Abstract
Background
Healthcare workers may pave the way for increased infections in hospitalized patients by coagulase-negative staphylococci (CoNS). Biofilm formation and antibiotic resistance are the major problems posed by CoNS in nosocomial infections. In this study, we determined biofilm production level and the distribution of biofilm-associated and virulence genes, including icaADBC, aap, bhp, atlE, embp, and fbe, as well as IS256, IS257, mecA, and ACME clusters (arc-A, opp-3AB) among 114 clinical (n = 57) and healthcare workers (n = 57) CoNS isolates in Kerman, Iran.
Results
In this study, more than 80% (n = 96) of isolates were methicillin-resistant CoNS (MR-CoNS). Out of 114 isolates, 33% (n = 38) were strong biofilm producers. Strong biofilm formation was found to be significantly different between clinical and healthcare workers’ isolates (P < 0.050). In addition, 28% (n = 32) of isolates were positive for icaADBC simultaneously, and all were strong biofilm producers. The prevalence of icaADBC, mecA, bhp, fbe, and IS256 in clinical isolates was higher than that in healthcare workers’ isolates (P < 0.050). A significant relationship was observed between clinical isolates and the presence of icaADBC, mecA, bhp, and IS256. Although these elements were detected in healthcare workers’ isolates, they were more frequent in clinical isolates compared to those of healthcare workers.
Conclusions
The high prevalence of ACME clusters in healthcare workers’ isolates and biofilm formation of these isolates partially confirms the bacterial colonization in the skin of healthcare workers. Isolating MR-CoNS from healthcare workers’ skin through similar genetic elements to clinical isolates, such as icaADBC, mecA, and IS256, calls for appropriate strategies to control and prevent hospital infections.
Similar content being viewed by others
Introduction
Given the increased infection risk factors in hospitalized patients, it is crucial to understand the presence of virulence genes and the prevalence of antibiotic resistance among bacteria that cause nosocomial infections [1]. Coagulase-negative staphylococci (CoNS) isolates are important opportunistic pathogens among hospitalized patients. In addition, Staphylococcus isolates are one of the major skin and mucous membrane microbiota [2, 3]. According to a previous study, CoNS isolates can spread among healthcare workers and hospitalized patients [3,4,5,6].
Various genes, including icaADBC (intracellular adhesion) operon, fbe (fibrinogen binding protein), bhp (bap homologous- protein), embp (extracellular matrix-binding protein), aap (accumulation-associated protein), and atlE (autolysin), are involved in biofilm formation, mediate the initial adhesion, and bind specifically to fibrinogen, fibronectin, and collagen. The ica operon can be regulated by insertion sequence (IS) elements. The integration of IS elements can alter genes expression, and IS256 confers phase variation phenomena concerning biofilm formation, modulation of antibiotic resistance and virulence genes in S. aureus and CoNS isolates [7,8,9,10,11].
Biofilms has considerably affect on evasion of host immune system and resistance to antibiotics in staphylococcal infections [12,13,14]. Biofilm formation and colonization on medical devices make the CoNS isolates one of the most frequent causes of nosocomial infections in hospitalized patients [12,13,14]. Nowadays, most clinical CoNS isolates, such as S. epidermidis and S. haemolyticus, are methicillin-resistant (MR-CoNS) and are commonly resistant to different antibiotic agents [3, 14].
Typically, the biofilm formation in CoNS isolates is based on the polysaccharide intercellular adhesion (PIA) matrix, which is also known as poly-N-acetyl-glucosamine (PNAG). This polysaccharide matrix is synthesized with proteins encoded by the icaADBC operon in Staphylococci species [12, 15]. On the other hand, the microbial surface components, which recognize adhesive matrix molecules (MSCRAMMs), such as Fbe and Embp, can bind to fibrinogen and fibronectin, respectively, and have an important role in attachment to the cells and biofilm production in CoNS [13]. Also, other virulence genes, such as atlE, bhp, and aap, are commonly found in S. epidermidis isolates and act as initial attachment factors in biofilm formation [6, 7]. Identifying and targeting polysaccharides and proteins involved in adhesion and biofilm formation can be suitable candidates for vaccine production that prevent staphylococcal infections [16,17,18]. The vaccine can be effective in preventing the infection. Although there is a good body of research on this issue, further practical studies should be undertaken on the vaccine to prevent infections [16,17,18].
