Abstract
Background
Chronic obstructive pulmonary disease (COPD) is underdiagnosed with the current gold standard measure pulmonary function test (PFT). A more sensitive and simple option for early detection and severity evaluation of COPD could benefit practitioners and patients.
Methods
In this multicenter retrospective study, frontal chest X-ray (CXR) images and related clinical information of 1055 participants were collected and processed. Different deep learning algorithms and transfer learning models were trained to classify COPD based on clinical data and CXR images from 666 subjects, and validated in internal test set based on 284 participants. External test including 105 participants was also performed to verify the generalization ability of the learning algorithms in diagnosing COPD. Meanwhile, the model was further used to evaluate disease severity of COPD by predicting different grads.
Results
The Ensemble model showed an AUC of 0.969 in distinguishing COPD by simultaneously extracting fusion features of clinical parameters and CXR images in internal test, better than models that used clinical parameters (AUC = 0.963) or images (AUC = 0.946) only. For the external test set, the AUC slightly declined to 0.934 in predicting COPD based on clinical parameters and CXR images. When applying the Ensemble model to determine disease severity of COPD, the AUC reached 0.894 for three-classification and 0.852 for five-classification respectively.
Conclusion
The present study used DL algorithms to screen COPD and predict disease severity based on CXR imaging and clinical parameters. The models showed good performance and the approach might be an effective case-finding tool with low radiation dose for COPD diagnosis and staging.
Similar content being viewed by others
Background
Chronic obstructive pulmonary disease (COPD) is a common pulmonary disease characterized by persistent respiratory symptoms and airflow limitation that is due to airway and/or alveolar abnormalities mainly caused by cigarette smoking [1]. Despite many decades of research on the pathogenesis and treatment of COPD, the medical community has failed to decrease its morbidity and mortality to the same degree that has been achieved in other major noncommunicable diseases, An important factor contributing to this slow progress may be that he previous COPD definition showed limitations as the lack of identification of the disorder at its early stages in the absence of flow limitation [2]. The proposed solutions are aimed to encourage novel treatments and translational studies: incorporating into the definition objectivable early computed tomography (CT) scan changes [3]. Epidemiological studies have shown that COPD is the third leading cause of death and accounts for 5% of all deaths worldwide each year [4]. The overall prevalence of COPD in people aged 40 years and older was 12.64% (95% CI 10.75%-14.65%) and 7.38% [5]. About a quarter of adults over 40 years have moderate airflow limitations, yet most of them are unaware of their conditions [6]. Early diagnosis of COPD is critical for early self-management and timely therapy to improve the overall prognosis [7, 8]. However, a considerable proportion of COPD patients are undiagnosed. An estimate of over 40% of COPD patients remain undiagnosed, particularly in develo** countries [9, 10], and only 12% of individuals with chronic airflow limitations had a previous spirometry-defined COPD diagnosis during the recent screening of 57,779 participants in China [11].
The conventional diagnosis and staging measure of COPD is pulmonary function test (PFT) according to the Global Initiative for Chronic Obstructive Lung Disease (GOLD) diagnostic criteria [12]. However, spirometry strongly depends on patients’ cooperation and COPD can either be misdiagnosed or missed entirely when using spirometry alone [13]. The 2017 GOLD report recommended that therapy should be based on clinical criteria rather than isolated PFT [14]. Furthermore, due to the shortage of experienced spirometry experts in poor areas of develo** countries, it’s hard to use PFT to screen asymptomatic patients in regular health examination on a large scale. Consequently, the need to develop new tools for early detection of COPD arises, and cost-effective strategies for case-finding are urgently needed.
