Abstract
Background
Endometrial cancer (EC) is one of the most common gynecological cancers. Herein, we aimed to define the role of specific myosin family members in EC because this protein family is involved in the progression of various cancers.
Methods
Bioinformatics analyses were performed to reveal EC patients’ prognosis-associated genes in patients with EC. Furthermore, colony formation, immunofluorescence, cell counting kit 8, wound healing, and transwell assays as well as coimmunoprecipitation, cycloheximide chase, luciferase reporter, and cellular thermal shift assays were performed to functionally and mechanistically analyze human EC samples, cell lines, and a mouse model, respectively.
Results
Machine learning techniques identified MYH14, a member of the myosin family, as the prognosis-associated gene in patients with EC. Furthermore, bioinformatics analyses based on public databases showed that MYH14 was associated with EC chemoresistance. Moreover, immunohistochemistry validated MYH14 upregulation in EC cases compared with that in normal controls and confirmed that MYH14 was an independent and unfavorable prognostic indicator of EC. MYH14 impaired cell sensitivity to carboplatin, paclitaxel, and progesterone, and increased cell proliferation and metastasis in EC. The mechanistic study showed that MYH14 interacted with MYH9 and impaired GSK3β-mediated β-catenin ubiquitination and degradation, thus facilitating the Wnt/β-catenin signaling pathway and epithelial–mesenchymal transition. Sesamolin, a natural compound extracted from Sesamum indicum (L.), directly targeted MYH14 and attenuated EC progression. Additionally, the compound disrupted the interplay between MYH14 and MYH9 and repressed MYH9-regulated Wnt/β-catenin signaling. The in vivo study further verified sesamolin as a therapeutic drug without side effects.
Conclusions
Herein, we identified that EC prognosis-associated MYH14 was independently responsible for poor overall survival time of patients, and it augmented EC progression by activating Wnt/β-catenin signaling. Targeting MYH14 by sesamolin, a cytotoxicity-based approach, can be applied synergistically with chemotherapy and endocrine therapy to eventually mitigate EC development. This study emphasizes MYH14 as a potential target and sesamolin as a valuable natural drug for EC therapy.
Graphical Abstract

Similar content being viewed by others
Background
Endometrial cancer (EC) is one of the most common gynecological cancers with increasing incidence [1, 2]. Despite the implementation of several available therapeutic strategies, the survival outcomes of patients with EC are unsatisfactory [3]. The combination of paclitaxel and carboplatin is the standard first-line chemotherapy for EC [4]. However, chemoresistance to this combination is one of the main factors contributing to poor EC prognosis [1M), indicating MYH14 as a prognosis-associated gene in patients with EC.
Bioinformatics analyses of the public database identify the prominent myosin family members correlated with EC prognosis. A Survival analysis displaying the relationship between myosin activities calculated by ssGSEA and the overall survival of EC patients in the TCGA UCEC dataset. Kaplan–Meier curves were plotted using the log-rank test. B, C Volcano plots presenting differentially expressed genes between EC and normal controls in TCGA UCEC and GSE17025 datasets, respectively. D, E The random forest model was applied to rank the identified 15 myosins that correlated with EC patient outcomes. The relationship between number of decision trees and model error; x axis, number of decision trees; and y axis, error rate of the obtained model are shown. Importance of all variables in the random forest classifier as determined by the Gini coefficient method; x axis, mean decrease in Gini index; and y axis, variables are shown. F The lasso regression model was used to identify prognosis-associated myosins in EC patients. Tenfold cross-validation was used to tune the parameters in the lasso model. G, H Scale independence and mean connectivity were plotted to reveal the soft threshold and scale-free topology model fit index according to the TCGA UCEC dataset. I, J A GeneTree and a cluster dendrogram were established based on the soft threshold according to the TCGA UCEC dataset. Different colors represent different co-expression modules. K The heatmap showing the relationship between gene modules and EC according to the TCGA UCEC dataset. Each row represents a module, and each column represents a clinical status. L The scatter plots presenting the association between genes and EC in the MEblue module. M A Venn diagram showing the overlap** gene correlated with the prognosis of EC patients. EC Endometrial cancer, GEO Gene Expression Omnibus, ssGSEA simple sample gene set enrichment analysis, TCGA The Cancer Genome Atlas, UCEC Uterine corpus endometrioid carcinoma
