Abstract
Background
Influenza viruses, especially Influenza A virus and Influenza B virus, are respiratory pathogens and can cause seasonal epidemics and pandemics. Severe influenza viruses infection induces strong host-defense response and excessive inflammatory response, resulting in acute lung damage, multiple organ failure and high mortality. Isoquercitrin is a Chinese medicine monomer, which was reported to have multiple biological activities, including antiviral activity against HSV, IAV, SARS-CoV-2 and so on. Aims of this study were to assess the in vitro anti-IAV and anti-IBV activity, evaluate the in vivo protective efficacy against lethal infection of the influenza virus and searched for the more optimal method of drug administration of isoquercitrin.
Methods
In vitro infection model (MDCK and A549 cells) and mouse lethal infection model of Influenza A virus and Influenza B virus were used to evaluate the antiviral activity of isoquercitrin.
Results
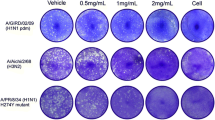
Isoquercitrin could significantly suppress the replication in vitro and in vivo and reduced the mortality of mouse lethal infection models. Compared with virus infection group, isoquercitrin mitigated lung and multiple organ damage. Moreover, isoquercitrin blocked hyperproduction of cytokines induced by virus infection via inactivating NF-κB signaling. Among these routes of isoquercitrin administration, intramuscular injection is a better drug delivery method.
Conclusion
Isoquercitrin is a potential Chinese medicine monomer Against Influenza A Virus and Influenza B Virus infection.
Similar content being viewed by others
Introduction
Influenza virus, belonging to the Orthomyxoviridae family, is a single-stranded, negative sense RNA virus and is one of the most significant pathogens threatening to human health. Influenza viruses are classified into four types (A, B, C, and D), of which influenza A virus (IAV) comprised the majority of global seasonal influenza case. Co-circulating with IAV, influenza B virus (IBV) is also responsible for about 25% of all influenza-related hospitalisations, especially cause high morbidity and mortality in children. [1, 2]. Continuing antigenic drift and shift of IAV and IBV results in new variants which facilitates the cross-species transmission and reduced efficacy of current vaccines and antiviral drugs. Thus, identifying new, more effective drugs against both IAV and IBV is of critical importance.
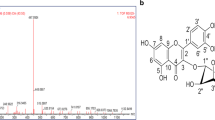
Isoquercitrin, also known as Quercetin-3-O-glucoside, is a kind of flavonol and widely exists in plants, like Houttuynia cordata, Hedyotis diffusa, et al. Currently, besides the direct extraction from plants, the chemical synthesis method was extensively used to prepare isoquercitrin. As a mono-glycoside derivative of quercetin which has diverse biological functions, isoquercitrin has been demonstrated to exhibit anti-tumor [3,4,5], anti-apoptotic [33], but in vivo, 10 mg/kg/day of isoquercitrin had equivalent efficacy to 25 mg/kg/day of oseltamivir phosphate, suggesting this drug is a better in vivo therapeutic drug. Given the continuous emergence of resistance of oseltamivir, perhaps isoquercitrin can become a new treatment option for drug-resistant strains of influenza A virus and influenza B virus.
Isoquercitrin is a flavonoid that can be extracted from natural plants and exhibits antiviral activity against multiple viruses. Isoquercitrin was reported to be the main component of some Traditional Chinese medicine (TCM) that suppressed HSV by inhibiting ROS production and NF-κB activation [9, 10], suppressed IAV (H1N1) replication by inhibiting ROS production [11] and exerted antiviral activity against SARS-CoV-2 [34] and HCoV-229E [13] by blocked Spike protein and Mpro. Isoquercitrin also blocked ZIKV infection through reduced ZIKV NS2B-NS3 protease and NS5 RNA dependent RNA polymerase (RdRp) activity in cells and Ifnar1−/− mice [16, 35]. Taken its inhibitory effect on many RNA viruses and even DNA viruses and results of our researches, isoquercitrin could become a broad-spectrum antiviral candidate.
