Abstract
Objective
The aim of this research was to examine how penehyclidine hydrochloride (PHC) impacts the occurrence of pyroptosis in lung tissue cells within a rat model of lung ischemia-reperfusion injury.
Methods
Twenty-four Sprague Dawley (SD) rats, weighing 250 g to 270 g, were randomly distributed into three distinct groups as outlined below: a sham operation group (S group), a control group (C group), and a test group (PHC group). Rats in the PHC group received a preliminary intravenous injection of PHC at a dose of 3 mg/kg. At the conclusion of the experiment, lung tissue and blood samples were collected and properly stored for subsequent analysis. The levels of malondialdehyde, superoxide dismutase, and myeloperoxidase in the lung tissue, as well as IL-18 and IL-1β in the blood serum, were assessed using an Elisa kit. Pyroptosis-related proteins, including Caspase1 p20, GSDMD-N, and NLRP3, were detected through the western blot method. Additionally, the dry-to-wet ratio (D/W) of the lung tissue and the findings from the blood gas analysis were also documented.
Results
In contrast to the control group, the PHC group showed enhancements in oxygenation metrics, reductions in oxidative stress and inflammatory reactions, and a decrease in lung injury. Additionally, the PHC group exhibited lowered levels of pyroptosis-associated proteins, including the N-terminal segment of gasdermin D (GSDMD-N), caspase-1p20, and nucleotide-binding oligomerization domain-like receptor protein 3 (NLRP3).
Conclusion
Pre-administration of PHC has the potential to mitigate lung ischemia-reperfusion injuries by suppressing the pyroptosis of lung tissue cells, diminishing inflammatory reactions, and enhancing lung function. The primary mechanism behind anti-pyroptotic effect of PHC appears to involve the inhibition of oxidative stress.
Similar content being viewed by others
Introduction
Lung ischemia-reperfusion injury (IRI), a type of aseptic acute lung injury, is a common surgical complication following single-lung ventilation [1, 2], open-heart surgery [3], trauma [4], and lung transplantation [Rat lung ischemia-reperfusion model All the rats were rendered unconscious through an intraperitoneal injection of 5 mg/100 g of pentobarbital sodium. Once they were adequately anesthetized, they were placed on an insulating blanket and had a tracheotomy performed for intubation. A tracheal tube was then connected to a small animal ventilator (Rayward, R415, Shenzhen, China) and ventilated with 100% oxygen. The tidal volume used was 6–8 ml per kilogram of body weight, following a volume-control mode, at a rate of 60–80 breaths per minute, with an inhalation-to-exhalation ratio of 1:2. After 30 min of intravenous administration of PHC (3 mg/kg) [13] through the femoral vein, the rats underwent a left thoracotomy, specifically during the expiratory phase, through the fourth intercostal space. Ten minutes later, following intravenous injection of sodium heparin (200 U/kg), the left hilum structure was carefully clamped using a non-invasive vascular clamp. After a duration of 45 min, the vascular clamp was removed, and pulmonary blood perfusion was maintained for a period of 2 h. Throughout the entire experiment, normal saline was continuously administered through the femoral vein at a rate of 10 ml per kilogram per hour. The blood pressure and temperature were consistently monitored using the Mindray iPM10 equipment from Shenzhen, China. The model rats were euthanized by exsanguination 2 h after reperfusion. Lung tissues and serum samples were collected for subsequent testing. A total volume of 1 ml of arterial blood was collected from the femoral artery. Blood gas analysis was conducted at two time points: before the thoracotomy (T0) and either at the conclusion of the experiment or after a 2-hour reperfusion period (T2). This analysis was carried out using the OPTI CCA-TS Blood Gas and Electrolyte Analyzer from the United States. Subsequently, the oxygenation index (PaO2/FiO2) was calculated to assess pulmonary function. After concluding the experiment, approximately 2–4 ml of blood were obtained from the femoral artery and subsequently spun at 2000 rpm at 4 °C for 15 min. The resulting supernatant was collected and preserved at -80 °C. The lung tissue was extracted, underwent a brief rinse with a cold (4°C) PBS solution, and was then subdivided into multiple smaller segments using a sharp blade. Some of these segments were promptly placed in a -80 °C freezer after pre-cooling with liquid nitrogen, while others were designated for pathological analysis and determination of the wet/dry weight ratio (W/D). To assess pulmonary edema, the process involved initially using absorbent paper to remove surface moisture from harvested upper lobe lung tissue. This tissue was then