The role of MYH14 in EC was further investigated by performing bioinformatics analyses. In both TCGA UCEC and GSE17025 datasets, the differential analyses indicated MYH14 upregulation in EC compared with that in normal controls (Fig. 2A, B). Furthermore, MYH14 expression was upregulated in high-grade EC samples compared with that in low-grade EC samples (Fig. 2C, D). The survival analyses based on the best cut-off value revealed a positive correlation between poor overall survival of patients with EC and upregulated MYH14 expression (Fig. 2E). Additionally, the subgroup analyses according to the best cut-off value showed that upregulated MYH14 expression conferred poor prognosis of patients with EC with young age, stage 3, and grade 3 (Fig. 2F–H). Then, univariate and multivariate Cox hazard analyses were performed to verify the relationship between MYH14 expression and patient prognosis in EC. The univariate Cox analysis indicated that upregulated MYH14 expression, elder age, advanced EC stage, and high grade conferred the patients with poor overall survival (Fig. 2I). The multivariate Cox analysis indicated that MYH14 expression independently served as a predictor of the overall survival time of patients with EC (hazard ratio: 1.610, 95% confidence interval: 1.014–2.554, P = 0.043) (Fig. 2J).
MYH14 expression was elevated in EC and conferred poor overall survival to patients according to the public database. A, B MYH14 expression was differentially expressed in EC and normal controls according to the TCGA UCEC and GSE17025 datasets. C, D MYH14 expression was differentially expressed in grades 1 and 2, and three EC tissues according to the TCGA UCEC and GSE17025 datasets. E Survival analysis displaying the association between MYH14 levels and the overall survival of EC patients. F–H Subgroup analyses exhibiting the overall survival time of EC patients in young (age ≤ 63 years) (F), stage 3 (G), and grade 3 (H). Kaplan–Meier curves were plotted using the log-rank test. I, J Univariate and multivariate Cox analyses disclosing the association between prognosis, clinicopathologic features, and MYH14 levels in EC patients. EC endometrial cancer, GEO Gene Expression Omnibus, TCGA The Cancer Genome Atlas
Collectively, these findings indicated that MYH14 functioned as a possible diagnostic and prognostic predictor of EC.
MYH14 levels were potentially associated with EC chemoresistance
Based on the association between MYH14 expression and the prognosis of patients with EC, ssGSEA was performed to determine the activities of biological processes and signaling pathways and correlate them with MYH14 expression. The correlation analyses revealed the positive association between TGF-β signaling, Wnt/β-catenin signaling, EMT, PI3K/AKT/mTOR signaling, Notch signaling, and MYH14 expression based on the HALLMARK geneset. Notably, the correlation analyses also revealed the positive association between TGF-β signaling, Wnt signaling, phosphatidylinositol signaling system, mTOR signaling, Notch signaling, and MYH14 expression based on the KEGG dataset (Additional file 1: Fig. S2A, B).
We further investigated the relationship between MYH14 expression and chemoresistance in patients with EC. Using ssGSEA, we calculated the platinum drug resistance index for patients with EC based on the TCGA database as we previously reported [24]. The analysis revealed a positive association between MYH14 expression and the platinum drug resistance index in the TCGA database on patients with EC (Additional file 1: Fig. S3A). Moreover, we used the oncoPredict tool in R and found a positive correlation between MYH14 expression and paclitaxel drug resistance in the GSE17025 dataset (Additional file 1: Fig. S3B). Furthermore, the GSCA database revealed that MYH14 levels were associated with cisplatin (correlation = 0.189, P < 0.001) and paclitaxel (correlation = 0.169, P < 0.001) drug resistance.