Cytokine is a kind of peptide or glycoprotein secreted by the cells and perform diverse biological functions, such as immune modulation, involvement in inflammatory responses, etc. Cytokine is a crucial component of antiviral innate immune, but severely viral infections are always accompanied by cytokine dysregulation and amplification, even becomes the main factor for viral pneumonia during the later phase after virus infection [36]. Considering that, reducing cytokine production is a potential treatment strategy. Thus, multiple immunomodulators, such as Janus kinase (JAK) inhibitors [37] and anti‐IL‐6 antibody [38], are developed to control virus-induced “cytokine storm” and mitigate disease progression. In addition to chemical molecules and antibodies, traditional Chinese medicine gradually become an alternative source for anti-influenza virus drugs [39]. Fructuscorni and Radix salviae miltiorrhizae possess resistance to influenza because they can enhance specific and non-specific immunity [40]. As a natural drug, isoquercitrin was reported to reduce the virus-induced ROS production and NF-κB activation [10]. Taken together, isoquercitrin not only has antiviral activity against IAV and IBV, but also represses the expression of pro-inflammatory cytokines by inactiviating NF-κB signal.
The pathogenicity of an influenza virus depends on many factors, such as cell tropism, replicate efficiency, and host immunity. Both IAV and IBV prefer to replicate in respiratory system and innate immune cells are recruited to lung and produce massive cytokines and chemokines after influenza virus infection, inducing pneumonia, even multi-organ failure [41]. Unexpectedly, adolescents and children are more susceptible to IBV and sometimes IBV-associated diseases develop earlier and more severe than IAV [42]. Characterized by inflammatory cell infiltration, alveolar epithelial cells sloughing, alveolar wall hyperemia and thickness, alveolar hemorrhage and damage, interstitial-alveolar edema, etc. the degree of which always becomes a standard for assess the pathogenicity of viruses and the efficacy of therapeutic drugs [43]. Fortunately, in this study, isoquercitrin was found as a treatment drug for viral pneumonia.
The dose and route of administration are main factors affecting the drug efficacy [44]. In other researches, isoquercitrin was demonstrated to exert therapeutic effect at lower doses, so we only explore its dose-dependent relationship. In a murine model, the common route for drug administration include tail-vein injection, intraperitoneal injection, intramuscular injection, subcutaneous injection, oral gavage and intranasal administration. Compared to tail-vein injection, which can increase the risk of infection and thromboembolic events [45], intraperitoneal injection, intramuscular injection and subcutaneous injection are more safe and long-acting. Oral administration is the predominant method of drug administration of natural products [46], but its bioavailability should be considered. In our study, isoquercitrin treatment via different administration could exert antiviral effect, suggesting that this drug may directly exert the pharmacodynamic effect in target organ, without requirement of metabolic conversion. Additionally, considering the influence of different drug administration on the antiviral effect of isoquercitrin, more experiments should be considered be implemented to identify the optimal dosing regimen.
Conclusion
Above findings suggest that isoquercitrin has potent antiviral activity against influenza A (H1N1) virus and influenza B virus in vitro and in vivo. Lethal infection of influenza virus induced excessive production of cytokines and severe lung damage, but these outcomes were suppressed by isoquercitrin, indicating that this drug could benefit severe patients with influenza virus infection. Additionally, through comparison between different drug delivery methods, the i.m. was proved as a better method for administration of isoquercitrin.
Availability of data and materials
The data produced from this study are available from the first author and the corresponding author on reasonable request.
References
Dumm RE, Heaton NS. The development and use of reporter influenza B viruses. Viruses. 2019;11(8):736.
Yazici Özkaya P, Turanli EE, Metin H, Aydın Uysal A, Çiçek C, Karapinar B. Severe influenza virus infection in children admitted to the PICU: comparison of influenza A and influenza B virus infection. J Med Virol. 2022;94(2):575–81.
