Abstract
Dysregulated mucosal immune responses and colonic fibrosis impose two formidable challenges for ulcerative colitis treatment. It indicates that monotherapy could not sufficiently deal with this complicated disease and combination therapy may provide a potential solution. A chitosan-modified poly(lactic-co-glycolic acid) nanoparticle (CS-PLGA NP) system was developed for co-delivering patchouli alcohol and simvastatin to the inflamed colonic epithelium to alleviate the symptoms of ulcerative colitis via remodeling immune microenvironment and anti-fibrosis, a so-called “two-birds-one-stone” nanotherapeutic strategy. The bioadhesive nanomedicine enhanced the intestinal epithelial cell uptake efficiency and improved the drug stability in the gastrointestinal tract. The nanomedicine effectively regulated the Akt/MAPK/NF-κB pathway and reshaped the immune microenvironment through repolarizing M2Φ, promoting regulatory T cells and G-MDSC, suppressing neutrophil and inflammatory monocyte infiltration, as well as inhibiting dendritic cell maturation. Additionally, the nanomedicine alleviated colonic fibrosis. Our work elucidates that the colon-targeted codelivery for combination therapy is promising for ulcerative colitis treatment and to address the unmet medical need.
Graphical Abstract

Similar content being viewed by others
Introduction
Ulcerative colitis (UC) is the major form of inflammatory bowel disease (IBD). UC is a relapsing and remitting mucosal inflammation that starts from the rectum and spreads continuously to the proximal segments of the colon [1]. The prevalence of UC has been rising in the newly industrialized countries in Africa, Asia, and South America [2]. For example, it is about 11.6 per 100,000 people in China [3]. Additionally, the incidence of colorectal cancer in Asian patients with UC has been also increasing [4]. This situation imposes a great need for effective UC treatment, but the current therapy methods cannot meet the expectations due to the unsustainable efficacy [5].
The maladjustment of the immune system affected by heredity, environment, and gut microbiota is closely related to the progress of UC [6]. The intestinal immune microenvironment consists of intestinal epithelial cells, macrophages, dendritic cells (DCs), regulatory T cells (Tregs), and inflammatory T cells, which collaboratively maintain immune homeostasis [7]. The inflammatory microenvironment can be a target for UC treatment. Macrophages (MΦ) are pivotal in coordinating the progress of UC [8]. Macrophages are characterized by their diversity and plasticity in response to environmental signals and are traditionally classified into M1Φ with pro-inflammatory/anti-microbial activity and M2Φ with anti-inflammatory activity/tissue repair [9]. An increase of M1Φ amount in the pathological site of colitis predicts the worsening disease stage [10]. Re-education from M1 to M2 phenotype is a potential strategy for UC treatment [11].
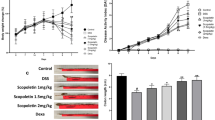
In addition to the aggravated inflammatory immune responses in colitis, excessive proliferation of fibroblasts and myofibroblasts contributes to the deposition of extracellular matrix (ECM) and the fibrosis of the intestinal wall. Severe intestinal fibrosis may result in intestinal obstruction and require surgical intervention [12]. Traditional treatments mainly focus on alleviating the symptoms of UC through anti-inflammatory approaches (e.g., 5-aminosalicylic acid, corticosteroids, immunosuppressants, or monoclonal antibodies), but their clinical application has been restrained because of unsustainable therapeutic effect, the recurrence after drug withdrawal, and off-target systemic side effects [1]. Moreover, these medications are of little help in solving the intestinal fibrosis problem that is a complication of UC [12]. Therefore, the synergy of immune regulation and anti-fibrosis may be a new strategy for UC treatment.