promptly weighed to determine its wet weight. Subsequently, it was placed in a 70 °C oven and subjected to a 72-hour baking period to ascertain the dry weight. Ultimately, the W/D weight ratio was computed as a means of assessing pulmonary edema. In summary, the lung tissue underwent a 72-hour immersion in a 4% paraformaldehyde solution, followed by embedding in paraffin and slicing into 4 μm-thick sections. These sections were subsequently stained with hematoxylin and eosin. A pathologist, unaware of the experimental design, examined the histological changes in the pulmonary tissue under a microscope and recorded the lung injury score. The parameters assessed included pulmonary interstitial edema, neutrophil infiltration, alveolar edema, and alveolar congestion [21]. The lung injury scores (LIS) were categorized as: 0 = normal or very mild, 1 = mild, 2 = moderate, 3 = severe. The concentrations of interleukin (IL)-1β and IL-18 in the serum were measured using enzyme-linked immunosorbent assay (ELISA) kits (Merck Bell Biotechnology, Wuhan, China) following the manufacturer’s provided instructions. Myeloperoxidase (MPO) function, the malondialdehyde (MDA) concentration, and superoxide dismutase (SOD) performance in lung tissue were assessed using a dedicated kit from Merck Bell Biotechnology (Wuhan, China). All assessments were conducted following the provided instructions from the manufacturer. In accordance with the manufacturer’s guidelines, lung tissue homogenates were processed for total protein extraction using RIPA lysis buffer (Beyotime Biotechnology, Shanghai, China). The resulting mixture was then subjected to centrifugation at 12,000 rpm at 4 °C for 15 min using a Sigma 3–39 K centrifuge (Germany). The concentration of total proteins was determined by employing a BCA kit (Beyotime Biotechnology, Shanghai, China) through the bicinchoninic acid method. Subsequently, the protein samples were obtained, underwent electrophoresis on a 10% sodium dodecyl sulfate-polyacrylamide gel (SDS-PAGE), and were transferred onto polyvinylidene fluoride (PVDF) membranes. These membranes were subsequently blocked with a 5% stacking gel at room temperature for 40 min. Following this, the membranes were subjected to overnight incubation with primary antibodies targeting NLRP3 (1:1,000, Abcam, Cambridge, USA), GSDMD-N (1:1,000, Cell Signaling Technology, USA), cysteinyl aspartate-specific proteinase (caspase-1) p20 (1:1,000, Biorigin, Bei**g, China), and anti-β-actin antibody at 4 °C. After overnight incubation, the membranes were treated with a secondary antibody (1:300, Merck, Wuhan, China) following three washes at room temperature. Finally, the immunological complexes were analyzed using ECL analysis kits (Beyotime Biotechnology, Shanghai, China), and the protein expression levels were quantified using image software. Transmission electron microscopy (TEM). To observe the morphology of type II alveolar epithelial cells, an expert who was blind to the study design made an electron microscope section of the lung tissue as soon as possible. The lung tissue was washed with 4 °C PBS solution, cut into small pieces (1 mm3), and fixed in 2.5% glutaraldehyde for 2 h at room temperature. Dates are presented as mean ± standard deviation. Statistical analyses were carried out using IBM SPSS Statistics 20.0 software. Group differences were assessed using a combination of one-way analysis of variance (ANOVA) with the Student’s t-test, and within-group comparisons were made using repeated measures ANOVA. A significance level of P < 0.05 was employed to determine statistical significance.Blood gas analysis
Sample processing and collection
Pulmonary tissue wet/dry weight ratio (W/D)
Histopathologic analysis of lung tissue
Levels of inflammatory factors in serum
Detection of oxidative stress in lung tissue
Detection of pyroptosis-related proteins
Statistical analysis
Results
Blood gas analysis
We employed the ratio of the fraction of inspired oxygen (FiO2) to the partial pressure of arterial oxygen (PaO2) as an indicator to assess the function of the lungs’ oxygenation system. PaO2/FiO2 was comparatively constant in the sham group. The PaO2/FiO2 in the sham group (428 ± 22.41 mmHg) was significantly higher than that in the control group (302 ± 30.49 mmHg) at the conclusion of the experiment, while the PaO2/FiO2 in the PHC group (343 ± 32.83 mmHg) was significantly higher than that in the control group (P < 0.05) (Table 1).
Inflammatoryresponse
While the W/D ratio of the lung tissue in the PHC group (5.2 ± 0.4) was lower than that in the control group (6.4 ± 0.5), it was higher than that in the sham group (4.1 ± 0.3). The serum concentrations of IL-18 and IL-1 as well as the lung tissue’s MPO activity showed a tendency that was similar to the W/D (P < 0.05) (Table 1).