Chen Q, Li P, Li P, Xu Y, Li Y, Tang B. Isoquercitrin inhibits the progression of pancreatic cancer in vivo and in vitro by regulating opioid receptors and the mitogen-activated protein kinase signaling pathway. Oncol Rep. 2015;33(2):840–8.
Huang G, Tang B, Tang K, Dong X, Deng J, Liao L, Liao Z, Yang H, He S. Isoquercitrin inhibits the progression of liver cancer in vivo and in vitro via the MAPK signaling pathway. Oncol Rep. 2014;31(5):2377–84.
Chen F, Chen X, Yang D, Che X, Wang J, Li X, Zhang Z, Wang Q, Zheng W, Wang L, Wang X, Song X. Isoquercitrin inhibits bladder cancer progression in vivo and in vitro by regulating the PI3K/Akt and PKC signaling pathways. Oncol Rep. 2016;36(1):165–72.
Wang H, **a W, Long G, Pei Z, Li Y, Wu M, Wang Q, Zhang Y, Jia Z, Chen H. Isoquercitrin Ameliorates Cisplatin-Induced Nephrotoxicity Via The Inhibition Of Apoptosis, Inflammation, And Oxidative Stress. Front Pharmacol. 2020;11: 599416.
Kim JH, Lee S, Cho EJ. Acer okamotoanum and isoquercitrin improve cognitive function via attenuation of oxidative stress in high fat diet and amyloid beta-induced mice. Food Funct. 2019;10(10):6803–14.
Liang S, Xu Z, Ruan Y, Niu T, Guo W, Jiang W, Hou J. Isoquercitrin attenuates renal ischemia/reperfusion injury through antioxidation, anti-inflammation, and anti-apoptosis in mice. Transplant Proc. 2020;52(3):1014–9.
Chen X, Wang Z, Yang Z, Wang J, Xu Y, Tan RX, Li E. Houttuynia cordata blocks HSV infection through inhibition of NF-κB activation. Antiviral Res. 2011;92(2):341–5.
Kim B, Kim YS, Hwang YH, Yang HJ, Li W, Kwon EB, Kim TI, Go Y, Choi JG. Quercus acuta Thunb (Fagaceae) and Its component, isoquercitrin, inhibit HSV-1 replication by suppressing virus-induced ROS production and NF-κB activation. Antioxidants. 2021;10(10):1638.
Nile SH, Kim DH, Nile A, Park GS, Gansukh E, Kai G. Probing the effect of quercetin 3-glucoside from Dianthus superbus L against influenza virus infection- In vitro and in silico biochemical and toxicological screening. Food Chem Toxicol. 2020;135: 110985.
Kim Y, Chang KO. Protein disulfide isomerases as potential therapeutic targets for influenza A and B viruses. Virus Res. 2018;247:26–33.
Zhu Y, Scholle F, Kisthardt SC, **e DY. Flavonols and dihydroflavonols inhibit the main protease activity of SARS-CoV-2 and the replication of human coronavirus 229E. Virology. 2022;571:21–33.
Johnson TO, Adegboyega AE, Ojo OA, Yusuf AJ, Iwaloye O, Ugwah-Oguejiofor CJ, Asomadu RO, Chukwuma IF, Ejembi SA, Ugwuja EI, Alotaibi SS, Albogami SM, Batiha GE, Rajab BS, Conte-Junior CA. A computational approach to elucidate the interactions of chemicals from Artemisia annua targeted toward SARS-CoV-2 main protease inhibition for COVID-19 treatment. Front Med. 2022;9: 907583.
Gaudry A, Bos S, Viranaicken W, Roche M, Krejbich-Trotot P, Gadea G, Desprès P, El-Kalamouni C. The flavonoid isoquercitrin precludes initiation of Zika virus infection in human cells. Int J Mol Sci. 2018;19(4):1093.