In the past few years, growing evidences have shown that chest quantitative computer tomography (CT) has potential in COPD diagnosis and stratification [15,16,17,18]. CT-based imaging can help improve COPD detection and evaluation in patients who cannot undergo PFT [19]. In a recent study, deep learning (DL) models that utilize computed tomography (CT) image data were developed for automated detection and staging of spirometry-defined COPD. The result showed chest CT-DL approach could automatically identify spirometry-defined COPD and categorize patients according to the GOLD scale [11]. In the research reported by Lin Zhang, et al., they trained and tested the deep convolutional neural network (CNN) based on CT images of lung parenchyma and bronchial wall to determine the presence of COPD and GOLD staging, using PFT as reference, so as to infer lung function and determine the existence and severity of COPD. The result demonstrated that CNN can identify emphysema and airway wall remodeling on CT images to infer lung function and determine the existence and severity of COPD. As the CNN reached AUCs of 0.853 to determine the presence of COPD in the training and external test cohorts, and the accuracies of CNN to determine COPD GOLD grade in three- and five- classifications were 77.4 and 67.9%, respectively [27,28]. It’s highly accurate for advanced emphysema [29], but only moderately sensitive in patients with mild to moderate emphysema [30,31,32]. Recently, machine learning (ML) technology is being assessed to perform medical tasks in almost every field of practice [33]. It has been successfully used in automated interpretation of PFT for differential diagnosis of obstructive lung diseases and COPD detection based on HRCT images [34,35,36]. However, none of the previous studies have used deep learning (DL) to predict COPD based on CXR images.
In the present study, we used DL algorithms to detect COPD and predict disease severity based on CXR imaging and clinical parameters, with the purpose to screen potential COPD patients while minimizing the need for additional radiographic examination. We hypothesized that applying DL algorithms to clinical and CXR imaging features would improve early diagnosis and prognosis prediction in COPD.
Methods
Study population
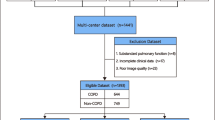
This was a multicenter retrospective study performed at the Third Affiliated Hospital of Sun Yat-sen University, the Third Affiliated Hospital of Sun Yat-sen University. Lingnan Hospital, the Six Affiliated Hospital of Guangzhou Medical University, Qingyuan People’s Hospital, and Huazhou Hospital of Traditional Chinese Medical. The data included frontal CXR images and clinical information of 1055 participants (535 patients with COPD and 520 controls) from outpatient, inpatient, and physical examination center settings between January 2019 and December 2021. This study was reviewed and approved by the Ethics Committee of the Third Affiliated Hospital of Sun Yat-sen University and requirements for written informed consent were waived due to the retrospective nature of the research.
Frontal images were identified by searching image databases for CXRs of the patients who also received PFT within one week, whereas lateral radiographs and oblique views were excluded. COPD diagnosis was confirmed by forced expiratory volume in 1 s (FEV1) to forced vital capacity (FVC) ratio less than 0.7 after inhalation of bronchodilators according to GOLD 2018. The severity of COPD is graded as GOLD 1 (FEV1%pred ≥ 80%), GOLD 2 (50%≤FEV1%pred < 80%), GOLD 3 (30%≤FEV1%pred < 50%), and GOLD 4 (FEV1%pred < 30%), as depending on the FEV1%pred value of PFT based on GOLD 2018. Since the diagnosis and severity of COPD depend on GOLD level based on PFT result, this study used the GOLD level as a reference standard to classify patients.
As COPD usually occurs in patients older than 40 years [6], all the subjects included in the present study were > 40 years old. The exclusion criteria were: (1) pregnant women; (2) other pulmonary diseases with abnormal CXR presentations, such as bronchiectasis, pulmonary fibrosis, atelectasis, pulmonary infectious disease, active pulmonary tuberculosis, pleural effusion, lung cancer, and pneumothorax; (3) severe renal insufficiency, severe liver disease, human immunodefciency virus, or other immune-related diseases; (4) previous chest surgery; (5) severe cardiac insufficiency; (6) in the acute phase of COPD. A total of 1224 subjects ranging in age from 41 to 86 years were recruited. Spirometry data, demographic information, smoking history, clinical indices and manifestations information were collected using a standardized data collection form. After excluding cases with incomplete clinical data (57 cases), substandard pulmonary function (42 cases), and poor CXR image quality (70 cases), finally 1055 participants were enrolled in the cohort. The 950 subjects recruited from the Third Affiliated Hospital of Sun Yat-sen University and the Third Affiliated Hospital of Sun Yat-sen University. Lingnan Hospital were randomly split into training set and internal test set, with a ratio of 70%:30% (Fig. 1). For the training set (n = 666), 49.4% were COPD patients (n = 329). Of the 284 subjects in internal test set, 49.3% (n = 140) were COPD patients. The 105 participants (66 COPD patients and 39 control subjects) from the Six Affiliated Hospital of Guangzhou Medical University, Qingyuan People’s Hospital, and Huazhou Hospital of Traditional Chinese Medical were used for external test set.