To address this issue, we proposed a combination therapy strategy using an oral nanomedicine for co-delivering patchouli alcohol (PA) and simvastatin (SV), a “two-birds-one-stone” nanotherapeutic strategy. We previously revealed that patchouli alcohol, a natural tricyclic sesquiterpene isolated from a Chinese herb Guang Huo ** a safe and effective drug for UC. The interaction between the inflammatory immune microenvironment and colitis-related fibrosis during the progression of UC has not been fully demonstrated yet, and further investigation and understanding will be helpful to better depict the underlying mechanisms and seek effective drug combinations.
Availability of data and materials
All data generated or analyzed during this study are included in this published article and its Additional file.
Abbreviations
- MTT:
-
3-(4,5-Dimethyl-2-thiazolyl)-2,5-diphenyl-2-H-tetrazolium bromide
- CS:
-
Chitosan
- JNK:
-
C-Jun NH2 terminal kinase
- COX-2:
-
Cyclooxygenase-2
- DCs:
-
Dendritic cells
- DSS:
-
Dextran sodium sulfate
- DiR:
-
1,1-Dioctadecyl-3,3,3,3-tetramethylindotricarbocyaine iodide
- DL:
-
Drug-loading capacity
- DMEM:
-
Dulbecco’s modified Eagle’s medium
- EE:
-
Encapsulation efficiency
- ECM:
-
Extracellular matrix
- ERK1/2:
-
Extracellular signal-regulated kinase
- FBS:
-
Fetal bovine serum
- FITC:
-
Fluorescein isothiocyanate
- GC–FID:
-
Gas chromatography–flame ionization detector
- G-MDSC:
-
Granulocytic myeloid-derived suppressor cells
- H&E:
-
Hematoxylin/eosin
- HPLC:
-
High-performance liquid chromatography
- iNOS:
-
Inducible nitric oxide synthase
- IBD:
-
Inflammatory bowel disease
- IPM:
-
Inflammatory peritoneal macrophages
- IFN-γ:
-
Interferon γ
- IL-4:
-
Interleukin-4
- LPS:
-
Lipopolysaccharide
- M-CSF:
-
Macrophage colony-stimulating factor
- MAPK:
-
Mitogen-activated protein kinases
- NF-κB:
-
Nuclear factor-κB
- PA:
-
Patchouli alcohol
- PI3K:
-
Phosphatidylinositol-3 kinases
- PLGA:
-
Poly (lactic-co-glycolic acid)
- PDI:
-
Polydispersity index
- PVA:
-
Polyvinyl alcohol
- Akt:
-
Protein kinase B
- ROS:
-
Reactive oxygen species
- qPCR:
-
Real-time quantitative polymerase chain reaction
- Tregs:
-
Regulatory T cells
- SV:
-
Simvastatin
- CMC-Na:
-
Sodium carboxymethyl cellulose
- TGF-β:
-
Transforming growth factor-β
- TEM:
-
Transmission electron microscope
- TNF-α:
-
Tumor necrosis factor α
- UC:
-
Ulcerative colitis
- XRD:
-
X-ray diffractometer
References
Ungaro R, Mehandru S, Allen PB, Peyrin-Biroulet L, Colombel JF. Ulcerative colitis. Lancet. 2017;389:1756–70.
Ng SC, Shi HY, Hamidi N, Underwood FE, Tang W, Benchimol EI, Panaccione R, Ghosh S, Wu JCY, Chan FKL, et al. Worldwide incidence and prevalence of inflammatory bowel disease in the 21st century: a systematic review of population-based studies. Lancet. 2017;390:2769–78.
Kaplan GG, Ng SC. Globalisation of inflammatory bowel disease: perspectives from the evolution of inflammatory bowel disease in the UK and China. Lancet Gastroenterol Hepatol. 2016;1:307–16.
Bopanna S, Ananthakrishnan AN, Kedia S, Yajnik V, Ahuja V. Risk of colorectal cancer in Asian patients with ulcerative colitis: a systematic review and meta-analysis. Lancet Gastroenterol Hepatol. 2017;2:269–76.