Oxidative stress response
The MDA levels in lung tissue in the PHC group (5.22 ± 1.09 mmol/l) and sham group (3.42 ± 0.46 mmol/l) were lower than those in the control group (7.73 ± 1.23 mmol/l) (P < 0.05) compared to the control group. Additionally, the MPO levels in lung tissue were greater in the PHC group than in the sham group. In contrast to MDA levels, SOD activity in lung tissue displayed the reverse direction (Table 1).
Lung injury scores
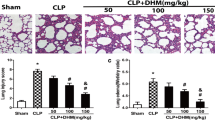
The sham group exhibited minimal pathological alterations in lung tissue due to the surgical procedure, while the control group displayed a substantial presence of neutrophil infiltration, intra-alveolar hemorrhage, and severe interstitial edema. In comparison to the control group, the PHC group demonstrated fewer pathological changes in lung tissue. The degree of neutrophil infiltration, as indicated by the LIS, was significantly higher in the control group [2.5(2–3)] than in the sham group [0.5(0–1) (P < 0.05), and in the PHC group [1.5(1–2)] (P < 0.05) (see Fig. 1).
Paraformaldehyde-fixed sections of lung tissues were stained with eosin and hematoxylin, and all pictures represent 40x original magnifications, the scale bar represents 100 μm (black). (A) sham group; (B) control group; (C) PHC group; (D) Lung injury score. *P < 0.05 vs. sham group, #P < 0.05 vs. control group
Pyroptosis-related protein
Caspase-1 p20 levels in the lung tissues were lower in the sham (0.42 ± 0.2) and PHC groups (0.69 ± 0.75) compared to the control group (0.96 ± 0.51) (P < 0.05). Similar trends were seen in the levels of NLRP3 and GSDMD-N as well as Caspase-1 p20 (Fig. 2).
Electron microscopy analysis of morphological changes in type II alveolar epithelial cells.
Ultrastructural alterations in lung tissue were examined using TEM. In the control group, microvilli displayed a slight reduction in length, with certain sections even experiencing a decrease or disappearance. Epithelium villi and lamellar corpuscles exhibited a decline, while swollen mitochondria and the merging of crista membranes were notably prominent. Additionally, the rough endoplasmic reticulum expanded, degranulation was observed, the perinuclear gap widened, and nuclear pyknosis occurred. Comparatively, fewer alterations were observed in the PHC group. However, in the sham group, type II alveolar epithelial cells resembled normal structures with minimal changes. Refer to Fig. 3.
Type II alveolar epithelial cell morphology was imaged with electron microscopy. (Control group), (PHC group), (Sham group), (original magnification, 10,000). As it was shown in the above pictures: in sham group, the type II epithelial alveolar cells were almost normal, villi and lamellar corpuscles were observed easily, with no obvious swelling or destruction of mitochondria. In the control group, the microvilli were slightly shorter, with some parts of the microvilli having reduced or even disappeared. There was a decrease in epithelium villi and lamellar corpuscles, while swollen mitochondria and crista membrane fusion were significant. Additionally, rough endoplasmic reticulum expanded, degranulation occurred, the perinuclear gap was wide and nuclear pyknosis occured, means the lung injury was severe in this group Compared with the control group in the PHC group, there were more epithelium villi and lamellar corpuscles, mitochondria swelling was slighter, crista membrane fusion was decreased and the perinuclear space widen slightly
Discussion
IRI is a pathological condition that arises when endogenous ligands are released [22]. This process occurs in tissues subjected to varying degrees of hypoxia and ischemia caused by various factors. Following the restoration of blood flow, organs and tissues undergo significant oxidative stress, resulting in changes in protein expression, disruptions in cellular metabolism, and structural injury [23]. Consequently, tissue damage is worsened, and in some cases, it becomes irreversible [24]. Lung ischemia-reperfusion injury is a common event during surgical procedures, especially in lung transplantation and cardiothoracic surgery. Nevertheless, the exact underlying mechanism remains unclear. Several theories propose that lung IRI may be linked to cell apoptosis [25], cell autophagy [26], and pyroptosis [8]. In this study, we demonstrated that the administration of PHC prior to ischemia can mitigate the development of lung injury caused by ischemia-reperfusion. This protective effect is associated with the prevention of pyroptosis, which is defined as gasdermin-mediated programmed necrosis and is linked to innate immunity and various diseases [27]. Research has shown a close connection between pyroptosis and conditions such as cardiovascular diseases [28] and tumors [29]. Recent evidence underscores the critical role of pyroptosis in host defense and its primary role in bridging innate and adaptive immunity [30]. However, excessive pyroptosis can lead to prolonged and heightened inflammatory responses, thereby contributing to the progression of inflammatory disorders [31].