Wong G, He S, Siragam V, Bi Y, Mbikay M, Chretien M, Qiu X. Antiviral activity of quercetin-3-β-O-D-glucoside against Zika virus infection. Virol Sin. 2017;32(6):545–7.
dos Santos AE, Kuster RM, Yamamoto KA, Salles TS, Campos R, de Meneses MD, Soares MR, Ferreira D. Quercetin and quercetin 3-O-glycosides from Bauhinia longifolia (Bong) steud show anti-mayaro virus activity. Parasit Vectors. 2014;7:130.
Kim CH, Kim JE, Song YJ. antiviral activities of quercetin and isoquercitrin against human herpesviruses. Molecules. 2020;25(10):2379.
Dai J, Gu L, Su Y, Wang Q, Zhao Y, Chen X, Deng H, Li W, Wang G, Li K. Inhibition of curcumin on influenza A virus infection and influenzal pneumonia via oxidative stress, TLR2/4, p38/JNK MAPK and NF-κB pathways. Int Immunopharmacol. 2018;54:177–87.
Dai JP, Wang QW, Su Y, Gu LM, Zhao Y, Chen XX, Chen C, Li WZ, Wang GF, Li KS. Emodin inhibition of influenza A virus replication and influenza viral pneumonia via the Nrf2, TLR4, p38/JNK and NF-kappaB pathways. Molecules. 2017;22(10):1754.
Ilyushina NA, et al. Comparative study of influenza virus replication in MDCK cells and in primary cells derived from adenoids and airway epithelium. J Virol. 2012;86:11725–34.
Kim Y, Narayanan S, Chang KO. Inhibition of influenza virus replication by plant-derived isoquercetin. Antiviral Res. 2010;88(2):227–35.
Smee DF, Hurst BL, Wong MH, Bailey KW, Tarbet EB, Morrey JD, Furuta Y. Effects of the combination of favipiravir (T-705) and oseltamivir on influenza A virus infections in mice. Antimicrob Agents Chemother. 2010;54(1):126–33.
Klomp M, Ghosh S, Mohammed S, Nadeem KM. From virus to inflammation, how influenza promotes lung damage. J Leukoc Biol. 2021;110(1):115–22.
Teijaro JR, Walsh KB, Cahalan S, Fremgen DM, Roberts E, Scott F, Martinborough E, Peach R, Oldstone MB, Rosen H. Endothelial cells are central orchestrators of cytokine amplification during influenza virus infection. Cell. 2011;146(6):980–91.
Kumar N, **n ZT, Liang Y, Ly H, Liang Y. NF-kappaB signaling differentially regulates influenza virus RNA synthesis. J Virol. 2008;82(20):9880–9.
Heikkinen T, Ikonen N, Ziegler T. Impact of influenza B lineage-level mismatch between trivalent seasonal influenza vaccines and circulating viruses, 1999–2012. Clin Infect Dis. 2014;59(11):1519–24.
Shi Y, Zhang B, Lu Y, Qian C, Feng Y, Fang L, Ding Z, Cheng D. Antiviral activity of phenanthrenes from the medicinal plant Bletilla striata against influenza A virus. BMC Complement Altern Med. 2017;17(1):273.
Garcia V, Aris-Brosou S. Comparative dynamics and distribution of influenza drug resistance acquisition to protein M2 and neuraminidase inhibitors. Mol Biol Evol. 2014;31(2):355–63.
Zhu Q, Bang TH, Ohnuki K, Sawai T, Sawai K, Shimizu K. Inhibition of neuraminidase by Ganoderma triterpenoids and implications for neuraminidase inhibitor design. Sci Rep. 2015;5:13194.
Hai R, Schmolke M, Leyva-Grado VH, Thangavel RR, Margine I, Jaffe EL, Krammer F, Solórzano A, García-Sastre A, Palese P, Bouvier NM. Influenza A(H7N9) virus gains neuraminidase inhibitor resistance without loss of in vivo virulence or transmissibility. Nat Commun. 2013;4:2854.