Altogether, these results suggested that MYH14 might be a chemoresistance-related oncogene in EC.
MYH14 expression was associated with EC pathology
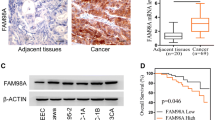
To further determine the clinical significance of MYH14 in EC, we performed an IHC analysis using 118 EC samples and 17 adjacent normal controls. The results revealed increased MYH14 expression in EC than in adjacent normal controls (Fig. 3A). Additionally, MYH14 expression varied among EC tissues (Fig. 3B). Further analysis indicated a positive correlation between MYH14 protein levels and Ki67 expression (Additional file 1: Table S4). Survival analysis showed a positive association between MYH14 expression and poor overall survival in patients with EC (Fig. 3C). Univariate and multivariate Cox hazard analyses confirmed MYH14 expression as an unfavorable and independent prognostic indicator for overall survival in patients with EC (hazard ratio: 1.477, 95% confidence interval: 1.002–2.178, P = 0.049) (Fig. 3D, E). Stratified analyses based on stage, grade, or age showed that MYH14 expression remained positively associated with poor overall survival in patients with EC (Fig. 3F–H).
MYH14 levels detected by IHC were elevated in EC and associated with poor patient prognosis. A Representative images presenting the differential MYH14 expression between EC and normal controls. B Representative images presenting differential MYH14 expression in EC. C Survival analysis revealing the association between MYH14 levels and the overall survival of EC patients. D, E Univariate and multivariate Cox analyses disclosing the association between MYH14 expression, clinicopathologic features and prognosis of EC patients. F–H Subgroup analyses were performed to elucidate the overall survival time of EC patients in stage 3 (F), grade 3 (G), and elderly (age > 55 years) (H). Kaplan–Meier curves were plotted using the log-rank test. EC endometrial cancer, H&E hematoxylin and eosin, IHC immunohistochemistry
To summarize, these findings revealed the crucial role of MYH14 in the progression of EC and suggested its potential clinical value as a biomarker.
MYH14 knockdown ameliorated therapy sensitivity and inhibited cell proliferation and metastasis in EC
The biological functions of MYH14 in the oncogenic process and signaling pathways were explored on the basis of the results obtained from bioinformatics analyses. MYH14 expression was silenced using siRNA in the Ishikawa and KLE cells (Fig. 4A). Subsequently, we evaluated the effect of carboplatin and paclitaxel on EC cells to determine the appropriate concentration for subsequent experiments (Additional file 1: Fig. S4A–D). Colony formation assays revealed that MYH14 knockdown sensitized EC cells to carboplatin and paclitaxel (Fig. 4B, C). Immunofluorescence analysis of the DNA damage marker (γH2AX) reported that MYH14 depletion increased the number of γ-H2AX-positive cells in carboplatin and paclitaxel-treated Ishikawa and KLE cells (Fig. 4D, E), further confirming the role of MYH14 in regulating EC chemosensitivity. Intriguingly, silencing MYH14 also sensitized EC cells to progesterone (Fig. 4F). Furthermore, MYH14 depletion was associated with a reduction in EC cell proliferation, as evidenced by CCK-8 assays (Fig. 4G). Wound healing and transwell assays further revealed the inhibitory effect of MYH14 silencing on the migration and invasion of EC cells (Fig. 4H, I).