The arginine catabolic mobile element (ACME) consists of two gene clusters: the arc-operon encoding a secondary arginine deaminase system and the opp3-operon encoding a putative oligopeptide permease system [19, 20]. These elements are homologs of virulence determinants in other bacterial species and enhance bacterial adaptability and colonization [20, 21]. The horizontal transfer of the ACME in intra-species of Staphylococci was confirmed in previous studies [19, 20]. However, the ACME was shown to contribute to the colonization and survival of CoNS isolates in hospital settings. ACME allotypes were classified as (1) ACME-I containing the arc and the opp-3 gene clusters, (2) ACME-II containing the arc, and (3) ACME-III containing the opp-3 [20, 22]. The transmission of infectious agents by healthcare workers was observed in hospital settings [2, 3]. Therefore, it is important to study the characteristics of typical flora isolates from hospital staff to control infection in hospitals. The present study determines biofilm formation ability and the prevalence of biofilm-associated and virulence genes, IS256, IS257, mecA, and ACME clusters in CoNS isolates collected from clinical staff and healthcare workers’ skin in Kerman, Iran
Results
Bacterial strains and methicillin-resistant CoNS detection
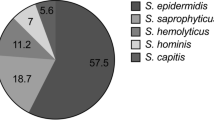
In total, 114 CoNS isolates were collected in this study. Different species of CoNS isolates, including S. epidermidis 77.2% (n = 88), S. haemolyticus 16.6% (n = 19), S. hominis 5% (n = 6), and S. saprophiticus 1% (n = 1), were detected. Among 114 clinical and healthcare workers’ CoNS isolates, 66.5% (n = 76), 10.5% (n = 12), 20% (n = 23), 6% (n = 7), 3.6% (n = 4), and 1.8% (n = 2) were collected from neonatal intensive care units (NICU), intensive care unit (ICU), infectious disease units (ID), pediatric intensive care units (PICU), bone marrow transplant (BMT), and oncology units, respectively. In addition, 96.5% (n = 56) of clinical and 70% (n = 40) of healthcare workers’ isolates were resistant to cefoxitin and considered MR-CoNS. All MR-CoNS isolates were positive for mecA.
Biofilm assay
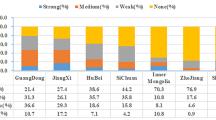
Biofilm formation was observed at strong, moderate, and weak levels in 96.5% (n = 55) of clinical and 91% (n = 52) of healthcare workers’ isolates in this study. Among clinical samples, 46% (n = 26) of isolates were categorized as strong biofilm producers, 37% (n = 21) were moderate biofilm producers, 14% (n = 8) were weak biofilm producers, and two isolates were non-biofilm producers. In healthcare workers’ isolates, 21% (n = 12) were strong biofilm forming, 30% (n = 17) were moderate biofilm forming, 40% (n = 23) were weak biofilm forming, and 9% (n = 5) isolates were non-biofilm forming. Also, the prevalence of strong biofilm formation in clinical isolates was higher than that of healthcare workers’ isolates (P < 0.004). Among 88 S. epidermidis, 40% (n = 36), 23% (n = 20), and 36% (n = 32) were strong, moderate, and weak biofilm producers, respectively. Moreover, among 19 S. heamolyticus, 89% (n = 17) and 10% (n = 2) were moderate and weak biofilm producers, respectively. Two S. hominis were strong, and 3 were moderate biofilm producers.
Detection of ica operon, ACME type, IS, and virulence genes
The profile of icaADBC, virulence genes, and methicillin resistance among biofilm producer of CoNS isolates are presented in Tables 1 and 2. The rate of icaA and icaB was 33% (n = 38), while the prevalence of icaC and icaD was 34% (n = 39) and 37.7% (n = 43), respectively. In clinical isolates, 42% (n = 24) were positive for icaADBC, while 17% (n = 10) of healthcare workers’ isolates were positive for this operon. There was a significant relationship between icaD and mecA in both groups of isolates, among 42 isolates were positive for icaD, 93% (n = 39) were positive for mecA simultaneously (P < 0.030). The fbe and bhp were detected in 11.4% (n = 17) and 6% (n = 7) of isolates, respectively. In addition, the prevalence of embp, aap, and atlE was found to be 55% (n = 63), 53.5% (n = 61), and 51% (n = 58) in isolates, respectively as shown in Table 3 (Fig. 1).
