Abstract
Background
It was always challenging to accurately and effectively reconstruct the complicated defects with three-dimensional tissue deficits in the extremities. Muscle-chimeric perforator flap is an excellent choice for repairing those complicated wound. However, problems like donor-site morbidity and time-consuming intramuscular dissection still exist. This purpose of this study was to present a novel design of the thoracodorsal artery perforator (TDAP) chimeric flap for the customized reconstruction of complex three-dimensional tissue defects in the extremities.
Methods
From January 2012 to June 2020, 17 patients with complex three-dimensional deficits in the extremities were retrospectively analyzed. All patients in this series underwent extremity reconstruction using latissimus dorsi (LD)-chimeric TDAP flap. Three different types of LD-chimeric TDAP flaps were performed.
Results
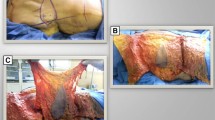
A total of seventeen TDAP chimeric flaps were successfully harvested for the reconstruction of those complex three-dimensional defects in extremities. Among them, Design Type A flaps were used in 6 cases, Design Type B flaps were performed in 7 cases, and Design Type C flaps were used in the remaining 4 cases. The sizes of the skin paddles ranged from 6 cm × 3 cm to 24 cm × 11 cm. Meanwhile, the sizes of the muscle segments ranged from 3 cm × 4 cm to 33 cm × 4 cm. All the flaps survived. Nevertheless, one case required re-exploration owing to venous congestion. Moreover, the primary closure of the donor site was successfully achieved in all patients, and the mean follow-up time was 15.8 months. Most of the cases displayed satisfactory contour.
Conclusion
The LD-chimeric TDAP flap is available for the reconstruction of complicated defects with three-dimensional tissue deficits in the extremities. It provided a flexible design for customized coverage of complex soft tissue defects with limited donor site morbidity.
Similar content being viewed by others
Introduction
High-energy trauma often leads to complicated wounds with three-dimensional deficits in the extremities involving superficial soft tissue defects and dead spaces of varying location, which are challenging to precisely and efficiently repair simultaneously [1,2,3]. The challenge of this type of reconstruction requires not only coverage of surface soft-tissue defects but also appropriate obliteration of dead space in a single procedure.
Numerous reconstructive strategies have been described for repairing complex three-dimensional soft tissue in the literature [4, 5]. Free latissimus dorsi muscle flaps have been widely accepted as a reliable option for reconstruct complicated defects with three-dimensionally inset multi-component tissue transfer. Nonetheless, reconstruction of this wound with a musculocutaneous flap always is more challenging for the reconstructive surgeon because of its restriction in the range of motion of muscle components. Moreover, those approaches are hindered by higher donor-site morbidity and poor contouring [6,7,8,9].
Recently, several studies have demonstrated that chimeric perforator flaps are one of the most valuable strategy for reconstructing complicated wounds with three-dimensional deficits, which can be attributed to their greater spatial freedom, more flexible design, the economy of donor incision, and superior aesthetic outcomes.[1, 10,11,12,13]. The latissimus dorsi (LD) muscle-chimeric thoracodorsal artery perforator (TDAP) flap has been introduced as a reliable strategy for repairing complicated wound [14]. However, harvesting chimeric perforator flaps with conventional design often lack the versatility to offer adequate tissue volume and allow precise tissue positioning to optimally cover the wound. Notably, when reconstructing complicated wounds with three-dimensional tissue deficits, customized chimeric perforator flap designs are necessitated for precisely coverage of superficial soft tissue defects and effective obliteration of dead space to facilitate wound healing. Moreover, the traditional designing of a LD muscle-chimeric TDAP flap typically requires time-consuming intramuscular dissection, leading to longer operative time and higher donor-site morbidity. To address this, three different types of LD muscle-chimeric TDAP flaps were designed to minimize donor-site morbidity and shorten the operative time. Thus, our report focused on the various designs for customized reconstruction of complex three-dimensional defects to minimize donor-site morbidity and shorten the operation time, which has rarely been addressed in previous studies. To the best of our knowledge, the use of individually designed chimeric perforator flaps to reconstruct complex three-dimensional soft tissue defects has not been studied in the context of free TDAP flaps.