MYH14 knockdown improved therapeutic sensitivity and suppressed cell proliferation and metastasis in EC. Cells were treated with CBP, PTX, or MPA for 48 h and subjected to further experiments. A qPCR and western blot assays were applied to assess MYH14 levels in MYH14-silenced Ishikawa and KLE cells and the controls. B–E Colony formation (B, C) and immunofluorescence (D, E) assays were adopted to measure the effect of MYH14 on the chemosensitivity of Ishikawa and KLE cells. F CCK-8 assays were employed to measure the sensitivity to endocrine therapy of MYH14-silenced Ishikawa and KLE cells and the controls. Ishikawa and KLE cells were treated with 40 μM and 50 μM MPA, respectively. G CCK-8 assays were used to determine the proliferation of MYH14-silenced Ishikawa and KLE cells and the controls. H, I Wound healing (H) and transwell (I) assays were applied for evaluating the metastasis of MYH14-silenced Ishikawa and KLE cells and the controls. * P < 0.05, ** P < 0.01, and *** P < 0.001 versus the control group. ### P < 0.001 versus the CBP or PTX group. EC endometrial cancer, CBP carboplatin, MPA medroxyprogesterone acetate, PTX paclitaxel, qPCR quantitative RT–PCR
These results indicated the inhibitory role of MYH14 in EC chemosensitivity and endocrine therapy sensitivity and suggested the stimulative role of MYH14 in the proliferation and metastasis of EC.
MYH14 interacted with MYH9 to facilitate Wnt/β-catenin signaling
To elucidate the potential mechanism of MYH14 in promoting EC progression, we investigated the interacting protein of MYH14. Co-IP along with mass spectrometry identified MYH9 as a potential interacting protein of MYH14 (Fig. 5A). Our previous studies have shown that MYH9 attenuated the GSK3β-mediated ubiquitination and degradation of β-catenin, thereby facilitating Wnt signaling activity and promoting proliferation, metastasis, and chemoresistance of cancer cells [9]. Additionally, the aforementioned bioinformatics analyses indicated the involvement of MYH14 in Wnt signaling. Therefore, we performed further investigations to determine the relationship between MYH14 and MYH9. Co-IP confirmed the binding between MYH14 and MYH9 in EC cells using Co-IP (Fig. 5B). The subsequent experiments aimed to determine how MYH14 affected the MYH9-mediated ubiquitination and degradation of β-catenin. MYH14 knockdown resulted in the upregulation of GSK3β expression and the downregulation of β-catenin expression, which could be reversed by MYH9 overexpression (Fig. 5C). Additionally, CHX chase assay showed that MYH14 depletion shortened the half-life of β-catenin protein (Fig. 5D). Furthermore, silencing MYH14 facilitated the formation of the GSK3β/β-catenin/ubiquitin complex, which could be reversed by MYH9 overexpression (Fig. 5E). Moreover, MYH14 downregulation restrained the activation of Wnt signaling, and this impact could be recovered by MYH9 overexpression (Fig. 5F). These results collectively indicated that MYH14 attenuated the GSK3β-mediated ubiquitination and degradation of β-catenin through its interaction with MYH9, thereby activating Wnt signaling in EC.