We did not observe a significant difference between clinical and healthcare workers’ isolates in terms of embp, aap, and atlE prevalence. The prevalence of IS257 was higher than IS256 in both clinical and healthcare workers’ isolates (83% vs. 46%). The frequency of fbe (23% vs. 7%) and IS256 (79% vs. 14%) in clinical isolates was higher than the one in healthcare workers’ isolates (P < 0.050). The distribution of icaA, icaB, icaC, icaD, and biofilm-associated virulence genes (fbe, bhp, embp, aap, and atlE) in clinical and healthcare workers’ isolates are presented in Table 3.
Among 37 strong biofilm producers, 32 isolates were positive for icaADBC, and we observed a significant relationship between the presence of icaADBC operon and strong biofilm production in isolates (P < 0.001). Furthermore, in moderate and weak biofilm-forming isolates, the frequency of icaADBC was lower compared to strong biofilm-producing isolates. The prevalence of aap in the moderate biofilm producers (79%, 30 out of 38) was higher compared with other isolates. Biofilm-associated genes among the biofilm-producing clinical and healthcare workers’ isolates are presented in Table 4. Fbe, bhp, embp, and IS256 were more frequently present in icaAD-positive isolates than in icaAD-negative isolates (P < 0.030).
In total, 31 isolates were positive for ACME clusters, including 40% (n = 23) of healthcare workers’ isolates and 14% (n = 8) of clinical isolates. Among the 23 healthcare workers’ ACME-positive isolates, 50% (n = 12) harbored ACME-III (opp-3AB positive), 39% (n = 9) harbored ACME-II (arcA positive), and 8.6% (n = 2) were ACME-І (arcA and opp-3AB positive). In addition, 8 clinical isolates were positive for ACME clusters, among which 37% (n = 3) were ACME-І and ACME-III, and 25% (n = 2) were ACME-II (Fig. 2). The frequency of ACME in healthcare workers’ isolates was considerably higher than that in clinical isolates (P < 0.001) (Table 3). We should note that the isolates with ACME were biofilm producers (strong, moderate, and weak), though no association was detected between ACME genes and strong biofilms or icaADBC. In ACME-harboring isolates, embp (64%, n = 20), aap (67%, n = 21), and atlE (61%, n = 19) were significantly prevalent compared to ACME-negative isolates (P < 0.010) (Fig. 3).
Discussion
In therapeutic and infection control strategies, it is important to identify the genetic and phenotypic characteristics of CoNS that cause nosocomial infections, including the presence of virulence genes, biofilm production, and antibiotic resistance [23]. Several characteristics of CoNS are advantageous for the pathogenesis, colonization, and transmission of these bacteria [13, 20]. CoNS are the reservoir of genes in hospitals and are considered in gene dissemination to other bacteria, and healthcare workers have an important role in transmitting these bacteria to hospitalized patients [24, 25]. In addition, MR-CoNS are often resistant to other antibiotics and present challenges in hospitals, especially in intensive care units [3]. In this study, we investigated biofilm formation and identified the biofilm-associated and virulence genes as well as IS256, IS257, mecA, and ACME clusters in CoNS clinical and healthcare workers’ isolates.
Biofilm formation is the most important mechanism of pathogenicity in CoNS isolates [3]. We found a significant difference between clinical and healthcare workers’ isolates in producing strong biofilm (P < 0.001), a finding that agrees with other studies [24, 26]. In addition, the prevalence of icaADBC was higher in clinical isolates than in healthcare workers’ isolates (42% vs. 12%) (P < 0.030). our study, similar to some other studies, 90% (n = 34) of strong biofilm-producer isolates were positive for icaADBC, which showed an association between ica operon and strong biofilm formation in isolates (P < 0.001) [25, 27, 28].
The co-existence of icaA and icaD increases N-acetyl-glucosaminyl-transferase activity for synthesizing polysaccharide intercellular adhesion (PIA) oligomers and improves biofilm formation [12]. According to earlier studies, PIA is considered a putative candid for vaccine development, though more studies on staphylococcal virulence factors are warranted [16,17,18]. In this study, 91% (n = 42) of healthcare workers’ isolates were biofilm producers, and 23.8% (10/42) were positive for icaADBC. This finding should be considered in preventing and controlling infection in hospitals. Our results showed that 63% (5/8) and 43% (20/46) of wound and blood infection samples were strong biofilm producers associated with icaAD and other virulence genes, such as embp and aap. This finding is not unusual because biofilm formation of Staphylococci on a medical device is common in hospital settings [29, 30]. More than 90% of the clinical isolates of Staphylococcus epidermidis with icaD and strong biofilm phenotype also carried mecA and we observed an association between the presence of the icaD and the presence of the mecA in these isolates. Other studies have reported the presence of the ica operon genes and the simultaneous presence of the mecA gene [24, 25]. Although the presence of ica-operon have the key role in strong biofilm producer isolates in our study, other isolates formed biofilm at strong, moderate, or weak levels in non-associated ica-operon. Therefore, biofilm production in staphylococci is associated with both ica operon and proteins, such as MSCRAMMs, which play an important role in biofilm formation in these bacteria [12, 13].
