR-874-3p is an independent prognostic factor and functions as an anti-oncomir in esophageal squamous cell carcinoma via targeting STAT3. Eur Rev Med Pharmacol Sci. 2018;22:7265–73.
Jiang T, Guan LY, Ye YS, Liu HY, Li R. MiR-874 inhibits metastasis and epithelial-mesenchymal transition in hepatocellular carcinoma by targeting SOX12. Am J Cancer Res. 2017;7:1310–21.
Sellam LS, Zappasodi R, Chettibi F, Djennaoui D, Yahi-AitMesbah N, Amir-Tidadini ZC, Touil-Boukoffa C, Ouahioune W, Merghoub T, Bourouba M. Silibinin down-regulates PD-L1 expression in nasopharyngeal carcinoma by interfering with tumor cell glycolytic metabolism. Arch Biochem Biophys. 2020;690: 108479.
Shan G, Tang T. Expression of cyclin D1 and cyclin E in urothelial bladder carcinoma detected in tissue chips using a quantum dot immunofluorescence technique. Oncol Lett. 2015;10:1271–6.
Qu C, Yan C, Cao W, Li F, Qu Y, Guan K, Si C, Yu Z, Qu Z. miR-128-3p contributes to mitochondrial dysfunction and induces apoptosis in glioma cells via targeting pyruvate dehydrogenase kinase 1. IUBMB Life. 2020;72:465–75.
Zeng FF, Xu CH, Liu YT, Fan YY, Lin XL, Lu YK, Zhang CX, Chen YM. Choline and betaine intakes are associated with reduced risk of nasopharyngeal carcinoma in adults: a case-control study. Br J Cancer. 2014;110:808–16.
Wang C, Lin XL, Fan YY, Liu YT, Zhang XL, Lu YK, Xu CH, Chen YM. Diet quality scores and risk of nasopharyngeal carcinoma in chinese adults: a case-control study. Nutrients. 2016;8:112.
Irungu CW, Oburra HO, Ochola B. Prevalence and predictors of malnutrition in nasopharyngeal carcinoma. Clin Med Insights Ear Nose Throat. 2015;8:19–22.
Reichlin S. Is leptin a secretion of the brain? J Clin Endocrinol Metab. 1999;84:2267–9.
Solberg R, Aas V, Thoresen GH, Kase ET, Drevon CA, Rustan AC, Reseland JE. Leptin expression in human primary skeletal muscle cells is reduced during differentiation. J Cell Biochem. 2005;96:89–96.
Bain GH, Collie-Duguid E, Murray GI, Gilbert FJ, Denison A, McKiddie F, Ahearn T, Fleming I, Leeds J, Phull P, et al. Tumour expression of leptin is associated with chemotherapy resistance and therapy-independent prognosis in gastro-oesophageal adenocarcinomas. Br J Cancer. 2016;114: e18.
Fan YL, Li XQ. Expression of leptin and its receptor in thyroid carcinoma: distinctive prognostic significance in different subtypes. Clin Endocrinol (Oxf). 2015;83:261–7.
Geng Y, Wang J, Wang R, Wang K, Xu Y, Song G, Wu C, Yin Y. Leptin and HER-2 are associated with gastric cancer progression and prognosis of patients. Biomed Pharmacother. 2012;66:419–24.
Duan X, Tang P, Zhang H, Yu Z. Expression of leptin and adiponectin in esophageal squamous cell carcinoma and their clinical significance. Zhonghua Zhong Liu Za Zhi. 2014;36:839–43.
Gallina S, Sireci F, Lorusso F, Di Benedetto DV, Speciale R, Marchese D, Costantino C, Napoli G, Tessitore V, Cucco D, et al. The immunohistochemical peptidergic expression of leptin is associated with recurrence of malignancy in laryngeal squamous cell carcinoma. Acta Otorhinolaryngol Ital. 2015;35:15–22.
Cascio S, Bartella V, Auriemma A, Johannes GJ, Russo A, Giordano A, Surmacz E. Mechanism of leptin expression in breast cancer cells: role of hypoxia-inducible factor-1alpha. Oncogene. 2008;27:540–7.
Garofalo C, Koda M, Cascio S, Sulkowska M, Kanczuga-Koda L, Golaszewska J, Russo A, Sulkowski S, Surmacz E. Increased expression of leptin and the leptin receptor as a marker of breast cancer progression: possible role of obesity-related stimuli. Clin Cancer Res. 2006;12:1447–53.
**e W, Liu L, He H, Yang K: Prognostic value of hypoxia-inducible factor-1 alpha in nasopharyngeal carcinoma: a meta-analysis. Int J Biol Markers 2018:1724600818778756.