MYH14 interacted with MYH9 to facilitate Wnt/β-catenin signaling. A Co-IP combined with silver staining identifying the interacting protein of MYH14. B Co-IP assays detecting the interplay between MYH14 and MYH9 in Ishikawa and KLE cells. C Western blot assays showing the effect of MYH14 and MYH9 on GSK3β and β-catenin expression. D CHX chase assay showing the effect of MYH14 on the half-life of β-catenin protein. E Co-IP assays were conducted for measuring the effect of MYH14 and MYH9 on the formation of GSK3β/β-catenin/ubiquitin complex in Ishikawa cells treated with MG132. F TOP/FOP luciferase reporter assays were applied for detecting the effect of MYH14 and MYH9 on Wnt signaling activity. *** P < 0.001 versus the control group. CHX cycloheximide, Co-IP coimmunoprecipitation
Sesamolin directly targeted MYH14 to attenuate EC progression
After establishing the oncogenic role of MYH14 in EC, we searched for a potential drug targeting MYH14. Previous research has suggested a direct interaction between sesamol and MYH14 [31]. Because sesamolin was shown to have superior anti-cancer properties than its metabolite sesamol [32], we investigated whether sesamolin or its metabolite sesamol could target MYH14. Molecular docking showed a higher probability of interaction between sesamolin and MYH14 compared with sesamol, as determined by CDOCKER interaction energy (Fig. 6A, B). Subsequently, we determined the potential interaction between sesamolin and MYH14. CETSA showed that sesamolin increased the protein stability of MYH14, confirming MYH14 as a direct target of sesamolin (Fig. 6C). An in vivo study confirmed the suppressive role of sesamolin on EC progression, as indicated by the evaluation of the size and weight of xenografts (Fig. 6D, E). Notably, there was no significant difference in the body weight of mice between the sesamolin-treated group and the control group, suggesting no distinct toxicity of sesamolin in vivo (Fig. 6F). Then, the biological function of sesamolin in EC progression was further determined. Colony formation assays showed that sesamolin sensitized EC cells toward carboplatin and paclitaxel in a dose-dependent manner (Fig. 7A, B). Immunofluorescence analysis indicated that sesamolin increased the percentage of γ-H2AX-positive cells in carboplatin and paclitaxel-treated EC cells (Fig. 7C, D). Additionally, sesamolin restored sensitivity to progesterone treatment in a dose-dependent manner, and it reduced the proliferation, migration, and invasion of EC cells (Fig. 7E–H). These findings suggested that sesamolin acted as an MYH14 inhibitor, inhibiting EC progression by directly targeting MYH14.
Sesamolin directly targeted MYH14 to attenuate EC progression. A, B Molecular docking revealing the potential interaction between sesamolin and MYH14. Three- and two-dimensional pattern diagrams showing the binding between sesamolin and MYH14. C CETSA was performed to assess the effect of sesamolin on the protein stability of MYH14. Cells were treated with Ses for 48 h. D, E An in vivo study was conducted to show the effect of sesamolin on the size and weight of xenografts. F For in vivo toxicity assessment, the body weight of mice was measured before and after sesamolin administration. * P < 0.05, and ** P < 0.01 versus the control group. EC endometrial cancer, CBP carboplatin, CETSA cellular thermal shift assay, PTX paclitaxel, Ses sesamolin
Sesamolin improved therapeutic sensitivity and attenuated cell proliferation and metastasis in EC. Cells were treated with CBP, PTX, MPA, and/or Ses for 48 h and subjected to further experiments. A–D Colony formation (A, B) and immunofluorescence (C, D) assays were adopted for evaluating the effect of sesamolin on the chemosensitivity of Ishikawa and KLE cells. E CCK-8 assays were adopted to measure the endocrine therapy sensitivity of sesamolin-treated Ishikawa and KLE cells and the controls. Ishikawa and KLE cells were treated with 40 μM and 50 μM MPA, respectively. F CCK-8 assays were conducted to reveal the proliferation of sesamolin-treated Ishikawa and KLE cells and the controls. G, H Wound healing (G) and transwell (H) assays were applied to elucidate the metastasis of sesamolin-treated Ishikawa and KLE cells and the controls. * P < 0.05, ** P < 0.01, and *** P < 0.001 versus the control group. ### P < 0.001 versus the CBP or PTX group. EC endometrial cancer, CBP carboplatin, MPA medroxyprogesterone acetate, PTX paclitaxel, Ses sesamolin
Sesamolin disrupted the interplay between MYH14 and MYH9 to inactivate Wnt/β-catenin signaling
Because sesamolin could mimic the inhibitory effect of MYH14 knockdown in EC progression, the effect of sesamolin on MYH14-mediated downstream signaling was subsequently determined to elucidate the mechanism of sesamolin in controlling EC progression. Co-IP assays reported that sesamolin could interfere with the interaction between MYH14 and MYH9 (Fig. 8A). Furthermore, sesamolin treatment upregulated GSK3β expression and downregulated β-catenin expression, which could be rescued by MYH14 overexpression (Fig. 8B). CHX chase assay showed that the half-life of β-catenin protein decreased due to sesamolin stimulation (Fig. 8C). Furthermore, co-IP showed that sesamolin treatment increased interaction between GSK3β, ubiquitin, and β-catenin, suggesting the formation of GSK3β/β-catenin/ubiquitin complex. TOP/FOP luciferase reporter indicated that the inactivation of Wnt signaling could be facilitated by sesamolin treatment. Nevertheless, the effect of sesamolin on MYH14-mediated downstream signaling could be reversed by MYH14 overexpression (Fig. 8D, E).