The adhesive surface proteins have an important role in biofilm formation in associated or non-associated ica genes [12]. In our results, fbe was detected in 23% of clinical, which was about similar to a study in India (20%) and lower than other studies in Germany (100%), and Iran (89.9%) [5, 31, 32]. Moreover, the frequency of bhp (6%) was lower than other studies in Iran (15.3%) and Germany (18.8%), also a study in India indicated none of the strong biofilm formers was positive for bhp [5, 31, 32]. In addition, 65% (47 of 72) of strong and moderate biofilm-producer isolates were positive for aap in our study. In our findings, fbe and bhp were detected in strong and moderate biofilm-producer isolates. By contrast, other studies in India and Brazil reported these genes in moderate biofilm-producer isolates or the ubiquitous presence of these genes in strong, moderate, and non-biofilm-producer isolates [5, 13]. In our study, aap, a gene encoded accumulating associated protein, was detected in 55% (n = 63) of isolates, this rate is difference from other studies in Iran (94.8% and 64.4%), Germany (93.8%) and India (10%) [5, 31,32,33]. In the present study, 73% (56 out of 76) of strong and moderate biofilm-producer isolates were positive for aap, indicating this protein’s importance in biofilm formation. aap was detected in clinical and healthcare workers’ isolates without significant differences. We found that the prevalence of atlE (51%) and embp (55%) differed from other studies in India and Brazil (30–100%), this discrepancy may be due to different sources of specimens in studies [5, 13]. Among 37 strong biofilm isolates that were positive for icaA and icaD, 20 (54%) isolates were positive for IS256, indicating an association between icaAD, biofilm formation, and IS256. This finding is similar to the results obtained by Petrelli et al. [34]. In total, the frequency of icaA, icaD, and IS256 was significantly higher in MR and clinical isolates in comparison with methicillin-sensitive and healthcare workers’ isolates, which indicates an association among mecA, icaA, icaD, and IS256, similar to earlier studies [25, 32, 35]. In addition, our study supported the idea that IS256 acts as a marker for clinical isolates [4, 25]. In our study, 40% of healthcare workers’ isolates were ACME-positive. The high prevalence of ACME among commensal Staphylococcus species notably affects the growth, survival, colonization, and spread of CoNS among healthcare workers in hospital settings [20]. Also, eight MR-clinical biofilm-producer isolates from blood and wound infections harbored ACME elements, and more than 70% carried icaADBC, embp, atlE, and aap.
However, CoNS are part of normal skin flora. The presence of virulence factors related to the attachment alone does not confirm the pathogenicity of this bacteria, and other factors should be considered. On the other hand, the presence and expression of a set of genes and environmental factors, such as temperature and osmolality, contribute to pathogenicity and biofilm formation in Staphylococci [36,37,38]. We detected all studied genetic elements in both clinical and healthcare workers’ isolates. Our results showed that all the ACME-positive isolates were biofilm producers, which increased their survival rate in the hosts. In our study, the high ACME prevalence in healthcare workers’ isolates and its dissemination might contribute to the spread of isolates due to the direct contact of contaminated healthcare workers’ hands with patients.
Conclusion
In this study, we compared biofilm formation and the frequency of genes involved in biofilm formation, virulence genes, and IS256, IS257, and ACME in clinical and healthcare workers’ CoNS isolates. We found a significant relationship between clinical isolates and the presence of icaADBC, mecA, and IS256. We detected some transposable elements contributed to pathogenicity, colonization, and transmission in CoNS isolates. In addition, strains with similar characteristics were found in the clinical and healthcare workers’ isolates, indicating the need for strategies to control and prevent infection in hospitals.