Demographic and clinical characteristics
A total of 1055 participants were finally included in the study: 535 COPD patients and 520 control subjects. The median age of COPD patients was higher than that of non-COPD participants (67 vs. 63, P < 0.001) and the majority of the COPD cohort was male (87.85%), which was consistent with COPD gender distributions in China [6]. A higher proportion of smokers (78.50% vs. 20.96%, P < 0.001), a reduced FEV1% (55.02% vs. 92.33%, P < 0.001), and a lower BMI (22.10 vs. 24.13, P = 0.001) were evident among COPD patients compared to control group. In addition, the symptoms of cough, sputum, and dyspnea were more common in COPD patients than in control subjects (77.94%, 51.40%, 50.47% vs. 13.46%, 8.46%, 2.31%, respectively). The percentages of stage 1, 2, 3, and 4 spirometry-defined COPD subjects on the GOLD scale were 18.50%, 42.06%, 25.61%, and 13.83%, respectively. Detailed demographic and clinical characteristics for the participants were provided in Table 1.
Data preprocessing
The data set consisted of clinical information and CXR images. The clinical information contained nine characteristics in all: gender, age, average body mass index (BMI), history of smoking, cough, expectoration, carbon dioxide retention, (arterial partial pressure of carbon dioxide greater than 45mmHg), dyspnea, respiratory failure (arterial partial oxygen pressure less than 60mmHg, with or without elevated carbon dioxide levels) and so on. Identity information including patient’s name, hospitalization ID and other DICOM information were removed from CXR images. In two classifications, the data were divided into normal control subjects without COPD (n = 520) and patients with COPD (n = 535). In three-classifications, the data were divided into three groups, normal (n = 520), GOLD 1/2 (n = 318), and GOLD 3/4 (n = 217), while in five-classifications, the data were divided into five groups, including normal (n = 520), GOLD 1 (n = 99), GOLD 2 (n = 225), GOLD 3 (n = 137), and GOLD 4 (n = 74).
Development of the COPD detection and COPD staging model
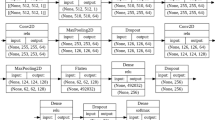
We first studied the comparison between normal control subjects and patients with COPD based on clinical data only. Clinical information were preprocessed through sk-learn’s data preprocessing interface StandardScaler, by filling in the missing values and standardizing the data to (-1, 1) interval. Three classical machine learning algorithms, decision tree, support vector machine and random forest were selected for modeling. The random forest algorithm showing best area under the curve (AUC) was determined to be the best algorithm for feature mining and extraction of clinical inform clinical information. For classification of CXR images, we generally chose transfer learning for modeling. Three classical deep learning convolution neural networks such as EfficientNet-B5, ResNet50 and DenseNet were selected for modeling [37, In this multicenter retrospective study, we used deep learning algorithms to predict COPD based on clinical parameters and CXR images, with the purpose to find a more sensitive and simple option for early detection of potential COPD patients. The diagnostic accuracies of COPD reached an AUC of 0.965 in internal test and 0.934 in external test to detect COPD according clinical characteristics and CXR images, better than the results of previous study, which used deep residual networks for automated detection of COPD based on low dosed CT images only, with an ACU of 0.889 [4] and 0.899 [49]. Through the gradcam interface of the EfficientNet model, we can get the hot focus areas of the CNN model for the diagnosis of COPD patients. The results indicated that the model paid specific attention to these lesions when distinguishing COPD subjects. This could make doctors and patients better understand what they have learned from DL, and whether they can rest assured that ML can assist them in diagnosis. There are several limitations in the present study. First, this was a retrospective study only performed in four medical institutes. The outer generalization of the deep learning algorithms needed to be tested further by prospective study including more centers. Second, the prediction was performed based on COPD and normal control. Subjects involved in the present study were mainly COPD patients without other complications or disease, as radiographic images with other lesion were eliminated. For better use in clinical practice, subsequent study with larger sample size including complication or comorbidities of COPD is needed. Third, a limited number of subjects with GOLD 1 and 4 were enrolled, which might constrain the model’s stratification capacity and resulted in the discrepancy of staging efficiency between groups. To improve the efficiency of detection and staging, we are currently recruiting more participants and aim to optimize our cohort in the future. In conclusion, we developed a more simple, sensitive and safer learning approach for detecting and staging of COPD. The proposed model approach achieved the desired performance and could serve as a powerful tool for COPD screening and evaluation, which may help clinicians easily identify possible suspected COPD patients. Nevertheless further studies are necessary to determine the feasibility of these outcomes in a prospective clinical setting.Discussion
Data availability
The datasets generated and analysed during the current study are not publicly available due for protection of participants’ privacy, but are available from the corresponding author on reasonable request.