Sesamolin disrupted the interplay between MYH14 and MYH9 to inactivate Wnt/β-catenin signaling. Cells were treated with sesamolin for 48 h. A Co-IP assays were performed for identifying the effect of sesamolin on the interaction between MYH14 and MYH9. B Western blot assays were conducted for showing the effect of sesamolin and MYH14 on GSK3β and β-catenin expression. C CHX chase assay was adopted to present the effect of sesamolin on the half-life of β-catenin protein. D Co-IP assays were used for revealing the effect of sesamolin and MYH14 on the formation of GSK3β/β-catenin/ubiquitin complex in Ishikawa cells treated with MG132. E TOP/FOP luciferase reporter assays were adopted to assess the effect of sesamolin and MYH14 on Wnt signaling activity. F Working model elucidating the effect of sesamolin-targeted MYH14 and MYH9-mediated Wnt/β-catenin signaling on chemosensitivity, endocrine therapy sensitivity, proliferation, and metastasis. This graph was generated using the Biorender website (https://biorender.com/). *** P < 0.001 versus the control group. CHX cycloheximide, Co-IP co-immunoprecipitation, Ses sesamolin
Altogether, these results suggested that sesamolin increased the chemosensitivity and endocrine therapy sensitivity of EC and repressed its proliferation and metastasis via the inhibition of MYH14-mediated downstream signaling.
Discussion
EC chemoresistance contributes to poor patient prognosis [33]. Progesterone resistance limited the efficiency of conservative therapy in patients with EC, especially in those suffering from advanced and recurrent EC [34]. The enhancement of EMT is regarded as a crucial mechanism of EC chemoresistance and progesterone resistance [35, 36]. Targeting myosin II, a member of the myosin family, could reduce chemoresistance in gynecological cancer [37]. In this study, we used machine learning techniques and identified a novel prognosis-associated myosin family member in patients with EC, namely MYH14. MYH14 decreased the overall survival of patients with EC. Furthermore, MYH14 impaired the chemosensitivity and endocrine therapy sensitivity of EC, increased the proliferation and metastasis of EC, and upregulated the expression of β-catenin (an EMT marker) [38] by triggering MYH9-mediated Wnt signaling. Interestingly, sesamolin, a natural compound, could directly target MYH14 to attenuate Wnt/β-catenin signaling, thus repressing EC progression.
Previous studies have shown that the ssGSEA algorithm can be used to establish novel indexes based on specific genesets. Zheng et al. showed the ability of the ssGSEA-based stemness index to aid the appointment of tumor grade and its possible therapeutic and diagnostic applications. The relative expression ordering (REO) based stemness indexes were REO-based signatures with high robustness against the batch effects and can be stably used in independent datasets [39]. Yi et al. reported that computationally derived Ras-dependency indexes (RDI) can represent a measure of Ras dependency in both patient samples and cancer cell lines. Furthermore, the ssGSEA score correlated with the original RDI datasets [40]. Therefore, we calculated the myosin activity on the basis of the geneset containing 52 myosin family members and counted the chemoresistance index as per the platinum drug resistance geneset extracted from KEGG. The negative correlation between calculated myosin activity and the prognosis of patients with EC suggested the participation of myosins in EC progression. Using machine learning techniques along with WGCNA, we identified MYH14 as the most significant myosin correlating with the prognosis of patients with EC. Furthermore, bioinformatics analyses of public data and IHC analysis of our patients with EC confirmed that MYH14 expression was upregulated in EC tissues and independently predicted the poor overall survival of patients with EC.