Materials and methods
Bacterial strains and methicillin-resistant CoNS detection
In this study, 114 CoNS isolates were collected from clinical samples (n = 57) and volunteer healthcare workers’ skin (n = 57) from January 2019 to December 2021. The healthcare workers included the nurses and physicians at Afzalipour referral hospital in Kerman, Iran. We excluded staff that cleaned their hands with antiseptics at the time of analysis. Clinical samples were isolated from blood, cerebrospinal fluid (CSF), and wounds. Systemic inflammation, absolute neutrophil count (ANC), C-reactive protein (CRP) levels, mono-microbial growth in cultures, and isolate detection from the second blood culture were the laboratory criteria to distinguish true infection and contamination [39]. The isolates were identified using standard biochemical tests, including colony morphology on Baird–Parker agar (Merck, Co, Germany), Gram staining, catalase production, oxidase test, coagulase-negative tests, susceptibility to novobiocin, and the utilization of xylose, arabinose, sucrose, maltose, mannitol, lactose, ribose, fructose, and mannose. Finally, they were confirmed as CoNS by multiplex-PCR, as described by Hirotaki et al. [40]. The multiplex-PCR reaction was performed in a volume of 50 µL containing: 25 µL of Taq DNA Polymerase Master Mix RED (Ampliqon, Co, Denmark), 2 µL DNA template, 0.25 µL of each primer (10pM), and DNase and RNase free water top up to 50 µL. multiplex-PCR amplification was carried out under the following conditions an initial denaturation step 5-min at 95 °C followed by 30 cycles (30 s of denaturation at 95 °C, 30 s of annealing at 58 °C, and 70 s of extension at 72 °C) and a final elongation step at 72 °C for 5 min. Staphylococcus epidermidis RP62A was used as a positive control strain in the M-PCR technique.
According to the clinical & laboratory standards institute (CLSI), cefoxitin (FOX, 30 µg) disc was used to screen methicillin-resistant isolates (MR-CoNS) [41]. Then, mecA gene was determined using the polymerase chain reaction (PCR) method in MR-CoNS, as previously described by Ruzauskas et al. [42].
Biofilm assay
Biofilm formation assay was determined according to the microtiter plate method described by Stepanovic et al. [43]. Briefly, The culture of each isolates was prepared in BHI medium overnight and adjusted to 0.5 McFarland. After incubation, the stationary-phase culture is vortexed and diluted 1:100, in medium for bioflim cultivation (TSB supplemented with glucose). The diluted bacteria are vortexed and then inoculated into a microtiter plate (200 mL per well). The microplates were incubated overnight for 24 h at 37 ̊C. Then, the wells were washed with PBS and fixed with methanol. The wells were stained with crystal violet and then Ethanol 95% were added to each wells. Finally The optical density of each plate was measured at 570 nm. All isolates were classified into the following categories: strong, moderate, weak and non-biofilm producer. The S. epidermidis strains ATCC 35,984 (formerly RP62A) was used as positive control.
Detection of ica operon, ACME type and virulence genes
Bacterial DNA was extracted according to the boiling method [44]. The presence of icaA, icaD, icaB, icaC, atlE, aap, bhp, embp, and fbe were determined by the PCR method [13, 25, 32]. The PCR program was run as follows: the initial denaturation at 94 °C for 3 min, 2) 30 cycles of 94 °C for 1 min, 3) annealing temperature (See Table 5) 1 min at 72 °C for extension step, and 4) final extension for 5 min at 72 °C. The products were detected on 1.5% agarose gel, and the band size was compared to a DNA marker (100 bp) Fig. 1. The allotypes of ACME were determined by the PCR [13]. ACME allotypes included ACME-I consisting of the arc and the opp-3 gene clusters, ACME-II containing the arc, and ACME-III constituting the opp-3. The primers used to identify ica operon and virulence genes are presented in Table 5. Staphlococcus epidermidis RP62A (ATCC 35,984) was used as positive control for aap, bhp, embP, and ica-operon genes in the PCR method.
IS256 and IS257 detection
The IS256 (1103-bp) and IS257 (621-bp) were detected by the PCR technique described by Kozitskaya et al. [4] The F-5′-TGAAAAGCGAAGAGATTCAAAGC and R-5′-ATGTAGGTCCATAAGAACGGC primers were used for IS256, and the primer sequences of F-5′-GCTAATTTCGTGGCATGGCG and R-5′- GTTATCACTGTAGCCGTTGG were used for IS257. The PCR program was run based on the following steps: (1) initial denaturation at 94 ºC for 4 min, (2) 30 cycles of denaturation at 92 ºC for 30s, (3) annealing step at 60 ºC for 40s, (4) elongation at 72 ºC for 30s, and (5) final extension at 72 ºC for 5 min. The product was visualized on 1.5% agarose gel, and the band size was compared to a DNA marker (100 bp).