Fukuda T, Naganuma M, Kanai T. Current new challenges in the management of ulcerative colitis. Intest Res. 2019;17:36–44.
de Souza HS, Fiocchi C. Immunopathogenesis of IBD: current state of the art. Nat Rev Gastroenterol Hepatol. 2016;13:13–27.
Eisenstein M. Gut reaction. Nature. 2018;563:S34–5.
Jones GR, Bain CC, Fenton TM, Kelly A, Brown SL, Ivens AC, Travis MA, Cook PC, MacDonald AS. Dynamics of colon monocyte and macrophage activation during colitis. Front Immunol. 2018;9:2764.
Liu YC, Zou XB, Chai YF, Yao YM. Macrophage polarization in inflammatory diseases. Int J Biol Sci. 2014;10:520–9.
Yang FC, Chiu PY, Chen Y, Mak TW, Chen NJ. TREM-1-dependent M1 macrophage polarization restores intestinal epithelium damaged by DSS-induced colitis by activating IL-22-producing innate lymphoid cells. J Biomed Sci. 2019;26:46.
Zhang J, Zhao Y, Hou T, Zeng H, Kalambhe D, Wang B, Shen X, Huang Y. Macrophage-based nanotherapeutic strategies in ulcerative colitis. J Control Release. 2020;320:363–80.
Wang J, Lin S, Brown JM, van Wagoner D, Fiocchi C, Rieder F. Novel mechanisms and clinical trial endpoints in intestinal fibrosis. Immunol Rev. 2021;302:211–27.
Zhao Y, Yang Y, Zhang J, Wang R, Cheng B, Kalambhe D, Wang Y, Gu Z, Chen D, Wang B, Huang Y. Lactoferrin-mediated macrophage targeting delivery and patchouli alcohol-based therapeutic strategy for inflammatory bowel diseases. Acta Pharm Sin B. 2020;10:1966–76.
Ikeda M, Takeshima F, Isomoto H, Shikuwa S, Mizuta Y, Ozono Y, Kohno S. Simvastatin attenuates trinitrobenzene sulfonic acid-induced colitis, but not oxazalone-induced colitis. Dig Dis Sci. 2008;53:1869–75.
Abe Y, Murano M, Murano N, Morita E, Inoue T, Kawakami K, Ishida K, Kuramoto T, Kakimoto K, Okada T, et al. Simvastatin attenuates intestinal fibrosis independent of the anti-inflammatory effect by promoting fibroblast/myofibroblast apoptosis in the regeneration/healing process from TNBS-induced colitis. Dig Dis Sci. 2012;57:335–44.
Rizvi F, Siddiqui R, DeFranco A, Homar P, Emelyanova L, Holmuhamedov E, Ross G, Tajik AJ, Jahangir A. Simvastatin reduces TGF-β1-induced SMAD2/3-dependent human ventricular fibroblasts differentiation: role of protein phosphatase activation. Int J Cardiol. 2018;270:228–36.
Liu W, Dong Z, Liu K, Lu Y, Wu W, Qi J, Chen Z. Targeting strategies of oral nano-delivery systems for treating inflammatory bowel disease. Int J Pharm. 2021;600:120461.
Arévalo-Pérez R, Maderuelo C, Lanao JM. Recent advances in colon drug delivery systems. J Control Release. 2020;327:703–24.
Chen SQ, Song YQ, Wang C, Tao S, Yu FY, Lou HY, Hu FQ, Yuan H. Chitosan-modified lipid nanodrug delivery system for the targeted and responsive treatment of ulcerative colitis. Carbohydr Polym. 2020;230:115613.
Joyce P, Wignall A, Peressin K, Wright L, Williams DB, Prestidge CA. Chitosan nanoparticles facilitate improved intestinal permeation and oral pharmacokinetics of the mast cell stabiliser cromoglycate. Int J Pharm. 2022;612:121382.
Cao X, Duan L, Hou H, Liu Y, Chen S, Zhang S, Liu Y, Wang C, Qi X, Liu N, et al. IGF-1C hydrogel improves the therapeutic effects of MSCs on colitis in mice through PGE(2)-mediated M2 macrophage polarization. Theranostics. 2020;10:7697–709.