Hosney M, Sabet S, El-Shinawi M, Gaafar KM, Mohamed MM. Leptin is overexpressed in the tumor microenvironment of obese patients with estrogen receptor positive breast cancer. Exp Ther Med. 2017;13:2235–46.
Xu YJ, Shao YF, Zhao X, Geng YT, Wang K, Yin YM. Expression and clinical significance of leptin, the functional receptor of leptin (OB-Rb) and HER-2 in non-small-cell lung cancer: a retrospective analysis. J Cancer Res Clin Oncol. 2011;137:1841–8.
Khabaz MN, Abdelrahman A, Butt N, Damnhory L, Elshal M, Aldahlawi AM, Ashoor S, Al-Maghrabi B, Dobson P, Brown B, et al. Immunohistochemical staining of leptin is associated with grade, stage, lymph node involvement, recurrence, and hormone receptor phenotypes in breast cancer. BMC Womens Health. 2017;17:105.
Chen L, Lin G, Chen K, Liang R, Wan F, Zhang C, Tian G, Zhu X. VEGF promotes migration and invasion by regulating EMT and MMPs in nasopharyngeal carcinoma. J Cancer. 2020;11:7291–301.
Pastushenko I, Blanpain C. EMT transition states during tumor progression and metastasis. Trends Cell Biol. 2019;29:212–26.
Xu M, Cao FL, Li N, Gao X, Su X, Jiang X. Leptin induces epithelial-to-mesenchymal transition via activation of the ERK signaling pathway in lung cancer cells. Oncol Lett. 2018;16:4782–8.
Yan D, Avtanski D, Saxena NK, Sharma D. Leptin-induced epithelial-mesenchymal transition in breast cancer cells requires beta-catenin activation via Akt/GSK3- and MTA1/Wnt1 protein-dependent pathways. J Biol Chem. 2012;287:8598–612.
Liberti MV, Locasale JW. The warburg effect: how does it benefit cancer cells? Trends Biochem Sci. 2016;41:211–8.
Dong Y, Tu R, Liu H, Qing G. Regulation of cancer cell metabolism: oncogenic MYC in the driver’s seat. Signal Transduct Target Ther. 2020;5:124.
Kim JW, Zeller KI, Wang Y, Jegga AG, Aronow BJ, O’Donnell KA, Dang CV. Evaluation of myc E-box phylogenetic footprints in glycolytic genes by chromatin immunoprecipitation assays. Mol Cell Biol. 2004;24:5923–36.
Chen X, Liang R, Zhu X. Anti-EGFR therapies in nasopharyngeal carcinoma. Biomed Pharmacother. 2020;131: 110649.
Chu C, Niu X, Ou X, Hu C. LAPTM4B knockdown increases the radiosensitivity of EGFR-overexpressing radioresistant nasopharyngeal cancer cells by inhibiting autophagy. Onco Targets Ther. 2019;12:5661–77.
Zheng LS, Yang JP, Cao Y, Peng LX, Sun R, **e P, Wang MY, Meng DF, Luo DH, Zou X, et al. SPINK6 promotes metastasis of nasopharyngeal carcinoma via binding and activation of epithelial growth factor receptor. Cancer Res. 2017;77:579–89.
Wang A, Bao Y, Wu Z, Zhao T, Wang D, Shi J, Liu B, Sun S, Yang F, Wang L, Qu L. Long noncoding RNA EGFR-AS1 promotes cell growth and metastasis via affecting HuR mediated mRNA stability of EGFR in renal cancer. Cell Death Dis. 2019;10:154.
Lin XM, Hu L, Gu J, Wang RY, Li L, Tang J, Zhang BH, Yan XZ, Zhu YJ, Hu CL, et al. Choline kinase alpha mediates interactions between the epidermal growth factor receptor and mechanistic target of rapamycin complex 2 in hepatocellular carcinoma cells to promote drug resistance and xenograft tumor progression. Gastroenterology. 2017;152:1187–202.
Barbato C, Frisone P, Braccini L, D'Aguanno S, Pieroni L, Ciotti MT, Catalanotto C, Cogoni C, Ruberti F: Silencing of Ago-2 interacting protein SERBP1 relieves KCC2 repression by miR-92 in neurons. Cells 2022, 11.
Niaz S. The AGO proteins: an overview. Biol Chem. 2018;399:525–47.
Treiber T, Treiber N, Meister G. Regulation of microRNA biogenesis and its crosstalk with other cellular pathways. Nat Rev Mol Cell Biol. 2019;20:5–20.
Pfaff J, Meister G. Argonaute and GW182 proteins: an effective alliance in gene silencing. Biochem Soc Trans. 2013;41:855–60.
Li X, Wang X, Cheng Z, Zhu Q. AGO2 and its partners: a silencing complex, a chromatin modulator, and new features. Crit Rev Biochem Mol Biol. 2020;55:33–53.