The purpose of this study was to present our experience on a novel design of the TDAP chimeric flap and its various designs for the customized reconstruction of complex three-dimensional defects in the extremities.
Patients and methods
Between January 2012 and June 2020, seventeen patients who suffered with complicated wound with three-dimensional tissue deficits were performed extremity reconstruction using a LD-muscle chimeric TDAP flap. Patient ages ranged from 2 to 39 years (mean age 20.24 years; 7 females, 10 males). Of these patients, five patients suffered from chronic osteomyelitis, two were injured by a crushing incident, three underwent tumor resection surgeries, and the remaining seven patients were injured in road traffic accidents. The complex soft-tissue defects were classified into three types according to the location of dead space. When the dead space was located at the center of the wound, it was classified as type I; when the dead space was located at the edge of the wound, it was classified as type II, while Type III was classified as a wound with extensive surface soft-tissue defect and dead space. Additionally, the type III category was further classified into two subtypes based on the location of dead space. When the dead space was located at the center of the wound, it was classified as type IIIA; when the dead space was at the edge of the wound, it was classified as type IIIB. Patient details are presented in Table 1. The study conformed to the ethical guidelines of the Hospital Ethical Committee of the ** scar-related issues at the donor site [35]. Our previous study also demonstrated that the transverse design of the scapular artery perforator flap presented a more aesthetic appearance. Moreover, the transverse design causes less lateral movement of breast mounds and nipples during donor-site closure. Considering the donor site's cosmetic appearance improvement, the recent practice showed that using a transverse designed flap was more effective than using a non-transverse one. Also, for female patients, the scar can be readily concealed with a brassiere.
The limitations of this study include the small number of cases as well as its retrospective character. And other limitations include the absence of a direct comparison group and a standardized assessment tool for the objective assessment of the result following reconstructive surgery. Thus, further studies, such as prospective case–control or randomized studies, are needed in future study.
Conclusions
To summarize, the TDA chimeric perforator flap is a reliable option for the reconstruction of complex three-dimensional defects of the extremities. This flexible approach provides various designs for customized coverage of complex three-dimensional defects with limited donor site morbidity. Moreover, the flexible shape of this flap permits alteration to match the shape of the recipient site. In addition, we introduced a novel classification system of chimeric perforator flaps for reconstructive surgeons and trainees to gain a better understanding of chimeric flaps design and allow safe, effective, and aesthetically superior reconstruction of complex three-dimensional defects.
Availability of data and materials
Data sets or analyses used in the current study are available from the corresponding author upon reasonable request.
References
Zhang YX, Hayakawa TJ, Levin LS, Hallock GG, Lazzeri D. The economy in autologous tissue transfer: Part 1. the kiss flap technique. Plast Reconstr Surg. 2016;137(3):1018–30.
Qing L, Wu P, Zhou Z, Yu F, Tang M, Tang J. A design for the dual skin paddle circumflex scapular artery perforator flap for the reconstruction of complex soft-tissue defects in children: anatomical study and clinical applications. Ann Plast Surg. 2019.
Qing L, Li X, Wu P, Zhou Z, Yu F, Tang J. Customized reconstruction of complex soft-tissue defect in the hand and forearm with individual design of chain-linked bilateral anterolateral thigh perforator flaps. J Plast Reconstr Aesthet Surg. 2019;72(12):1909–16.
Münchow S, Dragu A, Rammelt S, Reeps C, Stiehler M. Plastic-reconstructive treatment algorithm for dead space management following septic total hip arthroplasty removal: AV loop and two-stage free myocutaneous latissimus dorsi flap. Z Orthop Unfall. 2021;159(6):674–80.
Li BH, Jung HJ, Choi SW, Kim SM, Kim MJ, Lee JH. Latissimus dorsi (LD) free flap and reconstruction plate used for extensive maxillo-mandibular reconstruction after tumour ablation. J Craniomaxillofac Surg. 2012;40(8):e293-300.