Consequently, pyroptosis is a two-edged sword and has been observed in various types of cells, including those in the digestive system, reproductive system, cardiovascular system, and central nervous system [32]. Notably, some scholars have also confirmed the involvement of pyroptosis in the mechanism of pulmonary ischemia-reperfusion, aligning with our experimental findings. In the control group, there was an increase in the expression of proteins associated with pyronecrosis, such as NLRP3, caspase-1 p20, and GSDMD-N. Conversely, the expression of pyroptosis-related proteins in the sham group was minimal, and in the PHC group, it fell between the levels observed in the sham and control groups. This suggests that the protective effect of PHC on lung IRI may be linked to the regulation of pyroptosis. Pyroptosis represents an inflammatory type of programmed cell death characterized by cell membrane rupture and a robust inflammatory reaction. In a rat lung ischemia-reperfusion model, levels of pyroptosis-related proteins reached their highest point 2 h after reperfusion [33], which is why a 2-hour reperfusion period was selected for this study. The expression of pyroptosis-related proteins demonstrated that lung IRI is associated with pyroptosis mediated by the NLRP3 inflammasome. There is emerging evidence indicating that PHC offers protection against injuries caused by ischemia-reperfusion in various organs. These include acute lung injury resulting from renal ischemia-reperfusion and acute cerebral ischemia-reperfusion injury. These protective effects are attributed to the activation of the Nrf2 pathway [34] and the suppression of the JNK/p38MAPK signaling pathway [35]. PHC improves conditions related to oxidative stress, apoptosis, and inflammatory responses, effectively preventing renal ischemia-reperfusion [36]. Moreover, PHC elevates PDGF-B levels to activate the PI3K pathway in cells, ameliorating myocardial ischemia-reperfusion injury [37].
Furthermore, PHC also exhibits a certain preventive and protective effect against various types of lung injuries. In blunt chest trauma and hemorrhagic shock-induced acute lung injury, PHC plays a crucial role in preventing TLR4 signaling and inflammation [38]. Additionally, PHC has been demonstrated to be beneficial in cases of lung injury resulting from cecal ligation by enhancing microvascular permeability [39]. Research has also shown that PHC can alleviate apoptosis and endoplasmic reticulum stress [40] and trigger the Hes1/Notch1 pathway [41], effectively mitigating the effects of lipopolysaccharide-induced acute lung injury.
Chen et al. speculated that PHC may effectively alleviate lung injury resulting from mechanical ventilation in rats with COPD. The potential mechanism for this process may be related to the JNK/SAPK signaling pathway [19]. In our study, we discovered that pyroptosis is another potential mechanism underlying the protective effect of PHC against lung ischemia-reperfusion injury. Wang et al. [13] determined that the optimal dose of PHC pretreatment in a rat lung ischemia-reperfusion model is 3 mg/kg. Therefore, we used 3 mg/kg of PHC in our experiment and obtained similar results, confirming the significant protective effect of this dosage against lung ischemia-reperfusion injury. Additionally, PHC offers relief from pulmonary injury associated with cardiopulmonary bypass by diminishing the inflammatory response and oxidative stress reaction [42]. In this investigation, prior treatment with PHC demonstrated a significant reduction in inflammation markers, such as IL-18 and IL-1β levels in the bloodstream, MPO activity in lung tissue, and the W/D ratio of the lung.
Moreover, PHC effectively counteracts oxidative stress by reducing MDA activity and enhancing SOD activity, suggesting its potential to exert anti-inflammatory and antioxidant effects by decreasing inflammatory factors and increasing reactive oxygen species activity. These protective properties of PHC imply its capacity to shield against organ ischemia-reperfusion injury.
Nonetheless, our study has several limitations. First, we only examined the injury following a two-hour reperfusion period, necessitating further investigation into the long-term effects of PHC on lung IRI. Second, our observation of pyroptosis in pulmonary ischemia-reperfusion highlights the need for further exploration of the underlying mechanisms, potentially through in vitro cell culture experiments. Third, due to the small sample size and brief reperfusion duration, we were unable to fully record animal mortality rates, leaving uncertainty regarding the potential benefits of PHC in mitigating pulmonary ischemia-reperfusion injury-related mortality.