Zhao P, Zhang J, Wu A, Zhang M, Zhao Y, Tang Y, Wang B, Chen T, Li F, Zhao Q, Huang Y. Biomimetic codelivery overcomes osimertinib-resistant NSCLC and brain metastasis via macrophage-mediated innate immunity. J Control Release. 2021;329:1249–61.
Zhang X, Ma Y, Ma L, Zu M, Song H, **ao B. Oral administration of chondroitin sulfate-functionalized nanoparticles for colonic macrophage-targeted drug delivery. Carbohydr Polym. 2019;223:115126.
Neudecker V, Haneklaus M, Jensen O, Khailova L, Masterson JC, Tye H, Biette K, Jedlicka P, Brodsky KS, Gerich ME, et al. Myeloid-derived miR-223 regulates intestinal inflammation via repression of the NLRP3 inflammasome. J Exp Med. 2017;214:1737–52.
Rieder F, Fiocchi C, Rogler G. Mechanisms, management, and treatment of fibrosis in patients with inflammatory bowel diseases. Gastroenterology. 2017;152:340–50.
Frangogiannis N. Transforming growth factor-β in tissue fibrosis. J Exp Med. 2020;217: e20190103.
Burke JP, Watson RW, Murphy M, Docherty NG, Coffey JC, O’Connell PR. Simvastatin impairs smad-3 phosphorylation and modulates transforming growth factor beta1-mediated activation of intestinal fibroblasts. Br J Surg. 2009;96:541–51.
Guo LT, Wang SQ, Su J, Xu LX, Ji ZY, Zhang RY, Zhao QW, Ma ZQ, Deng XY, Ma SP. Baicalin ameliorates neuroinflammation-induced depressive-like behavior through inhibition of toll-like receptor 4 expression via the PI3K/AKT/FoxO1 pathway. J Neuroinflamm. 2019;16:95.
Feng FB, Qiu HY. Effects of Artesunate on chondrocyte proliferation, apoptosis and autophagy through the PI3K/AKT/mTOR signaling pathway in rat models with rheumatoid arthritis. Biomed Pharmacother. 2018;102:1209–20.
Fresno Vara JA, Casado E, de Castro J, Cejas P, Belda-Iniesta C, González-Barón M. PI3K/Akt signalling pathway and cancer. Cancer Treat Rev. 2004;30:193–204.
Haque MA, Jantan I, Harikrishnan H, Ghazalee S. Standardized extract of Zingiber zerumbet suppresses LPS-induced pro-inflammatory responses through NF-κB, MAPK and PI3K-Akt signaling pathways in U937 macrophages. Phytomedicine. 2019;54:195–205.
Chen YL, Yan DY, Wu CY, Xuan JW, ** CQ, Hu XL, Bao GD, Bian YJ, Hu ZC, Shen ZH, Ni WF. Maslinic acid prevents IL-1β-induced inflammatory response in osteoarthritis via PI3K/AKT/NF-κB pathways. J Cell Physiol. 2021;236:1939–49.
Zhou F, Mei J, Han X, Li H, Yang S, Wang M, Chu L, Qiao H, Tang T. Kinsenoside attenuates osteoarthritis by repolarizing macrophages through inactivating NF-κB/MAPK signaling and protecting chondrocytes. Acta Pharm Sin B. 2019;9:973–85.
Zhou H, Ivanov VN, Lien YC, Davidson M, Hei TK. Mitochondrial function and nuclear factor-kappaB-mediated signaling in radiation-induced bystander effects. Cancer Res. 2008;68:2233–40.
Ju L, Hu P, Chen P, Xue X, Li Z, He F, Qiu Z, Cheng J, Huang F. Huoxuezhitong capsule ameliorates MIA-induced osteoarthritis of rats through suppressing PI3K/Akt/NF-κB pathway. Biomed Pharmacother. 2020;129:110471.