Ugurlu K, Sacak B, Huthut I, Karsidag S, Sakiz D, Bas L. Reconstructing wide palatomaxillary defects using free flaps combining bare serratus anterior muscle fascia and scapular bone. J Oral Maxillofac Surg. 2007;65(4):621–9.
Serra MP, Longhi P, Carminati M, Righi B, Robotti E. Microsurgical scalp and skull reconstruction using a combined flap composed of serratus anterior myo-osseous flap and latissimus dorsi myocutaneous flap. J Plast Reconstr Aesthet Surg. 2007;60(10):1158–61.
Sano K, Hallock GG, Ozeki S, Suzuki H, Mawatari R, Yoshino K, et al. Devastating massive knee defect reconstruction using the cornucopian chimera flap from the subscapular axis: two case reports. J Reconstr Microsurg. 2006;22(1):25–32.
Milano F. A combined flap for root coverage. Int J Periodontics Restorative Dent. 1998;18(6):544–51.
Zhang YX, Messmer C, Pang FK, Ong YS, Feng SQ, Qian Y, et al. A novel design of the multilobed latissimus dorsi myocutaneous flap to achieve primary donor-site closure in the reconstruction of large defects. Plast Reconstr Surg. 2013;131(5):752e-e758.
Yoshimatsu H, Yamamoto T, Hayashi A, Iida T. Proximal-to-distally elevated superficial circumflex iliac artery perforator flap enabling hybrid reconstruction. Plast Reconstr Surg. 2016;138(4):910–22.
Lee KT, Wiraatmadja ES, Mun GH. Free latissimus dorsi muscle-chimeric thoracodorsal artery perforator flaps for reconstruction of complicated defects: does muscle still have a place in the domain of perforator flaps? Ann Plast Surg. 2015;74(5):565–72.
Agarwal JP, Agarwal S, Adler N, Gottlieb LJ. Refining the intrinsic chimera flap: a review. Ann Plast Surg. 2009;63(4):462–7.
Arikawa M, Miyamoto S, Fujiki M, Higashino T, Oshima A, Sakuraba M. Comparison of donor site drainage duration and seroma rate between latissimus dorsi musculocutaneous flaps and thoracodorsal artery perforator flaps. Ann Plast Surg. 2017;79(2):183–5.
He J, Qing L, Wu P, Zhou Z, Guliyeva G, Yu F, et al. One-stage reconstruction of extensive composite extremity defects with low donor site morbidity: a retrospective case series of combined transfer of a vascularized fibula flap and a perforator flap. Injury. 2022.
Qing L, Wu P, Yu F, Zhou Z, Tang J. Use of a sequential chimeric perforator flap for one-stage reconstruction of complex soft tissue defects of the extremities. Microsurgery. 2020;40(2):167–74.
Qing L, Wu P, Zhou Z, Yu F, Tang J. Customized reconstruction of complex three-dimensional defects in the extremities with individual design of vastus lateralis muscle-chimeric multi-lobed anterolateral thigh perforator flap. J Plast Surg Hand Surg. 2019;53(5):271–8.
Kim SW, Youn DG, Kim JT, Kim YH. A thoracodorsal artery perforator chimeric free flap for prevention of microvascular pedicle compression in lower extremity reconstruction. Microsurgery. 2015.
Philandrianos C, Moullot P, Gay AM, Bertrand B, Legre R, Kerfant N, et al. Soft tissue coverage in distal lower extremity open fractures: comparison of free anterolateral thigh and free latissimus dorsi flaps. J Reconstr Microsurg. 2018;34(2):121–9.
Miyamoto S, Fujiki M, Sakuraba M. Clinical analysis of 33 flow-through latissimus dorsi flaps. J Plast Reconstr Aesthet Surg. 2015;68(10):1425–31.
Hallock GG. Simultaneous transposition of anterior thigh muscle and fascia flaps: an introduction to the chimera flap principle. Ann Plast Surg. 1991;27(2):126–31.