Conclusion
Preconditioning with PHC can mitigate lung IRI, as it hinders the pyroptosis of lung tissue cells, diminishes the inflammatory response, and enhances lung function while primarily relying on the inhibition of oxidative stress as its key antipyroptotic mechanism.
Data availability
The datasets used and analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- PHC:
-
Penehyclidine hydrochloride
- LIS:
-
Lung injury score
- LPS:
-
Lipopolysaccharides
- COPD:
-
Chronic obstructive pulmonary disease
References
Lu D, Wang Z, Chen Z, Fan J, Zhai J, Fang D, Cai H, Liu X, Wu H, Cai K. Olmesartan attenuates single-lung Ventilation Induced Lung Injury via regulating pulmonary microbiota. Front Pharmacol. 2022;13:822615. https://doi.org/10.3389/fphar.2022.822615. PMID: 35401192; PMCID: PMC8984607.
Healthcare Engineering JO, Retracted. Effect of different doses of propofol on pulmonary function and inflammatory response in patients with Lung Ischemia Reperfusion Injury Induced by one-lung ventilation based on Big Data Analysis. J Healthc Eng. 2023;2023:9859680. https://doi.org/10.1155/2023/9859680. PMID: 37476778; PMCID: PMC10356442.
Zheng XM, Yang Z, Yang GL, Huang Y, Peng JR, Wu MJ. Lung injury after cardiopulmonary bypass: alternative treatment prospects. World J Clin Cases. 2022;10(3):753–61. https://doi.org/10.12998/wjcc.v10.i3.753. PMID: 35127892; PMCID: PMC8790450.
Wang Y, Wang C, Zhang D, Wang H, Bo L, Deng X. Dexmedetomidine protects against traumatic Brain Injury-Induced Acute Lung Injury in mice. Med Sci Monit. 2018;24:4961–7. https://doi.org/10.12659/MSM.908133. PMID: 30013022; PMCID: PMC6067036.
Liu R, Fang X, Meng C, **ng J, Liu J, Yang W, Li W, Zhou H. Lung inflation with hydrogen during the cold ischemia phase decreases lung graft injury in rats. Exp Biol Med (Maywood). 2015;240(9):1214–22. https://doi.org/10.1177/1535370214563895. Epub 2015 Feb 7. PMID: 25662956; PMCID: PMC4935368.
Liu S, Zhang J, Yu B, Huang L, Dai B, Liu J, Tang J. The role of autophagy in lung ischemia/reperfusion injury after lung transplantation in rats. Am J Transl Res. 2016;8(8):3593–602. PMID: 27648150; PMCID: PMC5009412.
Dong L, Liang F, Lou Z, Li Y, Li J, Chen Y, Ding J, Jiang B, Wu C, Yu H, Liu Y, Zhang W, Lu Y, Wu M. Necrostatin-1 alleviates Lung Ischemia-Reperfusion Injury via inhibiting necroptosis and apoptosis of lung epithelial cells. Cells. 2022;11(19):3139. https://doi.org/10.3390/cells11193139. PMID: 36231101; PMCID: PMC9563441.
Zheng P, Kang J, **ng E, Zheng B, Wang X, Zhou H. Lung inflation with Hydrogen during the Cold Ischemia Phase alleviates Lung Ischemia-Reperfusion Injury by inhibiting pyroptosis in rats. Front Physiol. 2021;12:699344. https://doi.org/10.3389/fphys.2021.699344. PMID: 34408660; PMCID: PMC8365359.
Wang X, Zhang B, Li G, Zhao H, Tian X, Yu J, Yin Y, Meng C. Dexmedetomidine alleviates lung oxidative stress Injury Induced by Ischemia-Reperfusion in Diabetic rats via the Nrf2-Sulfiredoxin1 pathway. Biomed Res Int. 2022;2022:5584733. https://doi.org/10.1155/2022/5584733. PMID: 35252452; PMCID: PMC8894003.
Hong H, Huang Q, Cai Y, Lin T, **a F, ** Z. Dexmedetomidine preconditioning ameliorates lung injury induced by pulmonary ischemia/reperfusion by upregulating promoter histone H3K4me3 modification of KGF-2. Exp Cell Res. 2021;406(2):112762. Epub 2021 Aug 2. PMID: 34352276.