Tsedenbal N, Tsend-Ayush A, Badarch D, Jav S, Pagbajab N. Influenza B viruses circulated during last 5 years in Mongolia. PLoS ONE. 2018;13(11): e0206987.
Cáceres CJ, Hu Y, Cárdenas-García S, Wu X, Tan H, Carnaccini S, Gay LC, Geiger G, Ma C, Zhang QY, Rajao D, Perez DR, Wang J. Rational design of a deuterium-containing M2–S31N channel blocker UAWJ280 with in vivo antiviral efficacy against both oseltamivir sensitive and—resistant influenza A viruses. Emerg Microbes Infect. 2021;10(1):1832–48.
Ye M, Luo G, Ye D, She M, Sun N, Lu YJ, Zheng J. Network pharmacology, molecular docking integrated surface plasmon resonance technology reveals the mechanism of Toujie Quwen Granules against coronavirus disease 2019 pneumonia. Phytomedicine. 2021;85: 153401.
Kumar S, El-Kafrawy SA, Bharadwaj S, Maitra SS, Alandijany TA, Faizo AA, Khateb AM, Dwivedi VD, Azhar EI. Discovery of bispecific lead compounds from Azadirachta indica against ZIKA NS2B-NS3 protease and NS5 RNA dependent RNA polymerase using molecular simulations. Molecules. 2022;27(8):2562.
Peiris JS, Yu WC, Leung CW, Cheung CY, Ng WF, Nicholls JM, Ng TK, Chan KH, Lai ST, Lim WL, Yuen KY, Guan Y. Re-emergence of fatal human influenza A subtype H5N1 disease. Lancet. 2004;363(9409):617–9.
Lin CM, Cooles FA, Isaacs JD. Basic mechanisms of JAK inhibition. Mediterr J Rheumatol. 2020;31(Suppl 1):100–4.
Vanderbeke L, Van Mol P, Van Herck Y, De Smet F, Humblet-Baron S, Martinod K, Antoranz A, Arijs I, Boeckx B, Bosisio FM, Casaer M, Dauwe D, De Wever W, Dooms C, Dreesen E, Emmaneel A, Filtjens J, Gouwy M, Gunst J, Hermans G, Jansen S, Lagrou K, Liston A, Lorent N, Meersseman P, Mercier T, Neyts J, Odent J, Panovska D, Penttila PA, Pollet E, Proost P, Qian J, Quintelier K, Raes J, Rex S, Saeys Y, Sprooten J, Tejpar S, Testelmans D, Thevissen K, Van Buyten T, Vandenhaute J, Van Gassen S, Velásquez Pereira LC, Vos R, Weynand B, Wilmer A, Yserbyt J, Garg AD, Matthys P, Wouters C, Lambrechts D, Wauters E, Wauters J. Monocyte-driven atypical cytokine storm and aberrant neutrophil activation as key mediators of COVID-19 disease severity. Nat Commun. 2021;12(1):4117.
Dai JP, Wang QW, Su Y, Gu LM, Deng HX, Chen XX, Li WZ, Li KS. Oxymatrine inhibits influenza A virus replication and inflammation via TLR4, p38 MAPK and NF-κB pathways. Int J Mol Sci. 2018;19(4):965.
Shen X, Zhao Z, Wang H, Guo Z, Hu B, Zhang G. Elucidation of the anti-inflammatory mechanisms of bupleuri and scutellariae radix using system pharmacological analyses. Mediators Inflamm. 2017;2017:3709874.
de Jong MD, Simmons CP, Thanh TT, Hien VM, Smith GJ, Chau TN, Hoang DM, Chau NV, Khanh TH, Dong VC, Qui PT, Cam BV, do Ha Q, Guan Y, Peiris JS, Chinh NT, Hien TT, Farrar J. Fatal outcome of human influenza A (H5N1) is associated with high viral load and hypercytokinemia. Nat Med. 2006;12(10):1203–7.