Hallock GG. The chimera flap: a quarter century odyssey. Ann Plast Surg. 2017;78(2):223–9.
Kim SW, Youn S, Kim JD, Kim JT, Hwang KT, Kim YH. Reconstruction of extensive lower limb defects with thoracodorsal axis chimeric flaps. Plast Reconstr Surg. 2013;132(2):470–9.
Huang WC, Chen HC, Wei FC, Cheng MH, Schnur DP. Chimeric flap in clinical use. Clin Plast Surg. 2003;30(3):457–67.
Hallock GG. Further clarification of the nomenclature for compound flaps. Plast Reconstr Surg. 2006;117(7):151e-e160.
Hallock GG. The complete nomenclature for combined perforator flaps. Plast Reconstr Surg. 2011;127(4):1720–9.
Bigdeli AK, Thomas B, Schmidt VJ, Kotsougiani D, Hernekamp FJ, Hirche C, et al. The conjoined parascapular and latissimus dorsi free flap for reconstruction of extensive knee defects. Microsurgery. 2018;38(8):867–75.
Azouz SM, Castel NA, Vijayasekaran A, Rebecca AM, Lettieri SC. Lower-limb reconstruction with chimeric flaps: the quad flap. Microsurgery. 2018.
Qing L, Wu P, Yu F, Zhou Z, Tang J. Use of dual-skin paddle anterolateral thigh perforator flaps in the reconstruction of complex defect of the foot and ankle. J Plast Reconstr Aesthet Surg. 2018.
Luo Z, Qing L, Zhou Z, Wu P, Yu F, Tang J. Reconstruction of large soft tissue defects of the extremities in children using the kiss deep inferior epigastric artery perforator flap to achieve primary closure of donor site. Ann Plast Surg. 2019;82(1):64–70.
Yang X, Zhang G, Liu Y, Yang J, Ding M, Tang M. Vascular anatomy and clinical application of anterolateral leg perforator flaps. Plast Reconstr Surg. (10) 2013;131(4):534e–43e.
Ogata F, Azuma R, Kikuchi M, Koshima I, Morimoto Y. Novel lymphography using indocyanine green dye for near-infrared fluorescence labeling. Ann Plast Surg. 2007;58(6):652–5.
Kuo YR, Seng-Feng J, Kuo FM, Liu YT, Lai PW. Versatility of the free anterolateral thigh flap for reconstruction of soft-tissue defects: review of 140 cases. Ann Plast Surg. 2002;48(2):161–6.
Koshima I, Fukuda H, Utunomiya R, Soeda S. The anterolateral thigh flap; variations in its vascular pedicle. Br J Plast Surg. 1989;42(3):260–2.
Lee KT, Kim A, Mun GH. Comprehensive analysis of donor-site morbidity following free thoracodorsal artery perforator flap harvest. Plast Reconstr Surg. 2016;138(4):899–909.
Acknowledgements
We would like to thank Umar Zeb Khan for hel** to revise and polish the language.
Funding
This publication was funded in part by the National Natural Science Foundation of China (To Dr Tang, No. 81472104 and To Dr Qing, No. 81901978).
Author information
Authors and Affiliations
Contributions
L.Q., J.T. and P.W. performed the surgery; L.Q., J.T. and P.W. significantly contributed to the analysis and preparation of the manuscript; G.L. and X.L. wrote the manuscript and performed the data analysis; the final manuscript was read and approved by the authors. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The study conformed to the ethical criterion of the **angya Hospital's Hospital Ethical Committee. The protocol was carried out in conformity with the ethical guidelines outlined in the 1975 Helsinki Declaration and all updates thereto.
Competing interests
The authors declare that they have no conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Qing, L., Luo, G., Li, X. et al. Individualized design of thoracodorsal artery perforator chimeric flap for customized reconstruction of complex three-dimensional defects in the extremities. J Orthop Surg Res 18, 367 (2023). https://doi.org/10.1186/s13018-023-03852-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13018-023-03852-z