Han W, **e B, Li Y, Shi L, Wan J, Chen X, Wang H. Orally deliverable nanotherapeutics for the synergistic treatment of colitis-associated colorectal cancer. Theranostics. 2019;9:7458–73.
Wang Q, Zhao Y, Guan L, Zhang Y, Dang Q, Dong P, Li J, Liang X. Preparation of astaxanthin-loaded DNA/chitosan nanoparticles for improved cellular uptake and antioxidation capability. Food Chem. 2017;227:9–15.
Du X, Wu Z, Xu Y, Liu Y, Liu W, Wang T, Li C, Zhang C, Yi F, Gao L, et al. Increased Tim-3 expression alleviates liver injury by regulating macrophage activation in MCD-induced NASH mice. Cell Mol Immunol. 2019;16:878–86.
Bauer C, Duewell P, Mayer C, Lehr HA, Fitzgerald KA, Dauer M, Tschopp J, Endres S, Latz E, Schnurr M. Colitis induced in mice with dextran sulfate sodium (DSS) is mediated by the NLRP3 inflammasome. Gut. 2010;59:1192–9.
Johansson ME, Gustafsson JK, Holmén-Larsson J, Jabbar KS, **a L, Xu H, Ghishan FK, Carvalho FA, Gewirtz AT, Sjövall H, Hansson GC. Bacteria penetrate the normally impenetrable inner colon mucus layer in both murine colitis models and patients with ulcerative colitis. Gut. 2014;63:281–91.
Kotla NG, Isa ILM, Rasala S, Demir S, Singh R, Baby BV, Swamy SK, Dockery P, Jala VR, Rochev Y, Pandit A. Modulation of gut barrier functions in ulcerative colitis by hyaluronic acid system. Adv Sci. 2021;9: e2103189.
Chang J, Leong RW, Wasinger VC, Ip M, Yang M, Phan TG. Impaired intestinal permeability contributes to ongoing bowel symptoms in patients with inflammatory bowel disease and mucosal healing. Gastroenterology. 2017;153:723–31.
Sharma A, Tirpude NV, Kumari M, Padwad Y. Rutin prevents inflammation-associated colon damage via inhibiting the p38/MAPKAPK2 and PI3K/Akt/GSK3β/NF-κB signalling axes and enhancing splenic Tregs in DSS-induced murine chronic colitis. Food Funct. 2021;12:8492–506.
Gordon IO, Agrawal N, Willis E, Goldblum JR, Lopez R, Allende D, Liu X, Patil DY, Yerian L, El-Khider F, et al. Fibrosis in ulcerative colitis is directly linked to severity and chronicity of mucosal inflammation. Aliment Pharmacol Ther. 2018;47:922–39.
Boland BS, He Z, Tsai MS, Olvera JG, Omilusik KD, Duong HG, Kim ES, Limary AE, ** W, Milner JJ, et al. Heterogeneity and clonal relationships of adaptive immune cells in ulcerative colitis revealed by single-cell analyses. Sci Immunol. 2020;5: eabb4432.
Brazil JC, Louis NA, Parkos CA. The role of polymorphonuclear leukocyte trafficking in the perpetuation of inflammation during inflammatory bowel disease. Inflamm Bowel Dis. 2013;19:1556–65.
Yamamoto S, Shimizu S, Kiyonaka S, Takahashi N, Wajima T, Hara Y, Negoro T, Hiroi T, Kiuchi Y, Okada T, et al. TRPM2-mediated Ca2+ influx induces chemokine production in monocytes that aggravates inflammatory neutrophil infiltration. Nat Med. 2008;14:738–47.
Acknowledgements
We thank the Molecular Imaging Center and TEM Facility at SIMM and the National Center for Protein Science Shanghai, CAS for the technical support.