Abbreviations
- COPD:
-
Chronic obstructive pulmonary disease
- PFT:
-
pulmonary function test
- GOLD:
-
Global Initiative for Chronic Obstructive Lung Disease
- CT:
-
computed tomography
- CXR:
-
chest X-ray
- ML:
-
machine learning
- DL:
-
deep learning
- FEV1:
-
forced expiratory volume in 1 s
- FVC:
-
forced vital capacity
- BMI:
-
body mass index
- SD:
-
standard deviation
- AUC:
-
area under the curve
- CO2:
-
carbon dioxide
- ACC:
-
accuracy
- PPV:
-
positive predictive value
- NPV:
-
negative predictive value
- F1:
-
false positive rate
- Grad-CAM:
-
Gradient-weighted class activation map**
References
Bakker JT, Klooster K, Vliegenthart R, et al. Measuring pulmonary function in COPD using quantitative chest computed tomography analysis. Eur Respir Rev. 2021;30:210031.
Confalonieri M, Braga L, Salton F, Ruaro B, Confalonieri P. Chronic obstructive Pulmonary Disease Definition: is it time to incorporate the Concept of failure of lung regeneration? Am J Respir Crit Care Med. 2023;207(3):366–7.
Celli B, Fabbri L, Criner G, et al. Definition and nomenclature of Chronic Obstructive Pulmonary Disease: time for its revision. Am J Respir Crit Care Med. 202;206(11):1317–25.
World Health Organization. The top 10 causes of death. www.who.int/news-room/fact-sheets/detail/the-top-10-causes-of-death Date last updated: 9 December 2020.
Al Wachami N, Guennouni M, Iderdar Y, et al. Estimating the global prevalence of chronic obstructive pulmonary disease (COPD): a systematic review and meta-analysis. BMC Public Health. 2024;24(1):297.
Wang C, Xu J, Yang L, et al. Prevalence and risk factors of chronic obstructive pulmonary disease in China (the China Pulmonary Health [CPH] study): a national cross-sectional study. Lancet. 2018;391:1706–17.
Tang LYW, Coxson HO, Lam S, et al. Towards large-scale case-finding: training and validation of residual networks for detection of chronic obstructive pulmonary disease using low-dose CT. Lancet Digit Health. 2020;2:e259–67.
Labaki WW, Han MK. Improving detection of early chronic obstructive pulmonary disease. Ann Am Thorac Soc. 2018;15:S243–8.
Perez-Padilla R, Thirion-Romero I, Guzman N. Underdiagnosis of chronic obstructive pulmonary disease: should smokers be offered routine spirometry tests? Expert Rev Respir Med. 2018;12:83–5.
Lamprecht B, Soriano JB, Studnicka M, et al. Determinants of underdiagnosis of COPD in national and international surveys. Chest. 2015;148:971–85.
Sun J, Liao X, Yan Y, et al. Detection and staging of chronic obstructive pulmonary disease using a computed tomography–based weakly supervised deep learning approach. Eur Radiol. 2022;32:5319–29.
Fischer AM, Varga-Szemes A, Martin SS, et al. Artificial Intelligence-based fully automated per lobe segmentation and emphysema-quantificationmbased on chest computed Tomography compared with Global Initiative for Chronic Obstructive Lung Disease severity of smokers. J Thorac Imaging. 2020;35:S28–34.
Miller MR, Levy ML. Chronic obstructive pulmonary disease: missed diagnosis versus misdiagnosis. BMJ. 2015;351:h3021.
