Abstract
Objectives
Primary blast lung injury (PBLI) is the main cause of death in blast injury patients, and is often ignored due to the absence of a specific diagnosis. Circular RNAs (circRNAs) are becoming recognized as new regulators of various diseases, but the role of circRNAs in PBLI remain largely unknown. This study aimed to investigate PBLI-related circRNAs and their probable roles as new regulators in PBLI in order to provide new ideas for PBLI diagnosis and treatment.
Methods
The differentially expressed (DE) circRNA and mRNA profiles were screened by transcriptome high-throughput sequencing and validated by quantitative real-time PCR (qRT-PCR). The GO and KEGG pathway enrichment was used to investigate the potential function of DE mRNAs. The interactions between proteins were analyzed using the STRING database and hub genes were identified using the MCODE plugin. Then, Cytoscape software was used to illustrate the circRNA-miRNA-hub gene network.
Results
A total of 117 circRNAs and 681 mRNAs were aberrantly expressed in PBLI, including 64 up-regulated and 53 down-regulated circRNAs, and 315 up-regulated and 366 down-regulated mRNAs. GO and KEGG analysis revealed that the DE mRNAs might be involved in the TNF signaling pathway and Fanconi anemia pathway. Hub genes, including Cenpf, Ndc80, Cdk1, Aurkb, Ttk, Aspm, Ccnb1, Kif11, Bub1 and Top2a, were obtained using the MCODE plugin. The network consist of 6 circRNAs (chr18:21008725–21020999 + , chr4:44893533–44895989 + , chr4:56899026–56910247-, chr5:123709382–123719528-, chr9:108528589–108544977 + and chr15:93452117–93465245 +), 7 miRNAs (mmu-miR-3058-5p, mmu-miR-3063-5p, mmu-miR-668-5p, mmu-miR-7038-3p, mmu-miR-761, mmu-miR-7673-5p and mmu-miR-9-5p) and 6 mRNAs (Aspm, Aurkb, Bub1, Cdk1, Cenpf and Top2a).
Conclusions
This study examined a circRNA-miRNA-hub gene regulatory network associated with PBLI and explored the potential functions of circRNAs in the network for the first time. Six circRNAs in the circRNA-miRNA-hub gene regulatory network, including chr18:21008725–21020999 + , chr4:44893533–44895989 + , chr4:56899026–56910247-, chr5:123709382–123719528-, chr9:108528589–108544977 + and chr15:93452117–93465245 + may play an essential role in PBLI.
Similar content being viewed by others
Introduction
Blast injuries are the most common fatal injuries in military actions, terrorist attacks, and peacetime accidents, such as industrial accidents and gas explosions [1]. In recent years, explosion accidents have occurred frequently, and blast injury has attracted increasing attention [2, 3]. At the moment of the explosion, the power is rapidly released in the form of heat, gas products and shock waves. Among these forms of power, shock waves are an important cause of injury, and can cause damage to multiple systems and organs, namely, primary blast injury [4]. The lung is the most vulnerable target organ in blast injury due to its air-containing. Clinical reports show that exposure to blast shock waves can easily cause primary blast lung injury (PBLI), leading to severe clinical manifestations, such as severe pulmonary contusion, hemorrhage, edema, and further acute lung injury (ALI), even more serious acute respiratory distress syndrome (ARDS), which eventually leads to death [5,
Methods
Experimental animals
Male C57BL/6 mice, aged 8–10 weeks and weighing 20–22 g, were purchased from Bei**g Vital River Laboratory Animal Technology Co., Ltd. (Beijiing, China). All mice were housed in a temperature-controlled and specific-pathogen-free environment with a 12 h light/dark cycle. All mice were free access to food and water. And all experimental procedures and protocols were conducted in accordance with the guidelines of the local animal care and use committee. Animal welfare and experimental design were approved by the Institutional Animal Care and Use Committee of Yi Shengyuan Gene Technology (Tian**) (YSY-DWLL-2021013). The study is reported in accordance with ARRIVE guidelines.
Establishment of primary blast lung injury model in mice
The self-made mini shock tube stimulation device was used to establish the PBLI model as previously described [20]. In brief, twelve mice were randomly divided into two groups: the control group and the PBLI group. For statistical analysis, six mice were included in each group. After anesthesia, the mice in PBLI group were placed in a prone position on a black rubber plate that attached to the device, with holes in the rubber plate located directly below the mice's chest. A shock wave with a peak pressure of 0.5 bar was generated using the device to cause lung injury in mice. The animals in the control group were anesthetized in the same manner but were not exposed to blast overpressure.