He D, Chiu AP, Lin Q, Yu D. Spatio-temporal patterns of proportions of influenza B cases. Sci Rep. 2017;7:40085.
Zaraket H, Hurt AC, Clinch B, Barr I, Lee N. Burden of influenza B virus infection and considerations for clinical management. Antiviral Res. 2021;185: 104970.
Sharma A, Anumanthan G, Reyes M, Chen H, Brubaker JW, Siddiqui S, Gupta S, Rieger FG, Mohan RR. Epigenetic modification prevents excessive wound healing and scar formation after glaucoma filtration surgery. Invest Ophthalmol Vis Sci. 2016;57(7):3381–9.
Liu G, Franssen E, Fitch MI, Warner E. Patient preferences for oral versus intravenous palliative chemotherapy. J Clin Oncol. 1997;15:110–5.
Li Y, Cao H, Liu M, Zhang B, Zhang X, Shi D, Guo L, Duan J, Zhou X, Zhu H, Zhang Q. Different modulation of Panax notoginseng on the absorption profiling of triptolide and tripterine from Tripterygium wilfordii in rat intestine. Chin Med. 2018;13:1.
Sima M, Lv C, Qi J, Guo J, Luo R, Deng X, Li Y, Wang T, Yue D, Gao Y. Anti-inflammatory effects of theaflavin-3′-gallate during influenza virus infection through regulating the TLR4/MAPK/p38 pathway. Eur J Pharmacol. 2023;938: 175332.
Acknowledgements
Not applicable.
Funding
This work was supported by the National Key Research and Development Program of China (2021YFC2301701), the Youth Science and Technology Innovation Outstanding Talents Special Project of Jilin Provincial Department of Science and Technology (NO.20210509004RQ) and the National Natural Science Foundation of Jilin (NO.YDZJ202201ZYTS174).
Author information
Authors and Affiliations
Contributions
YG, DY and TW designed experiments and provided guidance. Experiments were performed by RL and CL. XD and Mingwei Sima provided drugs. All viruses used in this research were provided by YL, WS. JG and JQ provided experimental guidance and manuscript revision.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Animal experiments were conducted following the protocols of Changchun Veterinary Research Institute, Chinese Academy of Agricultural Sciences (Approval Number: IACUC of AMMS-11-2022-036). Additionally, all influenza virus infection experiments are conducted in a biosafety level 2 laboratory of Changchun Veterinary Research Institute, Chinese Academy of Agricultural Sciences according to biosafety regulations.
Consent for publication
All authors consent to publish this manuscript.
Competing interests
Authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1:
Fig. S1. The effect of timing of drug administration and dose on anti-IAV A–B and anti-IBV C–D activity of isoquercitrin in MDCK cells and A549 cells. Colors annotate three drug administration protocols, pre-treatment (gray), co-treatment (black) and post-treatment (pink). Fig. S2. Effect of isoquercitrin on pathological damage caused by influenza virus infection. A HE staining of heart, liver, spleen and kidney tissues in influenza virus-infected mice and drug-treated mice at 5 dpi. B Immunohistochemistry staining of heart, liver, spleen and kidney tissues in influenza virus-infected mice and drug-treated mice at 5 dpi. C The percentage of NP positive cells in main tissues (heart, liver, spleen, kidney) in B. Fig. S3. Isoquercitrin inhibits cytokines induced by influenza A virus infection. Relative mRNA expression level of type I IFN (IFN-α and IFN-β), pro-inflammatory cytokines (TNF-α, IL-6 and IL-1β), anti-inflammatory cytokine (IL-10) and IFN induced gene (ISG54 and ISG56) in mouse lung tissues of each group at 5 dpi.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Luo, R., Lv, C., Wang, T. et al. A potential Chinese medicine monomer against influenza A virus and influenza B virus: isoquercitrin. Chin Med 18, 144 (2023). https://doi.org/10.1186/s13020-023-00843-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13020-023-00843-4