Statistical analysis
We analyzed the data on SPSS version 23 (IBM, Armonk, NY, USA). The chi-square and Fisher’s exact tests were used to compare categorical variables, and P values ≤ 0.05 were considered statistically significant.
Data Availability
The datasets used and/or analyzed during the study are available on reasonable requests from the corresponding author.
Abbreviations
- CoNS:
-
Coagulase negative staphylococci
- MR-CoNS:
-
Methicillin-resistant CoNS
- ACME:
-
Arginine catabolic mobile element
- fbe :
-
Fibrinogen binding protein
- bhp :
-
Bap homologous- protein
- embp :
-
Extracellular matrix-binding protein
- aap :
-
Accumulation-associated protein
- MSCRAMMs:
-
Microbial surface components recognizing adhesive matrix molecules
- PIA:
-
Polysaccharide intercellular adhesion
- PNAG:
-
Poly-N-acetyl-glucosamine
- NICU:
-
Neonatal intensive care units
- ICU:
-
Intensive care unit
- ID:
-
Infectious disease units
- PICU:
-
Pediatric intensive care units
- BMT:
-
Bone marrow transplant
References
Rn ALR, Abreu M, De, Rn A, Rn BE, Rn WC. Risk factors for health care–associated infection in hospitalized adults: Systematic review and meta-analysis. Am J Infect Control. 2017; Available from: https://doi.org/10.1016/j.2017.08.016.
Shahmoradi M, Faridifar P, Shapouri R. Determining the Biofilm Forming Gene Profile of Staphylococcus aureus Clinical isolates via multiplex colony PCR method. RBMB. 2019;7(2):1–8.
Becker K, Heilmann C, Peters G. Coagulase-negative Staphylococci. Am Soc Microbiol. 2014;27(4):870–926.
Kozitskaya S, Cho S, Dietrich K, Marre R, Naber K, Ziebuhr W. The bacterial insertion sequence element IS 256 occurs preferentially in Nosocomial Staphylococcus epidermidis isolates: Association with Biofilm formation and resistance to Aminoglycosides. Infect Immun. 2004;72(2):1210–5.
Suja KRS, Sheela P, Jyothis S, Radhakrishnan EK. Virulence factors associated with Coagulase negative staphylococci isolated from human infections. 3 Biotech. 2017;7(2):1–10.
Heilmann C, Ziebuhr W, Becker K. Are coagulase-negative staphylococci virulent ? *. Clin Microbiol Infect. 2019;25(9):1071–80.
Smith H, Narechania A, Kolokotronis S, Satola SW. Evolution of hypervirulence by a MRSA clone through acquisition of a transposable element. Mol Microbiol. 2015;93(4):664–81.
Unit M, Road SP, Kingdom U, Medical U. The staphylococcal insertion sequence IS 257 is active. Plasmid. 1995;205:198–205.
Ziebuhr W, Krimmer V, Rachid S, Go F, Lo I. A novel mechanism of phase variation of virulence in Staphylococcus epidermidis: evidence for control of the polysaccharide intercellular adhesin synthesis by alternating insertion and excision of the insertion sequence element IS256. Mol Microbiol. 1999;32:345–56.
Conlon KM, Humphreys H, Gara JPO. icaR encodes a transcriptional repressor involved in environmental regulation of ica Operon expression and biofilm formation in Staphylococcus epidermidis. J Bacteriol. 2002;184(16):4400–8.
Kleinert F, Hort M, Zweynert A, Szekat C, Nagel M, Bierbaum G. Crossm influence of IS 256 on genome variability and formation of small-colony variants. Antimicrob Agents Chemother. 2017;61(8):1–16.
Cue D, Lei MG, Lee CY. Genetic regulation of the intercellular adhesion locus in staphylococci. Front Cell Infect Microbiol. 2012;2(March):1–13.
Salgueiro VC, Lopes N, Iorio P, Ferreira MC, Chamon RC. Methicillin resistance and virulence genes in invasive and nasal Staphylococcus epidermidis isolates from neonates. BMC Microbiol. 2017;1–11. Available from: https://doi.org/10.1186/s12866-017-0930-9.
França A, Gaio V, Lopes N, Melo DR. Virulence factors in coagulase-negative Staphylococci. Pathogens. 2021;10:170.
Nasr RA, Abushady HM, Hussein HS. Biofilm formation and presence of icaAD gene in clinical isolates of staphylococci. Egypt J Med Hum Genet. 2012;13(3):269–74. Available from: https://doi.org/10.1016/j.2012.