Sample preparation
Six hours post-blast, euthanasia by cervical dislocation was performed and the lung of all mice was harvested. The right upper lobe of lungs was taken and placed in enzyme-free EP tubes, part of lung tissue of each mouse was cut and stored in liquid nitrogen for subsequent sequencing. The other part was added with 1 mL TRIzol reagent, homogenated and placed in the refrigerator at -80℃ for the RNA extraction. Every two tissues used for sequencing were mixed into one sample, that is, 3 samples each in the control group and the PBLI group for sequencing.
Total RNA isolation, library construction and sequencing
Total RNA from the lung tissue of the two groups of mice was isolated using TRIzol reagent (Invitrogen). The concentrations and purity of the RNA were measured using a NanoDrop ND-1000 instrument (Thermo, Waltham, MA, USA). OD260/OD280 values were used as the purity index of RNA, and OD260/OD280 values ranging from 1.8 to 2.1 were considered to be qualified. And RNA integrity and gDNA contamination were measured using denatured agarose gel electrophoresis.
The transcriptome high-throughput sequencing service was provided by CloudSeq Biotech (Shanghai, China). Briefly, rRNAs in total RNA were removed using the NEBNext® rRNA Depletion Kit (New England Biolabs, Inc., Massachusetts, USA) following the manufacturer's instructions. The TruSeq Stranded Total RNA Library Prep Kit (Illumina, USA) was used to construct RNA sequencing libraries with RNA-depleted RNAs. The BioAnalyzer 2100 instrument (Agilent Technologies, USA) was used to perform the library quality control and quantification. The 150 bp double-ended reads sequencing was performed on the Illumina NovaSeq 6000 instrument.
Data quality control
After image and base recognition, double-ended raw reads were harvested from the Illumina NovaSeq 6000 sequencer. Q30 was used for quality control, cutadapt software (v1.9.3) was used to remove connectors, low-quality reads were removed, and high-quality clean reads were obtained.
Differential expression of circRNAs in the lungs of PBLI mice
The high-quality reads were aligned to the reference genome/transcriptome with STAR software (v2.5.1b) and circRNAs were detected and identified with DCC software (v0.4.4). According to the alignment position of two ends of circRNAs, the circRNAs were divided into exonic circRNAs, intronic circRNAs, or inter-geneic circRNAs. The circRNAs were then identified using the circBase database and circ2Trait circRNA-disease database links based on the circRNA genome location. The newly identified circRNAs were labeled as novel.
The default TMM method of edgeR software (v3.16.5) was used to standardize the original junction reads, and the number of standardized reads was used to calculate the differential expression of circRNAs between the two groups. A fold change ≥ 2.0 and a p value ≤ 0.05 were taken as the threshold for DE circRNAs.
Differential expression of mRNAs in the lungs of PBLI mice
The high-quality reads were aligned to the mouse reference genome (UCSC MM10) with hisat2 software (v2.0.4). Then, FPKM values were obtained to create an expression profile of mRNA using cuffdiff software (v2.2.1, part of cufflinks) under the guidance of the Ensembl gtf gene annotation file. The cuffdiff software was used to identify DE mRNAs between the two groups. A fold change ≥ 2.0, p value ≤ 0.05, and FPKM value ≥ 0.5 in at least one sample were considered as differentially expressed.
Validation and quantification of circRNAs
The DE circRNAs were validated by quantitative real-time PCR (qRT-PCR). Two upregulated and two downregulated circRNAs, and three circRNAs in the ceRNA network were randomly selected. The housekee** gene β-actin was used as a reference for normalization, and the primers for circRNAs were specific divergent primers (Table 1). Total RNA was reverse transcribed into complementary DNA using the PrimeScript RT Reagent Kit (Perfect Real Time; TaKaRa, Osaka, Japan), and qRT-PCR was performed on a Roche LightCycler 96 Real-Time PCR System using the Hieff® qPCR SYBR Green Master Mix (Yeasen, Shanghai, China) according to the manufacturer’s instructions. Three independent assays were performed on all samples, in which samples were assessed in triplicate. The 2−ΔΔCt method was used to calculate the relative expression of the circRNAs.
Functional analysis
To better understand the pathogenesis of PBLI, we used Gene Ontology (GO) (http://www.geneontology.org/) analysis and Kyoto Encyclopedia of Genes and Genomes (KEGG) (http://www.genome.jp/kegg) pathway analysis to predict the potential function of linear transcripts of DE mRNAs [21,22,23]. GO analysis consists of three components: Molecular Function (MF), Biological Process (BP), and Cell Component (CC). A p value ≤ 0.05 in GO terms is considered statistically significant. The KEGG pathway analysis is the process of map** molecular data sets from genomics, transcriptomics, proteomics, and metabolomics onto the KEGG pathway map for the biological function interpretation of these molecules. A p value ≤ 0.05 was considered to indicate significant enrichment.
Construction of the PPI regulatory network and screening of hub genes
The STRING database (http://string-db.org) was used to predict the interaction of proteins, the minimum required interaction score was set to high confidence (0.7), and the PPI network was established by CytoScape 3.9.1 software, an open source software platform for visualizing complex networks and integrating these with any type of attribute data. The MCODE plug-in of Cytoscape software, a clustering algorithm that reclassifies existing proteins or genes, was run to predict the most significantly meaningful protein modules. The advanced options were set as degree cutoff = 2, haircut, node score cutoff = 0.2, k-core = 2, and max depth = 100 [24]. In addition, hub genes in the PPI network were filtered by the Maximal Clique Centrality (MCC) arithmetic of the Cytoscape plug-in cytoHubba, which ranks nodes according to their attributes in the network, mining key genes (hub genes) and sub networks [25, 26], the top ten nodes ranked by the MCC algorithm were considered as hub genes.
Prediction of circRNA-miRNA-hub gene associations
CircRNA-miRNA and miRNA-hub gene interactions were predicted by miRanda (http://www.miranda.org/) and TargetScan (http://www.targetscan.org/vert_71/), widely used databases that predict biological targets of miRNAs. For each circRNA/mRNA, the top 5 miRNAs that potentially bind are listed. Upregulated circRNA-miRNA-upregulated mRNA, and downregulated circRNA-miRNA-downregulated mRNA were selected, and the network was constructed by Cytoscape 3.9.1 software.
Results
Identification of circRNAs in mouse lung tissue
In the present study, a total of 3696 circRNAs were detected from the control group and the PBLI group. These circRNAs were located in the whole genomic region (Fig. 2a), including chrX (2.30%), chrY (0.14%), and chrM (0.11%). A total of 34.73% of host genes could produce two or more circRNAs, and some genes could generate more than ten circRNAs (0.14%) (Fig. 2b). For example, Ahank, a gene located on chr19, produced 29 circular intronic RNAs, which was the most in this study. The circRNAs found so far mainly arose from exons, accounting for 76.98% of the circRNAs in this study (Fig. 2c). Sequence length analysis revealed that the circRNA transcripts were mainly 200–1200 bp in length (75.24%) but that 11.74% were over 2000 bp in length (Fig. 2d). Among the 3696 circRNAs that were identified in this study, 2276 circRNAs were already recorded in the circBase database or PubMed, and 1420 were considered as novel (Fig. 2e).
Expression patterns of circRNAs in mouse lungs. a Chromosome distribution of the 3696 circRNAs identified in the two groups. b Number of circRNAs produced from one host gene. c Percentages of circRNAs arising from different genomic loci. d Length distributions of the identified circRNAs. e Sources of the annotated circRNAs
Identification and validation of DE circRNAs in PBLI in lung tissue
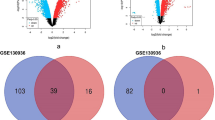
To investigate the possible circRNAs and mRNAs involved in PBLI, the DE circRNAs and mRNAs between the two groups were analyzed. Among the 3696 circRNAs identified in this study, 865 were detected only in the control group, 1332 were detected only in the PBLI group, and 1499 were detected in both groups (Fig. 3a). A total of 117 circRNAs were aberrantly expressed in the PBLI group, including 64 up-regulated and 53 down-regulated circRNAs (Fig. 3b, c). The top 20 up-regulated and down-regulated circRNAs are shown in Table 2. Two up-regulated (chr13:117249272–117249786 + and chr2:74568941–74573626-) and two down-regulated circRNAs (chr3:122794260–122804609 + and chr2:128675671–128676247-) were randomly selected for validation of DE circRNAs, which showed that the expression levels of DE circRNAs were consistent with the sequencing results (Fig. 3d).
Identification and validation of DE circRNAs. a Numbers of circRNAs detected in the two groups. b-c Histogram (b) and Heatmap (c) showing the significantly up-regulated and down-regulated circRNAs in the PBLI group. d Validation of differentially expressed circRNAs by qRT-PCR. The results are presented as the mean ± SD. *P < 0.05
Identification of DE mRNAs in PBLI
A total of 17,453 mRNAs were identified in the two groups, of which 470 were expressed only in the control group, 507 were expressed only in the PBLI group, and 16,417 were expressed in both groups (Fig. 4a). As shown in Fig. 4b and c, 681 mRNAs were aberrantly expressed in the PBLI group, among them, 315 were up-regulated and 366 were down-regulated. The top 20 up-regulated and down-regulated mRNAs were shown in Table 3.
GO and KEGG analyses of DE mRNAs
To investigate the potential function of the mRNAs, GO and KEGG pathway enrichment analyses of the DE mRNAs were performed. The GO analysis results, including BP, CC and MF categories are shown. For the up-regulated mRNAs, response to stress, extracellular region and protein binding are the most enriched GO terms in BP, CC and MF categories, respectively (Fig. 5a). The most enriched GO terms among the down-regulated mRNAs are mitotic cell cycle process in the BP category, chromosome, centromeric region in the CC category and carbohydrate derivative binding in the MFs category (Fig. 5b). KEGG pathway analysis indicated that the up-regulated mRNAs might be involved in the TNF signaling pathway and cytokine-cytokine receptor interaction (Fig. 5c). For the down-regulated mRNAs, the Fanconi anemia pathway and homologous recombination might play an essential role in PBLI (Fig. 5d).
PPI network, molecular complex detection analysis and hub gene identification
A PPI network including 234 nodes and 717 edges was built for the 681 DE mRNAs using the STRING database (Fig. 6). Subsequently, we used the MCODE method to screen hub genes from the PPI network, and the first module was selected after MCODE analysis (Fig. 7a). According to the MCC algorithm, the top 10 genes in the PPI network were obtained as hub genes, including Cenpf, Ndc80, Cdk1, Aurkb, Ttk, Aspm, Ccnb1, Kif11, Bub1 and Top2a (Fig. 7b), which are all down-regulated.
Hub gene identification and construction of a circRNA-miRNA-hub gene network. a Selected module of the PPI network. b Hub gene network. c circRNA-miRNA-hub gene network. The triangle, quadrilateral and circle represent miRNA, mRNA and circRNA, respectively. d Validation of circRNAs in circRNA-miRNA-hub gene network by qRT-PCR. The results are presented as the mean ± SD. *P < 0.05
Construction of a circRNA-miRNA-hub gene network
To construct the circRNA-miRNA-hub gene network, hub gene-related miRNAs, and top 20 DE circRNAs targeted miRNAs were predicted. As shown in Fig. 7c, the network consist of 6 circRNAs (chr18:21008725–21020999 + , chr4:44893533–44895989 + , chr4:56899026–56910247-, chr5:123709382–123719528-, chr9:108528589–108544977 + and chr15:93452117–93465245 +), 7 miRNAs (mmu-miR-3058-5p, mmu-miR-3063-5p, mmu-miR-668-5p, mmu-miR-7038-3p, mmu-miR-761, mmu-miR-7673-5p and mmu-miR-9-5p) and 6 mRNAs (Aspm, Aurkb, Bub1, Cdk1, Cenpf and Top2a) with 18 nodes and 14 edges was constructed. And chr18:21008725–21020999 + , chr4:44893533–44895989 + and chr4:56899026–56910247- were randomly selected for validation of circRNAs in the network. The results showed significant changes in the expression of these circRNAs in the lung tissue of mice exposed to the blast wave (Fig. 7d).
Discussion
Explosions can cause severe internal pulmonary injury despite tiny injuries observed on the external chest. This makes it difficult for PBLI to be diagnosed early. Therefore, early diagnosis and treatment are particularly important for victims suffering from PBLI [27]. Currently, PBLI is diagnosed with clinical manifestations such as hypoxemia and respiratory dysfunction, combined with chest X-ray and chest CT examination [28, 29], which are commonly performed in hospitals instead of rescue sites. In addition, although many studies and therapeutic trials have been conducted, at the present stage, the treatment of PBLI is predominantly supportive and symptomatic [6]. Due to the complexity and difficulty in diagnosis and treatment, the mortality rate of PBLI is extremely high. Therefore, the development of rapid diagnosis and specific treatment strategies is particularly important for reducing mortality. In this study, we constructed a circRNA-miRNA-hub gene network to explore the role of circRNAs in PBLI, aiming to provide new ideas for the PBLI diagnosis and treatment.