Wang G, Li JY, Weng YQ, Ding M, Yu HL, Wang Q, Ren HC, Xu RB, Yu WL. Protective effect of ulinastatin combined with dexmedetomidine on lung injury after cold ischemia-reperfusion in rats. Eur Rev Med Pharmacol Sci. 2018;22(17):5712–5718. https://doi.org/10.26355/eurrev_201809_15839. PMID: 30229849.
Saito M, Chen-Yoshikawa TF, Suetsugu K, Okabe R, Takahagi A, Masuda S, Date H. Pirfenidone alleviates lung ischemia-reperfusion injury in a rat model. J Thorac Cardiovasc Surg. 2019;158(1):289–96. Epub 2018 Sep 28. PMID: 30385019.
Wang Y, Lin D, Tan H, Gao Y, Ma J. Penehyclidine hydrochloride preconditioning provides pulmonary and systemic protection in a rat model of lung ischaemia reperfusion injury. Eur J Pharmacol. 2018;839:1–11. Epub 2018 Sep 7. PMID: 30201378.
Sun X, Sun C, Zhai L, Dong W. A selective M1 and M3 receptor antagonist, Penehyclidine Hydrochloride, exerts antidepressant-like Effect in mice. Neurochem Res. 2019;44(12):2723–32. https://doi.org/10.1007/s11064-019-02891-5. Epub 2019 Oct 12. PMID: 31606838.
Kang Y, Li Y, Wen H, Zhu J, Zheng J, Feng Z. Prevention of renal ischemia and reperfusion injury by penehyclidine hydrochloride through autophagy activation. Mol Med Rep. 2020;21(5):2182–92. https://doi.org/10.3892/mmr.2020.11024. Epub 2020 Mar 12. PMID: 32186764; PMCID: PMC7115187.
Wang D, Jiang Q, Du X. Protective effects of scopolamine and penehyclidine hydrochloride on acute cerebral ischemia-reperfusion injury after cardiopulmonary resuscitation and effects on cytokines. Exp Ther Med. 2018;15(2):2027–31. https://doi.org/10.3892/etm.2017.5646. Epub 2017 Dec 15. PMID: 29434800; PMCID: PMC5776622.
Kong Q, Wu X, Duan W, Zhan L, Song X. Penehyclidine hydrochloride exerts protective effects in rats with acute lung injury via the Fas/FasL signaling pathway. Exp Ther Med. 2019;17(5):3598–606. https://doi.org/10.3892/etm.2019.7340. Epub 2019 Mar 4. PMID: 30988742; PMCID: PMC6447765.
Feng M, Wang L, Chang S, Yuan P. Penehyclidine hydrochloride regulates mitochondrial dynamics and apoptosis through p38MAPK and JNK signal pathways and provides cardioprotection in rats with myocardial ischemia-reperfusion injury. Eur J Pharm Sci. 2018;121:243–50. https://doi.org/10.1016/j.ejps.2018.05.023. Epub 2018 May 31. PMID: 29860115.
Chen ZY, Zhang Y, Wu JH, Gao XH, Huang CL, Lin YM, Xu XT, Li Y. The Mechanism of Penehyclidine Hydrochloride and Its Effect on the Inflammatory Response of Lung Tissue in Rats with Chronic Obstructive Pulmonary Disease During Mechanical Ventilation. Int J Chron Obstruct Pulmon Dis. 2021;16:877–885. https://doi.org/10.2147/COPD.S295329. Retraction in: Int J Chron Obstruct Pulmon Dis. 2023;18:1003–1004. PMID: 33833508; PMCID: PMC8020330.
Zheng F, **ao F, Yuan QH, Liu QS, Zhang ZZ, Wang YL, Zhan J. Penehyclidine Hydrochloride Decreases Pulmonary Microvascular Endothelial Inflammatory Injury Through a Beta-Arrestin-1-Dependent Mechanism. Inflammation. 2018;41(5):1610–1620. https://doi.org/10.1007/s10753-018-0804-9. PMID: 29766401.
Kozian A, Schilling T, Fredén F, Maripuu E, Röcken C, Strang C, Hachenberg T, Hedenstierna G. One-lung ventilation induces hyperperfusion and alveolar damage in the ventilated lung: an experimental study. Br J Anaesth. 2008;100(4):549–59. https://doi.org/10.1093/bja/aen021. Epub 2008 Feb 27. PMID: 18308740.