Bahonar S, Ghazvinian M, Haghshenas MR, Goli HR, Mirzaei B. Purification of PIA and rSesC as putative vaccine candidates against Staphylococcus aureus.RBMB. 2019;8(2).
Mirzaei B. Staphylococcal Vaccine Antigens related to biofilm formation. Hum Vaccin Immunother. 2021;17(1):293–303. Available from: https://doi.org/10.1080/21645515.2020.
Mirzaei B, Moosavi SF, Babaei R. Purification and evaluation of Polysaccharide Intercellular Adhesion (PIA) Antigen from Staphylococcus epidermidis. Curr Microbiol. 2016;73(5):611–7.
Diep BA, Gill SR, Chang RF, Phan TH, Chen JH, Davidson MG, et al. Complete genome sequence of USA300, an epidemic clone of community-acquired meticillin-resistant Staphylococcus aureus. Lancet. 2006;367:731–9.
Miragaia M, Lencastre H, De, Perdreau-remington F, Chambers HF, Sullam PM, Lin J, et al. Genetic diversity of Arginine Catabolic Mobile element in Staphylococcus epidermidis. PLoS ONE. 2009;4(11):e7722.
Onishi M, Urushibara N, Kawaguchiya M, Ghosh S, Shinagawa M, Infection. Genetics and Evolution Prevalence and genetic diversity of arginine catabolic mobile element (ACME) in clinical isolates of coagulase-negative staphylococci: Identification of ACME type I variants in Staphylococcus epidermidis. Infect Genet Evol. 2013;20:381–8. Available from: https://doi.org/10.1016/j.2013.09.018.
Miragaia M, Carric JA, Thomas JC, Couto I, Enright MC, Lencastre H, De. Comparison of molecular ty** methods for characterization of Staphylococcus epidermidis: proposal for clone definition. J Clin Microbiol. 2008;46(1):118–29.
Dalton KR, Rock C, Carroll KC, Davis MF. One health in hospitals: how understanding the dynamics of people, animals, and the hospital built-environment can be used to better inform interventions for antimicrobial-resistant gram-positive infections. Antimicrob Resist Infect Control. 2020;2:1–17.
Cafiso V, Bertuccio T, Santagati M, Campanile F, Amicosante G, Perilli MG et al. Presence of the ica operon in clinical isolates of Staphylococcus epidermidis and its role in biofilm production. Clin Microbiol Infect. 2004;10(12):1081–8. Available from: https://doi.org/10.1111/j.1469-0691.2004.01024.x.
Zhou S, Chao X, Fei M, Dai Y, Liu B. Analysis of S. Epidermidis icaA and icaD genes by polymerase chain reaction and slime production: a case control study. BMC Infect Dis. 2013;13(1):1.
Letizia S, Nentwich MM, Mino H. Methicillin resistance and biofilm production of Staphylococcus epidermidis isolates from infectious and normal flora conjunctiva. Int Ophthalmol. 2017;819–25.
Begun J, Gaiani JM, Rohde H, Mack D, Calderwood SB, Ausubel FM et al. Staphylococcal Biofilm Exopolysaccharide protects against Caenorhabditis elegans Immune defenses. PLoS ONE. 2007;3(4).
Gara JPO. ica and beyond: bio ¢ lm mechanisms and regulation in Staphylococcus epidermidis and Staphylococcus aureus. FEMS. 2007;270:179–88.
Neopane P, Shrestha R, Uehara O, Abiko Y. In vitro biofilm formation by Staphylococcus aureus isolated from wounds of hospital-admitted patients and their association with antimicrobial resistance. Int J Gen Med. 2018;25–32.
Zalipour M, Ebrahim-saraie HS, Sarvari J, Khashei R. Detection of Biofilm Production Capability and icaA / D Genes Among Staphylococci Isolates from Shiraz, Iran. Jundishapur J Microbiol. 2016;9(12):9 (12).
Peerayeh SN, Moghadas AJ, Behmanesh M. Prevalence of virulence-related determinants in clinical Isolates of Staphylococcus epidermidis. Jundishapur J Microbiol. 2016;9(8).
Rohde H, Kalitzky M, Kro N, Scherpe S, Horstkotte MA, Knobloch JK, et al. Detection of Virulence-Associated genes not useful for discriminating between Invasive and Commensal Staphylococcus epidermidis strains from a bone marrow transplant unit. J Clin Microbiol. 2004;42(12):5614–9.