Funding
National Key Research and Development Program of China (2021YFE0103100, 2021YFC2400600); NFSC (81925035, 8201101172, and 81803736); Shanghai SciTech Innovation Initiative (19431903100, 18430740800).
Author information
Authors and Affiliations
Contributions
JZ: investigation, methodology, data curation, formal analysis, visualization, writing—original draft. AO: investigation, validation, formal analysis, data curation. XT, RW, YZ, PZ: investigation, resources. YF: investigation, validation. YF, DC: investigation, data curation. BW: project administration, supervision. YH: conceptualization, project administration, formal analysis, writing—review and editing. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
All the animal experimental procedures were complied with the institutional ethical guidelines and approved by the Institutional Animal Care and Use Committee (IACUC), Shanghai Institute of Materia Medica, Chinese Academy of Sciences (IACUC No. SYXK2015-0027).
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1: Figure S1.
Effect of PA on macrophage repolarization and synergistic effect with SV. (A–C) The mRNA levels of M1-associated pro-inflammatory cytokines (e.g., IL-1β, IL-6, and TNF-α) in PA-treated RAW264.7 macrophages, as measured by qPCR. (D, E) Western blot analysis of Akt/MAPK/NF-κB pathway-related biomarkers and M2-related MR expression after PA treatment. (F) The mRNA levels of the M1-related pro-inflammatory molecules (e.g., TNF-α, iNOS, and COX-2) and M2-related Arg1 in SV-treated RAW264.7 macrophages, as measured by qPCR. (G) IL-6 mRNA levels in LPS-induced peritoneal macrophages treated with PA (10 μM) and SV (0, 1, and 2 μM). Figure S2. Cytotoxicity study of PA and SV on (A) Caco-2 cells, (B) RAW264.7 cells, and (C) L929 cells. Cytotoxicity of the NPs in (D) L929 cells and (E) M2Φ. Figure S3. Anti-colitis treatment of synergistic drugs. (A) Schematic diagram of DSS-induced colitis and treatment. (B) Changes in daily bodyweight of each group during the trial period. (C) Statistical analysis and (D) images of colon lengths in each group (n = 4). Figure S4. Preliminary biosafety assessment of PA and SV. (A) Organ coefficients. (B) H&E staining of the major organs (Scale bar: 100 μm). Figure S5. Fluorescence images of (A) M1Φ and (B) L929 after incubation with the coumarin 6-labeled NPs (scale bar: 50 µm). (C, E) Histogram and (D, F) mean fluorescence intensity of the NPs-internalized M1Φ (LPS-induced RAW264.7 cells) and L929 cells were analyzed by flow cytometry (n = 3). Figure S6. Specific accumulation of CS-PLGA NPs in inflamed colons. Ex vivo imaging and radiant efficiency of (A, B) organs and (C) colons at 3 h. Ex vivo imaging and radiant efficiency of (D, E) organs and (F) colons at 5 h (n = 3). Figure S7. (A–E) Individual bodyweight curves and (F–J) DAI curves in CMC-Na, DSS, PA/SV, PLGA NPs, or CS-PLGA NPs groups (n = 6). Figure S8. Preliminary biosafety assessment. (A) Organ coefficients. (B) H&E staining of the major organs (Scale bar: 100 μm). Figure S9. The dot plots of M2Φ and DCs in the colon tissue. Figure S10. The dot plots of neutrophils, inflammatory monocytes, and G-MDSCs in the colon tissue. Figure S11. The dot plots of Tregs in the colon tissue. Table S1. The primer sequence used in qPCR. Table S2. Disease activity index (DAI) scoring. Table S3. Characterization of the NPs. Table S4. Drug encapsulation efficiency and drug loading efficacy.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Zhang, J., Ou, A., Tang, X. et al. “Two-birds-one-stone” colon-targeted nanomedicine treats ulcerative colitis via remodeling immune microenvironment and anti-fibrosis. J Nanobiotechnol 20, 389 (2022). https://doi.org/10.1186/s12951-022-01598-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12951-022-01598-0