Meister G, Landthaler M, Patkaniowska A, Dorsett Y, Teng G, Tuschl T. Human Argonaute2 mediates RNA cleavage targeted by miRNAs and siRNAs. Mol Cell. 2004;15:185–97.
Hu B, Zhong L, Weng Y, Peng L, Huang Y, Zhao Y, Liang XJ. Therapeutic siRNA: state of the art. Signal Transduct Target Ther. 2020;5:101.
Acknowledgements
We thank Chang Gung Medical Foundation Kaohsiung Chang Gung Memorial Hospital Tissue Bank Core Lab (CLRPG8F1702) for excellent technical support.
Funding
This study was supported by the grants CMRPG8H1011-3 from Chang Gung Memorial Hospital, Kaohsiung, Taiwan, by the grant RD11005 from Chung Shan Medical University, Taichung, Taiwan.
Author information
Authors and Affiliations
Contributions
SDL, CCH, and CHC: Experimental design and manuscript draft. SDL, CFH, TJC and SHL: Clinical sample collection, interpretation and analysis. HTT and YLH: Experimental manipulation and analyzed the results related to these assays. CCH: Writing-editing. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This study was approved by the Medical Ethics Committee and animal Use and Management Committee of Chang Gung Memorial Hospital. IRB approval (No. 201700700B0) was provided by Chang Gung Memorial Hospital. IACUC approval (No. 2017050402) was provided by the Institutional Animal Care and Use Committee of Kaohsiung Chang Gung Memorial Hospital.
Consent for publication
Written informed consent was obtained from all patients participating in this study.
Competing interests
The authors declare no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1:
Supplementary Figure S1. Leptin expression profiles are investigated in publicly database and NPC cell lines. (a to d) The mRNA expression levels of leptin in Oncomine HNC databases were determined. (e) Analysis of leptin mRNA and protein levels in NPC cell lines.
Additional file 2:
Supplementary Figure S2. Leptin promotes cell growth in NPC. (a) The mRNA and protein expression levels of leptin were investigated in gain-of-function of leptin in TW02 cells by QPCR and Western blotting. (b) MTS assays was performed to assess the cell proliferation of TW02 cells after transfection of overexpression plasmid. (c) The foci numbers of TW02-leptin transfectants were assessed. The representative images and fold change of foci formation were shown. (d and e) The mRNA and protein expression levels of leptin were investigated in loss-of-function of leptin in TW02 and TW06 cells by QPCR and Western blotting. (f and g). The effect of siLeptin-TW02 on cell proliferation was determined by MTS and colony formation assays. *P<0.05, **P<0.01, ***P<0.001.
Additional file 3:
Supplementary Figure S3. Leptin promotes NPC cells motility by inducing EMT. (a and c) Wound healing assays demonstrated that the overexpression or knockdown of leptin modulated the migratory ability of TW02 cells. (b and d) Transwell assays were performed to examine the change of invasive ability of TW02 cells with leptin overexpression or knockdown. (e) Expressions of EMT markers were detected by Western blotting in TW02 cells with overexpression or knockdown of leptin. *P<0.05, **P<0.01, ***P<0.001.
Additional file 4:
Supplementary Figure S4. Leptin regulates the expressions of cell cycle-related molecules and EGFR/MAPK/c-Myc pathway in TW02 cells. (a) Western blot indicated the expressions of cyclin D1, cyclin E, CDK4, p21 and p27 in leptin-overexpression and leptin-depleted TW02 cells. (b) Western blot analysis was performed to detect the protein levels of p-EGFR, EGFR, p-ERK1/2, ERK1/2, p-AKT, AKT, p-mTOR, mTOR and c-Myc in leptin-overexpression and leptin-depleted TW02 cells.
Additional file 5:
Supplementary Figure S5. miR-874-3p suppresses the malignant properties of TW02 cells. (a and b) MTS assays and colony formation assays were performed to assess the cell proliferation of TW02 cells after transfecting miR-874-3p mimics or miR-874-3p inhibitor and their corresponding negative control. The representative images and fold change of foci formation were shown. (c and d) The migratory and invasive abilities of TW02 cells transfected with miR-874-3p mimics or miR-874-3p inhibitor and their corresponding negative control were assessed by wound healing and Transwell assays. *P<0.05, **P<0.01, ***P<0.001.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Luo, SD., Tsai, HT., Hwang, CF. et al. Aberrant miR-874-3p/leptin/EGFR/c-Myc signaling contributes to nasopharyngeal carcinoma pathogenesis. J Exp Clin Cancer Res 41, 215 (2022). https://doi.org/10.1186/s13046-022-02415-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13046-022-02415-0