Li W, Terada Y, Tyurina YY, Tyurin VA, Bery AI, Gauthier JM, Higashikubo R, Tong AY, Zhou D, Nunez-Santana F, Lecuona E, Hassan A, Hashimoto K, Scozzi D, Puri V, Nava RG, Krupnick AS, Lavine KJ, Gelman AE, Miller MJ, Kagan VE, Bharat A, Kreisel D. Necroptosis triggers spatially restricted neutrophil-mediated vascular damage during lung ischemia reperfusion injury. Proc Natl Acad Sci U S A. 2022;119(10):e2111537119. https://doi.org/10.1073/pnas.2111537119. Epub 2022 Mar 1. PMID: 35238643; PMCID: PMC8917381.
Sun Y, Zhang T, Zhang Y, Li J, ** L, Sun Y, Shi N, Liu K, Sun X. Ischemic postconditioning alleviates cerebral ischemia-reperfusion Injury through activating Autophagy during early reperfusion in rats. Neurochem Res. 2018;43(9):1826–40. https://doi.org/10.1007/s11064-018-2599-3. Epub 2018 Jul 25. PMID: 30046966; PMCID: PMC6096887.
Mariscal A, Caldarone L, Tikkanen J, Nakajima D, Chen M, Yeung J, Cypel M, Liu M, Keshavjee S. Pig lung transplant survival model. Nat Protoc. 2018;13(8):1814–1828. https://doi.org/10.1038/s41596-018-0019-4. PMID: 30072720.
Dong L, Yin L, Li R, Xu L, Xu Y, Han X, Qi Y. Dioscin alleviates lung ischemia/reperfusion injury by regulating FXR-mediated oxidative stress, apoptosis, and inflammation. Eur J Pharmacol. 2021;908:174321. https://doi.org/10.1016/j.ejphar.2021.174321. Epub 2021 Jul 9. PMID: 34252440.
Zhang W, Zhang J. Dexmedetomidine preconditioning protects against lung injury induced by ischemia-reperfusion through inhibition of autophagy. Exp Ther Med. 2017;14(2):973–80. https://doi.org/10.3892/etm.2017.4623. Epub 2017 Jun 16. PMID: 28810549; PMCID: PMC5526121.
Man SM, Karki R, Kanneganti TD. Molecular mechanisms and functions of pyroptosis, inflammatory caspases and inflammasomes in infectious diseases. Immunol Rev. 2017;277(1):61–75. https://doi.org/10.1111/imr.12534. PMID: 28462526; PMCID: PMC5416822.
Jia C, Chen H, Zhang J, Zhou K, Zhuge Y, Niu C, Qiu J, Rong X, Shi Z, **ao J, Shi Y, Chu M. Role of pyroptosis in cardiovascular diseases. Int Immunopharmacol. 2019;67:311–8. Epub 2018 Dec 17. PMID: 30572256.
Pezuk JA. Pyroptosis in combinatorial treatment to improve cancer patients’ outcome, is that what we want? EBioMedicine. 2019;41:17–8. https://doi.org/10.1016/j.ebiom.2019.03.007. Epub 2019 Mar 14. PMID: 30879924; PMCID: PMC6443691.
Hsu SK, Li CY, Lin IL, Syue WJ, Chen YF, Cheng KC, Teng YN, Lin YH, Yen CH, Chiu CC. Inflammation-related pyroptosis, a novel programmed cell death pathway, and its crosstalk with immune therapy in cancer treatment. Theranostics. 2021;11(18):8813–35. https://doi.org/10.7150/thno.62521. PMID: 34522213; PMCID: PMC8419056.
Rao Z, Zhu Y, Yang P, Chen Z, **a Y, Qiao C, Liu W, Deng H, Li J, Ning P, Wang Z. Pyroptosis in inflammatory diseases and cancer. Theranostics. 2022;12(9):4310–29. https://doi.org/10.7150/thno.71086. PMID: 35673561; PMCID: PMC9169370.
Wei Y, Yang L, Pandeya A, Cui J, Zhang Y, Li Z. Pyroptosis-Induced inflammation and tissue damage. J Mol Biol. 2022;434(4):167301. https://doi.org/10.1016/j.jmb.2021.167301. Epub 2021 Oct 13. PMID: 34653436; PMCID: PMC8844146.
Xu KY, Wu CY, Tong S, **ong P, Wang SH. The selective Nlrp3 inflammasome inhibitor Mcc950 attenuates lung ischemia-reperfusion injury. Biochem Biophys Res Commun. 2018;503(4):3031–7. Epub 2018 Aug 23. PMID: 30146255.
Liu Z, Li Y, Yu L, Chang Y, Yu J. Penehyclidine hydrochloride inhibits renal ischemia/reperfusion-induced acute lung injury by activating the Nrf2 pathway. Aging. 2020;12(13):13400–21. https://doi.org/10.18632/aging.103444. Epub 2020 Jul 11. PMID: 32652517; PMCID: PMC7377887.