Vogelmeier CF, Criner GJ, Martinez FJ, et al. Global strategy for the diagnosis, management, and Prevention of Chronic Obstructive Lung Disease 2017 Report: GOLD Executive Summary. Am J Respir Crit Care Med. 2017;195:557–82.
Labaki WW, Martinez CH, Galbàn CG, et al. The role of chest CT in the evaluation and management of patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2017;196:1372–9.
Lynch DA, Moore CM, Wilson C, et al. CT-based visual classification of emphysema: association with mortality in the COPDGene study. Radiology. 2018;288:859–66.
Bhatt SP, Washko GR, Hoffman EA et al. Imaging advances in chronic obstructive pulmonary disease. Insights from the genetic epidemiology of Chronic Obstructive Pulmonary Disease (COPDGene) study. Am J Respir Crit Care Med 199:286–301.
Park J, Hobbs BD, Crapo JD, et al. Subty** COPD by using visual and quantitative CT imaging features. Am J Respir Crit Care Med. 2019;199:286–301.
Castillo-Saldana D, Hague CJ, Coxson HO, et al. Using quantitative computed tomographic imaging to understand chronic obstructive pulmonary disease and fibrotic interstitial lung disease: state of the art and future directions. J Thorac Imaging. 2020;35:246–54.
Zhang L, Jiang B, Wisselink HJ, Vliegenthart R, **e X. COPD identification and grading based on deep learning of lung parenchyma and bronchial wall in chest CT images. Br J Radiol. 2022;95(1133):20210637.
Paoletti M, Cestelli L, Bigazzi F, et al. Chronic obstructive pulmonary disease: pulmonary function and CT lung attenuation do not show linear correlation. Radiology. 2015;276:571–8.
Mettler FA Jr, Huda W, Yoshizumi TT, Mahesh M. Effective doses in radiology and diagnostic nuclear medicine: a catalog. Radiology. 2008;248:254–63.
Larke FJ, Kruger RL, Cagnon CH, et al. Estimated radiation dose associated with low-dose chest CT of average-size participants in the National Lung Screening Trial. AJR Am J Roentgenol. 2011;197:1165–9.
Willer K, Fingerle AA, Noichl W, et al. X-ray dark-field chest imaging for detection and quantification of emphysema in patients with chronic obstructive pulmonary disease: a diagnostic accuracy study. Lancet Digit Health. 2021;3:e733–44.
den Harder AM, de Boer E, Lagerweij SJ, et al. Emphysema quantification using chest CT: influence of radiation dose reduction and reconstruction technique. Eur Radiol Exp. 2018;2:30.
Cavigli E, Camiciottoli G, Diciotti S, et al. Whole-lung densitometry versus visual assessment of emphysema. Eur Radiol. 2009;19:1686–92.
Singla S, Gong M, Riley C, et al. Improving clinical disease subty** and future events prediction through a chest CTbased deep learning approach. Med Phys. 2021;48:1168–81.
Goldin JG. Imaging the lungs in patients with pulmonary emphysema. J Thorac Imaging. 2009;24:163–70.
Miniati M, Monti S, Stolk J, et al. Value of chest radiography in phenoty** chronic obstructive pulmonary disease. Eur Respir J. 2008;31:509–15.
Washko GR. Diagnostic imaging in COPD. Semin Respir Crit Care Med. 2010;31:276–85.
Meinel FG, Schwab F, Schleede S, et al. Diagnosing and map** Pulmonary Emphysema on X Ray Projection images: Incremental Value of Grating Based X-Ray Dark-Field Imaging. PLoS ONE. 2013;8:e59526.
Hellbach K, Yaroshenko A, Meinel FG, et al. In vivo dark-field radiography for early diagnosis and staging of pulmonary emphysema. Invest Radiol. 2015;50:430–5.
Rajkomar A, Dean J, Kohane I. Machine learning in Medicine. N Engl J Med. 2019;380:1347–58.
Das N, Topalovic M, Janssens W. Artificial intelligence in diagnosis of obstructive lung disease: current status and future potential. Curr Opin Pulm Med. 2018;24:117–23.
Topalovic M, Laval S, Aerts J-M, et al. Automated interpretation of pulmonary function tests in adults with respiratory complaints. Respiration. 2017;93:170–8.