TGF-β signaling, Wnt/β-catenin signaling, and PI3K/AKT signaling are well-established promoters of EMT [41], with EMT being a determinant of cancer cell chemoresistance [42]. In the present study, the positive association between TGF-β signaling, Wnt/β-catenin signaling, PI3K/AKT signaling, EMT, and MYH14 expression determined by bioinformatics analyses prompted us to investigate the role of MYH14 in EC chemoresistance. The oncoPredict package in the R and GSCA database has been successfully used to estimate cancer chemoresistance [43, 51,60]. The existing evidence indicates sesamolin as a novel and promising therapeutic drug for the treatment of EC.
Conclusions
In this study, we showed that MYH14 was a novel oncogene associated with EC prognosis and therapy sensitivity, independently contributing to poor overall survival of patients with EC. MYH14 interacted with MYH9, inhibiting the GSK-3β-mediated ubiquitination and degradation of β-catenin. This interaction activated the Wnt/β-catenin signaling pathway, thereby promoting EC progression. Furthermore, MYH14 could be directly targeted by sesamolin which disrupted MYH14-mediated downstream signals, thus improving the chemosensitivity and endocrine therapy sensitivity of EC while also suppressing the proliferation and metastasis of EC (Fig. 8F). Effective chemotherapy sensitization can inhibit the escape of tumor cells and mitigate serious side effects. Co-administration of sesamolin or a MYH14 inhibitor and carboplatin or paclitaxel presents a promising synergistic strategy for the treatment of advanced and recurrent EC. To conclude, our study reports MYH14 as a potential therapeutic target and sesamolin as a valuable natural compound for EC therapy.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- CBP:
-
Carboplatin
- CCK-8:
-
Cell counting kit 8
- CETSA:
-
Cellular thermal shift assay
- CHX:
-
Cycloheximide
- Co-IP:
-
Co-immunoprecipitation
- EC:
-
Endometrial cancer
- EMT:
-
Epithelial-mesenchymal transition
- FBS:
-
Fetal bovine serum
- GEO:
-
Gene Expression Omnibus
- GSCA:
-
Gene Set Cancer Analysis
- IHC:
-
Immunohistochemistry
- KEGG:
-
Kyoto Encyclopedia of Genes and Genomes
- MPA:
-
Medroxyprogesterone acetate
- PPI:
-
Protein–protein interaction
- PTX:
-
Paclitaxel
- qRT-PCR:
-
Quantitative reverse transcription polymerase chain reaction
- Ses:
-
Sesamolin
- ssGSEA:
-
Simple sample gene set enrichment analysis
- TCGA:
-
The Cancer Genome Atlas
- UCEC:
-
Uterine corpus endometrioid carcinoma
- WGCNA:
-
Weighted gene co-expression network analysis
References
Crosbie EJ, Kitson SJ, McAlpine JN, Mukhopadhyay A, Powell ME, Singh N. Endometrial cancer. Lancet. 2022;399(10333):1412–28.
Zhou J, Lin Y, Yang X, Shen B, Hao J, Wang J, et al. Metabolic disorders sensitise endometrial carcinoma through endoplasmic reticulum stress. Cell Mol Biol Lett. 2022;27(1):110.
Matsuzaki S, Klar M, Matsuzaki S, Roman LD, Sood AK, Matsuo K. Uterine carcinosarcoma: contemporary clinical summary, molecular updates, and future research opportunity. Gynecol Oncol. 2021;160(2):586–601.
Eskander RN, Sill MW, Beffa L, Moore RG, Hope JM, Musa FB, et al. Pembrolizumab plus chemotherapy in advanced endometrial cancer. N Engl J Med. 2023;388(23):2159–70.
**ao Y, ** L, Deng C, Guan Y, