Abstract
Background
Magnesium chelatase plays an important role in photosynthesis, but only a few subunits have been functionally characterized in cassava.
Results
Herein, MeChlD was successfully cloned and characterized. MeChlD encodes a magnesium chelatase subunit D, which has ATPase and vWA conservative domains. MeChlD was highly expressed in the leaves. Subcellular localization suggested that MeChlD:GFP was a chloroplast-localized protein. Furthermore, the yeast two-hybrid system and BiFC analysis indicated that MeChlD interacts with MeChlM and MePrxQ, respectively. VIGS-induce silencing of MeChlD resulted in significantly decreased chlorophyll content and reduction the expression of photosynthesis-related nuclear genes. Furthermore, the storage root numbers, fresh weight and the total starch content in cassava storage roots of VIGS-MeChlD plants was significantly reduced.
Conclusion
Taken together, MeChlD located at the chloroplast is not only required for chlorophyll biosynthesis and photosynthesis, but also affecting the starch accumulation in cassava. This study expands our understanding of the biological functions of ChlD proteins.
Similar content being viewed by others
Background
Cassava (Manihot esculenta Crantz) is an important food staple for raising nearly a billion people in the tropics, playing vital functions in food security [1]. Cassava not only used as industrial raw materials, but also act as edible food in China, attributed to its starchy roots [2, 3]. In recent decades, China became a major country of cassava production in the world during the long-term cultivation, the harvest area and yield were increased in China [4]. However, the production of cassava in China could not afford the domestic consumption, self-sufficiency rate was only 18%, most of cassava dry slices and starch dependent on import [5]. Therefore, it is an urgence to solve the disadvantaged situation by cultivating cassava varieties harboring the excellent characteristic with high-yield and high starch accumulation. Furthermore, cassava is a typical source-to-sink tuberous crop [6, 7]. Thus, to better understanding the molecular regulation mechanism of chlorophyll biosynthesis is the initial step for cassava breeding.
Chlorophyll is synthesized from Mg-protoporphyrin IX in the biosynthesis pathway. Magnesium chelatase is the first committed enzyme for Mg-porphyrin branch, which catalyzed Mg2+ in to protoporphyrin IX by consumption of ATP. Magnesium chelatase consists of at least three different subunits, ChlI, ChlH and ChlD subunits [8]. These subunits have been reported in plants, especially in Oryza sativa [9,10,11], Nicotiana benthamiana [12, 13], Arabidopsis thaliana [14, 15], Glycine max [16,17,18], Pisum sativum [19], Zea mays [20], which have specific functions in chlorophyll biosynthesis and photosynthesis. AtChlH protein involves in plastid-to-nucleus signal transduction [21, 22]. Mutations in ChlD also impairs retrograde signaling [23]. Additionally, ChlH and ChlI except ChlD also affect ABA signaling in stomatal guard cell in A. thaliana and N. benthamiana [24, 25]. These results indicated these subunits have multiple roles in plants. Thus, although the regulatory mechanism of chlorophyll biosynthesis is currently being elucidated in many species, there are still many details that required further investigation in cassava.
Virus-induced gene silencing (VIGS) system was widely and successfully used to characterized gene functions in many plants [26,27,28,29,30]. VIGS is mostly drive on RNA viruses, such as tobacco rattle virus (TRV), tobacco mosaic virus (TMV), cucumber mosaic virus (CMV), potato virus X (PVX) and cassava common mosaic virus (CsCMV). Especially, TRV and CsCMV are widely used in cassava for rapid gene function analysis [29,30,31,32], due to high silencing efficiency, long silencing duration, and obvious virus-induced disease symptoms. A magnesium chelatase subunit I (ChlI) was successfully silenced in tobacco and different cassava varieties by CsCMV-mediate VIGS system [30]. Therefore, CsCMV mediated-VIGS system can be used as a new tool for rapidly characterized gene functions in cassava.
In this study, MeChlD was successfully cloned and characterized, which encodes a magnesium chelatase subunit D. The tissue specific expression, subcellular localization, protein interactions and VIGS-inducing the phenotypes were characterized. These results suggested that MeChlD was chlorophyll-localized is required for chlorophyll biosynthesis, photosynthesis and starch metabolism in cassava.
Methods
Plant materials and growth conditions
Cassava variety South China 9 (SC9) was used in this study, which provided from National Cassava Germplasm Repository (Danzhou city, Hainan province). Tobacco (N. benthamiana) was provided by Prof. Yao from Institute of Tropical Bioscience and Biotechnology, Chinese academy of Tropical Agricultural Science, which was used in this study to analyzed subcellular localization and protein interactions. All plants were kept in an air-conditioned growth room (22 °C; 16-h photoperiod, 2000 lx light intensity).
Gene cloning and plasmid construction
Gene sequence of MeChlD was cloned using cDNA as a template. DNA sequences encoding proteins (MeChlD, MeChlM and MePrxQ) without signal peptides were cloned into prokaryotic expression vector pET28a (Novagen, Merck).
To analyze localization of GFP-tagged MeChlD, the coding sequence of MeChlD was PCR-amplified, and then inserted at the NcoI site of the binary vector pCAMBIA1302 (yielding a 35Spro-MeChlD:GFP construct).
To examine the interaction relationship between MeChlD and MeChlM/MePrxQ by using the yeast two-hybrid system, the vectors pGBKT7-MeChlD, pADT7-MeChlM, pADT7-MePrxQ, pGBKT7-MeChlM, pGBKT7-MePrxQ and pADT7-MeChlD were constructed and transformed into yeast competent cell AH109, respectively.
To further confirm the interaction by using BiFC, these vectors pNC-BiFC-Enn-MeChlD with N-terminal enhanced yellow fluorescence protein (nYFP), pNC-BiFC-Ecc-MeChlM and pNC-BiFC-Ecc-MePrxQ with C-terminal enhanced YFP (cYFP) were constructed by seamless cloning (ClonExpress II One Step Cloning Kit, Vazyme Biotech Co., Ltd), and transformed into Agrobacterium tumefaciens GV3101-pSoup-p19.
To investigate the role of MeChlD, 300 bp DNA fragment was PCR-amplified using cDNA of SC9 and finally cloned into the a VIGS vector pCsCMV-NC as described [30], yielding pCsCMV-MeChlD.
All constructs were verified by DNA sequencing. Primer sequences for plasmid construction are shown in Table S1.
Identification and bioinformatic analysis of MeChlD
The MW (molecular weight), pI (theoretical isoelectric point) and positive/negative charge residues of MeChlD were predicted by ExPASy [33]. The ProtScale program (http://web.expasy.org/ protscale/) was used to analyze the hydrophilicity of the ChlD protein. The gene expression of MeChlD was characterized in cassava expression atlas as described [34].
For the identification of MeChlD and related proteins, the amino acid sequence of MeChlD was used as a query sequence for blast at the NCBI (https://blast.ncbi.nlm.nih.gov/Blast.cgi). MEGA X [35] was used to multiple alignment analysis of MeChlD and homologous proteins and a phylogenetic tree was constructed using the Neighbor-joining method.
Measurement of the net photosynthetic rate in cassava accessions
The net photosynthetic rate under nesting scenarios along with the temperature, CO2 concentration, and the photon flux density was measured by LI-6800 portable photosynthesis system (Li-COR company, Nebraska, USA). The required temperature was set 25 °C by temperature controlling module, the CO2 injection volume ratio was set 400 μL/L, the photo flux density was set to 1000 μmol/(m2·s) by LED light source module, the relative humidity was set 50–70% by water control module. Fully expanded functional leaves of cassava were randomly selected. The timing for measuring was selected at 09:00 and 12:00 am to avoid the effects of stomatal closure on this study.
The growth of the recombinant proteins under different concentrations of Mg2+ treatments
The vectors pET-28a-MeChlD, pET-28a-MeChlM, pET-28a-MePrxQ and pET-28a were transformed into E. coil (DE3). These recombinant proteins MeChlD, MeChlM and MePrxQ were treated by different concentrations of magnesium ion as described [36].
Subcellular localization of MeChlD in tobacco leaves
The vectors pCAMBIA1302-35Spro-MeChlD:GFP and pCAMBIA1302 were transformed into A. tumefaciens strain GV3101, and then infiltrated into N. benthamiana leaves as described. After 3 days post inoculation (dpi), GFP signals (395–509 nm) were detected using a laser confocal microscope.
Y2H assay
These vectors pGBKT7-MeChlD/pADT7, pGBKT7-MeChlM/pADT7 and pGBKT7-MePrxQ/pADT7, pGBKT7-MeChlD/pADT7-MeChlM, pGBKT7-MeChlD/pADT7-MePrxQ and pGBKT7-MeChlM/pADT7-MePrxQ were transformed into yeast competent cell AH109, and then the positive clones were screened on the selective SD dropout medium (SD-Leu-Trp and SD-Leu-Trp-His-Ade).
Bimolecular fluorescence complementation (BiFC) analysis
These vectors pNC-BiFC-Enn-MeChlD, pNC-BiFC-Ecc-MeChlM and pNC-BiFC-Ecc-MePrxQ were transformed into A. tumefaciens strain GV3101-psoup-p19, and then infiltrated into N. benthamiana leaves (growth of 30 days) as described [37]. The yellow fluorescence (514–527 nm) in the infiltrated leaf areas was examined using a laser confocal microscope.
VIGS assay in cassava
The best VIGS target region of MeChlD CDS sequence (Manes. 14G005700) was analyzed by online software (SGN VIGS Tool), and then we amplified this region (274–573 bp of CDS sequence) and constructed the vectors pCsCMV-MeChlD. The vectors pCsCMV-MeChlD and pCsCMV-NC were transformed into A. tumefaciens strain GV3101-pSoup-p19, and then infiltrated the leaves of cassava variety SC9 by a syringe injection as described [30].
The VIGS-mediated silent plants were grown in the air-condition room (22 °C; 16-h photoperiod). After 3 weeks post inoculation, the expression level of MeChlD in silent plants of cassava was measured by qRT-PCR to further calculate the silence efficient. Currently, chlorophyll content in different silent plants of cassava was performed as described [38]. The content of the total starch was measured as described [32].
RNA extraction and qRT-PCR verification
Total RNA was extracted from cassava leaves using a RNAprep Pure Plant plus Kit (Tiangen, China). One-Step gDNA Removal and cDNA Synthesis SuperMix (TransGen, China) was used for first-strand cDNA synthesis. The reactions solutions were performed in 10 mL volume in Real-time Thermal Cycler fluorescence quantification apparatus (Thermo Fisher Scientific Inc., Göteborg, Sweden). MeActin was used as an internal reference gene. All samples were performed in triplicate per sample. The relative expression levels of genes were calculated by the formula 2−ΔΔCT method. The qRT-PCR primers were shown in Table S1.
Statistical analysis
Microsoft Excel 2010 was used for data processing; SPSS 23.0, DPS 7.0 and Kruskal–Wallis software was used for data analysis; GraphPad 6.0 and Origin2021 software were used to create experimental figures.
Results
MeChlD encodes a magnesium chelatase subunit D
A magnesium chelatase subunit D, MeChlD, was successfully amplified and sequencing in cassava variety SC9 (Figure S1). The length of coding sequence is 2250 bp, encodes 749 amino acids. The information of MV, pI and positive/negative charge residues were 82.27 kDa, 5.81, 100 and 92, respectively (Table S2). Bioinformatic analysis indicated that MeChlD belongs to stable protein and hydrophilic protein (Figure S2).
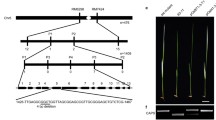
To further characterize ChlD protein, we performed a study with ChlD of M. esculenta and homologues from HbChlD (Hevea brasiliensis), RcChlD (Ricinus communis), AtChlD (A. thaliana), NtChlD (N. tabacum), OsChlD (Oryza sativa), GsChlD (G. soja), CsChlD (Citrus sinensis) and GhChlD (Gossypium hirsutum). An amino acid sequence alignment of these proteins and a corresponding phylogenetic tree is shown (Fig. 1). Most of these ChlD proteins have conservative domains, ATPase domain without ATPase activity, and vWA structural domain with binding site for metal ion (Fig. 1a). MeChlD shows more than 81% identities with these proteins, especially has the closer relationship with HbChlD (96.74% identities) and RcChlD (90.60% identities) (Fig. 1b).
Alignment and phylogenetic analysis of MeChlD and MeChlD-related proteins in plants. a Alignment of amino acid sequences of MeChlD and MeChlD-related proteins. The alignment was performed with GENEIOUS software (https://www.geneious.com/). Identical amino acid residues are indicated with a black background, homologous residues with a grey background and dashes indicate gaps. The blue line and green line indicate ATPase domain and vWA structural domain, respectively. b A corresponding phylogenetic tree of MeChlD and related amino acid sequences. MeChlD characterized in this study are highlighted in blue. The tree was constructed by using the Neighbor-joining method. The horizontal bar represents a distance of 0.03 substitutions per site
Mg2+ affects the growth of the recombinant protein MeChlD
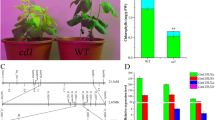
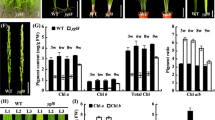
Magnesium as an essential nutrient for plant growth and development, involved in the photosynthesis process. To investigate the roles of MeChlD in photosynthesis, ChlM and PrxQ were selected as positive controls [30, 32]. In this study, VIGS-induced reduction of MeChlD expression resulted in mosaic leaves or fully yellow leaves of VIGS-MeChlD plants compared with VIGS-NC control plants (Fig. 6b). This finding could be attributed to the reduction in chlorophyll content (Fig. 6c). These changes were consistent with those in previous studies on VIGS in Mg-chelatase subunits [17, 19, 30]. Furthermore, the changes in chlorophyll biosynthesis were significantly correlated with the reduced expression of photosynthesis-related genes (Fig. 6d), but not of MeChlH silencing. In this study, the expression of MeChlI was significantly reduced in VIGS-MeChlD plants, consistent with the result of previous studies, which showed that ChlI stabilizes ChlD, and acts as a chaperone [45]. The transcript level of MeChlH was significantly up-regulated in VIGS-MeChlD plants, and similar results have been reported in Arabidopsis ChlM mutant [46]. However, the different results indicated that ChlH accumulation without changing the expression of VIGS-ChlD plants in pea [17]. These findings suggested that the regulation of ChlH is a complex process.
To our knowledge, cassava is a typical source-to-sink tuberous crop, and its sink capacity depends on the delivery of forms of sources [47]. In this study, the phenotype of yellow leaves or mosaic leaves was observed after 4 months of plantation in the field (Fig. 7a). The content of total starch, the number and fresh weight of cassava storage roots (4 months) in VIGS-MeChlD-2 plants significantly decreased compared with those in VIGS-NC control plants (Fig. 7b). This result indicated that the effect of VIGS could be maintained for at least 4 months in cassava storage roots, thus proving the hypothesis as described [30]. In conclusion, MeChlD not only possesses an important role in source biosynthesis, but also affects starch accumulation in cassava.
Availability of data and materials
The sequence of MeChlD protein analyzed during the current study are available in the UniPort repository (Accession number: A0A2C9UIM7; https://www.uniprot.org/uniprotkb/A0A2C9UIM7/entry).
References
Cai J, Zhang J, Ding Y, Yu S, Lin HX, Yuan ZQ, et al. Different fertilizers applied alter fungal community structure in rhizospheric soil of cassava (Manihot esculenta Crantz) and increase crop yield. Front Microbiol. 2021;12: 663781.
Zhu F. Composition, structure, physicochemical properties, and modifications of cassava starch. Carbohyd Polym. 2015;122:456–80.
De Souza AP, Massenburg LN, Jaiswal D, Cheng S, Shekar R, Long SP. Rooting for cassava: insights into photosynthesis and associated physiology as a route to improve yield potential. New Phytol. 2017;213:50–65.
Hailing Fu, Yi Qu, Pan Yi. Efficiency of cassava production in China: empirical analysis of field surveys from six provinces. Appl Sci. 2018;8:1356.
Liang HB, Huang J, An FF, Wei YX. Analysis of present situation of cassava industry in China. Acta Agriculturae Jiangxi. 2016;28:22–6.
Alves AAC. “Cassava botany and physiology,” in Cassava: Biology, Production and Utilization, eds R. J. Hillocks, J. M. Thresh, and A. Bellotti (Wallingford: CABI), 2022:67–89.
Yan W, Wu XY, Li YN, Liu GH, Cui ZF, Jiang TL, et al. Cell wall invertase 3 affects cassava productivity via regulating sugar allocation from source to sink. Front Plant Sci. 2019;10:541.
Masuda T. Recent overview of the Mg branch of the tetrapyrrole biosynthesis leading to chlorophylls. Photosynthesis Res. 2008;96:121–43.
Zhou S, Sawicki A, Willows RD, Luo M. C-terminal residues of Oryza sativa GUN4 are required for the activation of the ChlH subunit of magnesium chelatase in chlorophyll synthesis. FEBS Lett. 2012;586:205–10.
Zhang H, Liu L, Cai M, Zhu S, Zhao J, Zheng T, et al. A point mutation of magnesium chelatase OsCHLI gene dampens the interaction between CHLI and CHLD subunits in rice. Plant Mol Biol Rep. 2015;33:1975–87.
Luo S, Luo T, Liu Y, Li Z, Fan S, Wu C. N-terminus plus linker domain of Mg-chelatase D subunit is essential for Mg-chelatase activity in Oryza sativa. Biochem Bioph Res Co. 2018;497:749–55.
Papenbrock J, Gräfe S, Kruse E, Hänel F, Grimm B. Mg-chelatase of tobacco: identification of a Chl D cDNA sequence encoding a third subunit, analysis of the interaction of the three subunits with the yeast two-hybrid system, and reconstitution of the enzyme activity by co-expression of recombinant CHLD. CHLH and CHLI The Plant J. 1997;12:981–90.
Papenbrock J, Pfündel E, Mock HP, Grimm B. Decreased and increased expression of the subunit CHLI diminishes Mg chelatase activity and reduces chlorophyll synthesis in transgenic tobacco plants. The Plant J. 2000;22:155–64.
Rissler HM, Collakova E, DellaPenna D, Whelan J, Pogson JB. Chlorophyll biosynthesis. expression of a second Chl I gene of magnesium chelatase in Arabidopsis supports only limited chlorophyll synthesis. Plant Physiol. 2002;128:770–9.
Kobayashi K, Mochizuki N, Yoshimura N, Motohashi K, Hisabori T, Masuda T. Functional analysis of Arabidopsis thaliana isoforms of the Mg-chelatase CHLI subunit. Photoch Photobio Sci. 2008;7:1188–95.
Nakayama M, Masuda T, Bando T, Yamagata H, Ohta H, Takamiya KI. Cloning and expression of the soybean chlH gene encoding a subunit of Mg-chelatase and localization of the Mg2+ concentration-dependent ChlH protein within the chloroplast. Plant Cell Physiol. 1998;39:275–84.
Luo T, Luo S, Araújo WL, Schlicke H, Rothbart M, Yu J, et al. Virus-induced gene silencing of pea CHLI and CHLD affects tetrapyrrole biosynthesis, chloroplast development and the primary metabolic network. Plant Physiol Biochem. 2013;65:17–26.
Zhang D, Chang E, Yu X, Chen Y, Yang Q, Cao Y, et al. Molecular characterization of Magnesium Chelatase in soybean [Glycine max (L.) Merr.]. Front Plant Sci. 2018;9:720.
Wu CJ, Wang J, Zhu J, Ren J, Yang YX, Luo T, et al. Molecular characterization of Mg-chelatase CHLI subunit in pea (Pisum sativum L.). Front Plant Sci. 2022;13:821683.
Sawers RJ, Viney J, Farmer PR, Bussey RR, Olsefski G, Anufrikova K, et al. The maize Oil yellow1 (Oy1) gene encodes the I subunit of magnesium chelatase. Plant Mol Bio. 2006;60:95–106.
Mochizuki N, Brusslan JA, Larkin R, Nagatani A, Chory J. Arabidopsis genomes uncoupled 5 (GUN5) mutant reveals the involvement of Mg-chelatase H subunit in plastid-to-nucleus signal transduction. P Nat Acad Sci USA. 2001;98:2053–8.
Nott A, Jung HS, Koussevitzky S, Chory J. Plastid-to-nucleus retrograde signaling. Annu Rev Plant Biol. 2006;57:739–59.
Strand A, Asami T, Alonso J, Ecker JR, Chory J. Chloroplast to nucleus communication triggered by accumulation of Mg-protoporphyrinIX. Nature. 2003;421:79–83.
Tsuzuki T, Takahashi K, Inoue SI, Okigaki Y, Tomiyama M, Hossai MA, et al. Mg-chelatase H subunit affects ABA signaling in stomatal guard cells, but is not an ABA receptor in Arabidopsis thaliana. J Plant Res. 2011;124:527–38.
Du SY, Zhang XF, Lu Z, **n Q, Wu Z, Jiang T, et al. Roles of the different components of magnesium chelatase in abscisic acid signal transduction. Plant Mol Biol. 2012;80:519–37.
Wang XY, LÜ K, Cai CP, Xu J, Guo WZ. Establishment and application of TRV-mediated virus-induced gene silencing in cotton. Acta Agronomica Sinica. 2014;40:1356.
Tian J, Pei HX, Zhang S, Chen JW, Chen W, Yang RY, et al. TRV-GFP: a modified tobacco rattle virus vector for efficient and visualizable analysis of gene function. J Exp Bot. 2014;65:311–22.
Anu K, Jessymol KK, Chidambareswaren M, Gayathri GS, Manjula S. Down-regulation of osmotin (PR5) gene by virus-induced gene silencing (VIGS) leads to susceptibility of resistant Piper colubrinum to the oomycete pathogen Phytophthora capsici Leonian. Indian J Exp Bot. 2015;53:329–34.
Zeng H, **e Y, Liu G, Wei Y, Hu W, Shi H. Agrobacterium-mediated gene transient overexpression and tobacco rattle virus (TRV)-based gene silencing in cassava. Int J Mol Sci. 2019;20:3976.
Tuo D, Zhou P, Yan P, Cui H, Liu Y, Wang H, et al. A cassava common mosaic virus vector for virus-induced gene silencing in cassava. Plant Methods. 2021;17:1–12.
Li X, Liu W, Li B, Liu G, Wei Y, He C, Shi H. Identification and functional analysis of cassava DELLA proteins in plant disease resistance against cassava bacterial blight. Plant Physiol Biochem. 2018;124:70–6.
An F, **ao X, Chen T, Xue J, Luo X, Ou W, Li KM, Cai J, Chen SB. Systematic analysis of bHLH transcription factors in cassava uncovers their roles in postharvest physiological deterioration and cyanogenic glycosides biosynthesis. Front Plant Sci. 2022;13:901128.
Artimo P, Jonnalagedda M, Arnold K, Baratin D, Csardi G, De Castro E, et al. ExPASy: SIB bioinformatics resource portal. Nucleic Acids Res. 2012;40:597–603.
Wilson MC, Mutka AM, Hummel AW, Berry J, Chauhan RD, Vijayaraghavan A, et al. Gene expression atlas for the food security crop cassava. New Phytol. 2017;213:1632–41.
Kumar S, Stecher G, Li M, Knyaz C, Tamura K. MEGA X: molecular evolutionary genetics analysis across computing platforms. Mol Biol Evol. 2018;35:1547–9.
Zhou SX. Kinetic and catalytic analysis of the magnesium chelatase in plants. doctoral dissertation, Wuhan: Huazhong agricultural university, China, 2012.
Sparkes IA, Runions J, Kearns A, Hawes C. Rapid, transient expression of fluorescent fusion proteins in tobacco plants and generation of stably transformed plants. Nat Protoc. 2006;1:2019–25.
Chaiareekitwat S, Latif S, Mahayothee B, Khuwijitjaru P, Nagle M, Amawan S, Müller J. Protein composition, chlorophyll, carotenoids, and cyanide content of cassava leaves (Manihot esculenta Crantz) as influenced by cultivar, plant age, and leaf position. Food Chem. 2022;372: 131173.