Pourmand MR, Abdossamadi Z, Salari MH, Hosseini M. Original article slime layer formation and the prevalence of mecA and aap genes in Staphylococcus epidermidis isolates. J Infect Dev Ctries. 2011;(15):034–40.
Petrelli D, Zampaloni C, Ercole SD, Prenna M. Analysis of different genetic traits and their association with biofilm formation in Staphylococcus epidermidis isolates from central venous catheter infections. Eur J Clin Microbiol Infect Dis. 2006;773–81.
Conlon KM, Humphreys H, Gara JPO. Inactivations of rsbU and sarA by IS 256 represent Novel Mechanisms of Biofilm phenotypic variation in Staphylococcus epidermidis. J Bacteriol. 2004;186(18):6208–19.
Clin A, Antimicrob M, Manandhar S, Singh A, Varma A, Pandey S et al. Phenotypic and genotypic characterization of biofilm producing clinical coagulase negative staphylococci from Nepal and their antibiotic susceptibility pattern. Ann Clin Microbiol Antimicrob. 2021;1–11. Available from: https://doi.org/10.1186/s12941-021-00447-6.
Arciola CR, Campoccia D, Ravaioli S, Montanaro L. Polysaccharide intercellular adhesin in biofilm: structural and regulatory aspects. Front Cell Infect Microbiol. 2015;5(February):1–10.
Foulston L, Elsholz AKW, Defrancesco AS, Losick R. The Extracellular Matrix of Staphylococcus aureus Biofilms comprises cytoplasmic Proteins that associate with the cell surface in response to decreasing pH. MBio. 2014;5(5):1–9.
Tong SYCC, Davis JS, Eichenberger E, Holland TL, Fowler VG. Staphylococcus aureus Infections: Epidemiology, Pathophysiology, Clinical Manifestations, and management. Clin Microbiol Rev. 2015;28(3):603–61.
Hirotaki S, Sasaki T, Kuwahara-arai K, Hiramatsu K. Rapid and Accurate Identification of Human-Associated Staphylococci by Use of Multiplex PCR. J Clin Microbiol. 2011;49(10):3627–31.
M100. Performance Standards for Antimicrobial Susceptibility Testing A CLSI supplement for global application. 31st Edition.
Ruzauskas M, Siugzdiniene R, Klimiene I, Virgailis M, Mockeliunas R, Vaskeviciute L. Prevalence of methicillin-resistant Staphylococcus haemolyticus in companion animals: a cross-sectional study. Antibiotics. 2014;1–7.
Vukovic D, Bonaventura GDI, Djukic S, Ruzicka F. Quantification of biofilm in microtiter plates: overview of testing conditions and practical recommendations for assessment of biofilm production by staphylococci. APMIS. 2007;891–9.
Alexopoulou K, Foka A, Petinaki E, Jelastopulu E, Dimitracopoulos G, Spiliopoulou I. Comparison of two commercial methods with PCR restriction fragment length polymorphism of the tuf gene in the identification of coagulase-negative staphylococci. Lett Appl Microbiol. 2006;43:450–4.
Acknowledgements
The authors would like to thank the staff at the Afzalipour teaching hospital in Kerman, Iran.
Funding
This work was supported by the Kerman University of Medical Sciences (Grant number 98000886).
Author information
Authors and Affiliations
Contributions
Davood Kalantar-Neyestanaki, Shahla Mansouri and Elham Isaei designed the experiments; Elham Isaei and Davood Kalantar-Neyestanaki performed experiments and collected data; Davood Kalantar-Neyestanaki, Shahla Mansouri, Omid Tajrobehkar and Elham Isaei analysed data, discussed the results and strategy; Davood Kalantar-Neyestanaki and Shahla Mansouri Supervised, directed and managed the study. The author(s) read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All methods were carried out following respective guidelines and regulations. Informed consent was obtained from the participants. The present study was approved by the Ethics Committee of the Research Council of Kerman University of Medical Sciences, Kerman, Iran (IR.KMU.REC.1398.650).
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Kalantar-Neyestanaki, D., Mansouri, S., Tadjrobehkar, O. et al. The frequency of adherence, biofilm-associated, Arginine Catabolic Mobile element genes, and biofilm formation in clinical and healthcare worker coagulase-negative staphylococci isolates. BMC Microbiol 23, 222 (2023). https://doi.org/10.1186/s12866-023-02959-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12866-023-02959-x