CircRNAs are a unique class of non-coding RNAs, as they are more abundant, more stable, and have certain tissue specificity than other types of RNA. CircRNAs are considered to be promising biomarkers and potential therapeutic targets of many diseases. CircRNA has been reported to be associated with a variety of diseases. For example, Wang et al., reviewed circRNAs that associated with cancers, including gastric cancer, breast cancer, lung cancer and pancreatic cancers [12]. Yang et al., demonstrated that circ_0054633 is highly expressed in lipopolysaccharides (LPS)-induced acute lung injury model both in vivo and in vitro. Silencing of circ_0054633 alleviates LPS-induced ALI via the NF-κB signaling pathway, suggesting that circ_0054633 may be a potential biomarker for the diagnosis and treatment of ALI [30]. In this study, the DE circRNAs in PBLI were selected. The changes in the expression of these circRNAs induced by PBLI indicate that these circRNAs may be involved in PBLI. Which provides a basis for the screening of PBLI diagnostic and treatment markers.
There is growing evidence that circRNAs can act as ceRNA by sponging miRNA by mitigating the inhibition of miRNAs on their target. As the mechanism of PBLI is still unclear, the circRNA related to PBLI is basically not studied, the correlation between circRNA and PBLI is unknown, and the corresponding PBLI database and circRNA-PBLI association prediction methods are lacking. The application of bioinformatics methods to predict DE circRNA-targeted miRNAs and construct ceRNA networks is crucial for screening possible circRNAs and inferring the possible mechanism of action of circRNAs in PBLI. In this study, the circRNA-miRNA-hub gene network consisting of 6 circRNAs, 7 miRNAs and 6 mRNAs was constructed. These circRNA-targeted mRNAs play an important role in inflammation and oxidative stress, which is responsible for the high mortality rate of PBLI [31, 32]. For example, it has been reported that in hepatocellular carcinoma, the expression levels of CDK1 was positively correlated with the infiltration levels of CD4+ T cells, CD8+ T cells, neutrophils, macrophages and dendritic cells, suggesting that this gene may be involved in the recruitment and regulation of infiltrating cells in the immune microenvironment [33]. And in Bleomycin-induced ALI, the protein expression of Top2a was up-regulated, which leads to persistent inflammation and aggravation of alveolar epithelium [34]. Thus, these genes may be involved in immune regulation and inflammation balance in PBLI. And the 6 circRNAs (chr18:21008725–21020999 + , chr4:44893533–44895989 + , chr4:56899026–56910247-, chr5:123709382–123719528-, chr9:108528589–108544977 + and chr15:93452117–93465245 +) that in the circRNA-miRNA-hub gene network may also play important regulatory roles in the progression of PBLI, and may be used as targets for the diagnosis and treatment of PBLI.
As mentioned above, we used bioinformatics methods to explore PBLI-associated circRNAs. At present, more and more computational methods have been widely used in the field of bioinformatics, and computational models can effectively predict potential circRNA-disease associations, which is time-saving and inexpensive. These computational models can be broadly divided into two categories, those based on network algorithms and those based on machine learning [12]. For example, Zhao et al. developed the computational method of IBNPKAT and Ge et al., developed LLCDC [35, 36]. These methods obtain circRNA information, disease information and circRNA-disease associations from the database, and use circRNA similarity and disease similarity to infer potential circRNA-disease relationship. Case studies show that these methods are effective in predicting circRNAs associated with human disease and do not require negative samples. With the help of these computational models, the correlation between circRNAs and disease can be quickly deduced and potential circRNAs can be screened. In addition to circRNAs, computational models successfully developed for other research areas will significantly accelerate the prediction of PBLI related ncRNAs. For instance, based on network distance analysis model (NDALMA), and graph convolutional neural network and conditional random field model (GCNCRF) for lncRNA-miRNA association prediction, as well as based on deep learning predictive model (DMFGAM) for predicting drug-target interactions [37,38,39]. It is believed that these computational biology methods will provide new insights into the molecular mechanism and treatment strategies of PBLI in the future.
Conclusions
In conclusion, this study delineated the expression profiles of circRNAs and constructed circRNA-miRNA-hub gene network in PBLI for the first time, which may provide new ideas for the diagnosis and treatment of PBLI.