González G, Ash SY, Vegas-Sánchez-Ferrero G, et al. Disease staging and prognosis in smokers using deep learning in chest computed tomography. Am J Respir Crit Care Med. 2018;197:193–203.
Huang G, Liu Z, Pleiss G, Maaten LV, Weinberger KQ. Convolutional Networks with dense connectivity. IEEE Trans Pattern Anal Mach Intell. 2022;44(12):8704–16.
He K, Zhang X, Ren S, Sun J. Deep residual learning for image recognition. ar**v:1512.03385.
Tan M, Quoc V, Le. EfficientNet: rethinking Model scaling for convolutional neural networks. ar**v:1905.11946.
Chandra TB, Singh BK, Jain D. Integrating patient symptoms, clinical readings, and radiologist feedback with computer-aided diagnosis system for detection of infectious pulmonary disease: a feasibility study. Med Biol Eng Comput. 2022;60(9):2549–65.
Chandra TB, Singh BK, Jain D. Disease localization and Severity Assessment in chest X-Ray images using Multi-stage superpixels classification. Comput Methods Programs Biomed. 2022;222:106947.
Chandra TB, Verma K, Singh BK, Jain D, Netam SS. Coronavirus disease (COVID-19) detection in chest X-Ray images using majority voting based classifier ensemble. Expert Syst Appl. 2021;165:113909.
Diab N, Gershon AS, Sin DD, et al. Underdiagnosis and overdiagnosis of chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2018;198:1130–9.
Feng Y, Wang Y, Zeng C, et al. Artificial Intelligence and Machine Learning in Chronic Airway diseases: Focus on Asthma and Chronic Obstructive Pulmonary Disease. Int J Med Sci. 2021;18:2871–89.
Xu C, Qi S, Feng J, et al. DCT-MIL: Deep CNN transferred multiple instance learning for COPD identification using CT images. Phys Med Biol. 2020;65:145011.
Matsumura K, Ito S. Novel biomarker genes which distinguish between smokers and chronic obstructive pulmonary disease patients with machine learning approach. BMC Pulm Med. 2020;20(1):29.
Westcott A, Capaldi D, McCormack DG, et al. Chronic obstructive Pulmonary Disease: Thoracic CT Texture Analysis and Machine Learning to Predict Pulmonary Ventilation. Radiology. 2019;293:676–84.
Andreeva E, Pokhaznikova M, Lebedev A, et al. Spirometry is not enough to diagnose COPD in epidemiological studies: a follow-up study. NPJ Prim Care Respir Med. 2017;27:62.
Esteva A, Robicquet A, Ramsundar B, et al. A guide to deep learning in healthcare. Nat Med. 2019;25:24–9.
Acknowledgements
Not applicable.
Funding
This work was supported by National Key Technology R&D Program (2018YFC1311900) and Shenzhen Science and Technology Program (No. JCYJ20220530145001002).
Author information
Authors and Affiliations
Contributions
TianTuo Zhang and LanQing Han had full access to all of the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. **aoLing Zou, Yong Ren and HaiLing Yang equally contributed to this work.Concept and design: **aoLing Zou, Yong Ren, HaiLing YangAcquisition, analysis, or interpretation of data: Yong Ren, HaiLing Yang, ManMan Zou, ** Meng, LiYi Zhang, MingJuan Gong, WenWen DingStatistical analysis: **aoLing Zou, Yong Ren, HaiLing YangAdministrative, technical, or material support: **aoLing Zou, Yong Ren, HaiLing Yang, TianTuo Zhang, LanQing Han Drafting of the manuscript: **aoLing Zou, Yong RenSubmission of the study: **aoLing ZouSupervision: TianTuo Zhang, LanQing Han.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
This study was reviewed and approved by the Ethics Committee of the Third Affiliated Hospital of Sun Yat-sen University. The need for informed consent was waived by the Ethics Committee of the Third Affiliated Hospital of Sun Yat-sen University.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
TianTuo Zhang and LanQing Han contributed to this work as corresponding authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Zou, X., Ren, Y., Yang, H. et al. Screening and staging of chronic obstructive pulmonary disease with deep learning based on chest X-ray images and clinical parameters. BMC Pulm Med 24, 153 (2024). https://doi.org/10.1186/s12890-024-02945-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12890-024-02945-7