**ao LG, Yang RC. Molecular cloning and characterization of a stress-induced peroxiredoxin Q gene in halphyte suaeda salsa. Plant Sci. 2004;167:969–75.
Wang Z, Hong X, Hu K, Wang Y, Wang X, Du S, et al. Impaired magnesium protoporphyrin IX methyltransferase (ChlM) impedes chlorophyll synthesis and plant growth in Rice. Front Plant Sci. 2017;8:1694.
Moss DN, Musgrave RB. Photosynthesis and crop production. Adv Agron. 1971;23:317–36.
Croce R, van Amerongen H. Natural strategies for photosynthetic light harvesting. Nat Chem Biol. 2014;10:492–501.
Hu W, Ji C, Shi H, Liang Z, Ding Z, Ye J, et al. Allele-defined genome reveals biallelic differentiation during cassava evolution. Mol Plant. 2021;14:851–4.
Nyaboga E, Njiru J, Nguu E, Gruissem W, Vanderschuren H. Unlocking the potential of tropical root crop biotechnology in east Africa by establishing a genetic transformation platform for local farmer-preferred cassava cultivars. Front Plant Sci. 2013;4:526.
Lake V, Olsson U, Willows RD, Hansson M. ATPase activity of magnesium chelatase subunit I is required to maintain subunit D in vivo. Eur J Biochem. 2004;271:2182–8.
Huang YS, Li HM. Arabidopsis CHLI2 can substitute for CHLI1. Plant Physiol. 2009;150:636–45.
Lemoine R, Camera SL, Atanassova R, Dédaldéchamp F, Allario T, Pourtau N, et al. Source-to-sink transport of sugar and regulation by environmental factors. Front Plant Sci. 2013;4:272.
Acknowledgements
We would like to thank Dr. Wentao Shen, Pu Yan and Decai Tuo for sharing the VIGS vectors. We also acknowledge the technical support for using LI-6800 portable photosynthesis system provided Dr. Jungang Wang and Dr. Xu Shen.
Funding
This work was financially supported by the National Key Research and Development Program of China (No. 2019YFD1000500), Hainan Provincial Natural Science Foundation of China (320RC730), Central Public-interest Scientific Institution Basal Research Fund for Chinese Academy of Tropical Agricultural Sciences (No. 1630032022007) and earmarked fund for China Agriculture Research System (CARS-11-HNLKM).
Author information
Authors and Affiliations
Contributions
CSB and LKM designed the experiments; YXA performed the experiment; YXA, CJ, XJJ, LXQ, ZWL, XXH, AFF analyzed the data; XMF provided plant materials; CJ wrote the manuscript, AFF, CJ and YXA and revised the manuscript. All authors have agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
The plant experiments were performed in accordance to institutional guidelines. The experiments on live vertebrates and/or higher invertebrate were not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1:
Table S1. Primers were used in this study.
Additional file 2:
Table S2. The information of MV, pI and positive/negative charge residues of MeChlD.
Additional file 3:
Figure S1. MeChlD was successfully amplified in SC9. M: DL 15000 bp Marker; Line 1-3: the amplification bands of MeChlD CDS
Additional file 4:
Figure S2. Physical and chemical properties and structure analysis of protein. (a) MeChlD protein affinity and hydrophobicity prediction analysis chart. (b) Secondary structure of MeChlD protein. (c) Tertiary structure of MeChlD protein
Additional file 5:
Figure S3. The auto-inducing activities of MeChlD, MeChlD and MerPrxQ were detected in yeast.
Additional file 6:
Figure S4. The Y2H assay analysis of MeChlD, MerPrxQ and MeChlD in vitro.
Additional file 7:
Figure S5. qRT-PCR analysis of the expression of MeChlD, in the yellow leaves of MeChlD-1 and MeChlD-3 plants.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Yang, X., Cai, J., Xue, J. et al. Magnesium chelatase subunit D is not only required for chlorophyll biosynthesis and photosynthesis, but also affecting starch accumulation in Manihot esculenta Crantz. BMC Plant Biol 23, 258 (2023). https://doi.org/10.1186/s12870-023-04224-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12870-023-04224-9