Shu Y, Yang Y, Zhang P. Neuroprotective effects of penehyclidine hydrochloride against cerebral ischemia/reperfusion injury in mice. Brain Res Bull. 2016;121:115–23. https://doi.org/10.1016/j.brainresbull.2016.01.008. Epub 2016 Jan 21. PMID: 26802510.
Wang YP, Li G, Ma LL, Zheng Y, Zhang SD, Zhang HX, Qiu M, Ma X. Penehyclidine hydrochloride ameliorates renal ischemia-reperfusion injury in rats. J Surg Res. 2014;186(1):390–7. https://doi.org/10.1016/j.jss.2013.07.041. Epub 2013 Aug 12. PMID: 23972620.
Lu Y, Zi C, Zhang L, Cui B, Li L, Ma J. Penehyclidine Hydrochloride protects rat cardiomyocytes from Ischemia- Reperfusion Injury by platelet-derived growth Factor-B. Comb Chem High Throughput Screen. 2023;26(6):1204–13. https://doi.org/10.2174/1386207325666220715090505. PMID: 35838232; PMCID: PMC10236564.
Wu XJ, Liu HM, Song XM, Zhao B, Leng Y, Wang EY, Zhan LY, Meng QT, **a ZY. Penehyclidine hydrochloride inhibits TLR4 signaling and inflammation, and attenuates blunt chest trauma and hemorrhagic shock-induced acute lung injury in rats. Mol Med Rep. 2018;17(5):6327–36. Epub 2018 Feb 27. PMID: 29488614; PMCID: PMC5928610.
Zhan J, **ao F, Li JJ, Zhang ZZ, Chen K, Wang YP, Wang YL. Penehyclidine hydrochloride decreases pulmonary microvascular permeability by upregulating beta arrestins in a murine cecal ligation and puncture model. J Surg Res. 2015;193(1):391-8. https://doi.org/10.1016/j.jss.2014.07.002. Epub 2014 Jul 5. PMID: 25096356.
Ye S, Yang X, Wang Q, Chen Q, Ma Y. Penehyclidine Hydrochloride alleviates Lipopolysaccharide-Induced Acute Lung Injury by ameliorating apoptosis and endoplasmic reticulum stress. J Surg Res. 2020;245:344–53. https://doi.org/10.1016/j.jss.2019.07.080. Epub 2019 Aug 16. PMID: 31425874.
Weng J, Chen M, Lin Q, Chen J, Wang S, Fang D. Penehyclidine hydrochloride defends against LPS-induced ALI in rats by mitigating endoplasmic reticulum stress and promoting the Hes1/Notch1 pathway. Gene. 2019;721:144095. https://doi.org/10.1016/j.gene.2019.144095. Epub 2019 Aug 30. PMID: 31476403.
He M, Zhao Y, Li S, Luo A, Chen H. Effect of penehyclidine hydrochloride on inflammatory response and oxidative stress in rats with cardiopulmonary bypass related-lung injury. Acta Cir Bras. 2022;37(4):e370406. https://doi.org/10.1590/acb370406. PMID: 35766672; PMCID: PMC9239558.
Acknowledgements
The authors thank Dr. Hongjie Wang for advice in study design.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
Gao JG, Liu RF and Zhang XG conceived the idea and conceptualised the study. Liu RF and Zhang XG collected the data. Yan J, Liu S, Li YL and Wu GY analysed and interpreted the data. Yan J, Liu S, Li YL and Wu GY statistically analyzed the data. No external funding received to conduct this study. Liu RF and Zhang XG drafted the manuscript. Gao JG reviewed the manuscript. All authors read and approved the final draft.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All experiments were evaluated and approved by Animal Welfare and Ethical Committee of Hebei University (No. IACUC-2023004SR). The animal use protocol listed below has been reviewed and approved by the Animal Ethical and Welfare Committee (AEWC). The study is reported in accordance with ARRIVE guidelines.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Material 1
: Supplemental figure: The groups of WB gels are: Sham group, Control group, PHC group, Sham group, Control group, PHC group, Sham group, Control group, PHC group
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Liu, R., Zhang, X., Yan, J. et al. Penehyclidine hydrochloride alleviates lung ischemia-reperfusion injury by inhibiting pyroptosis. BMC Pulm Med 24, 207 (2024). https://doi.org/10.1186/s12890-024-03018-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12890-024-03018-5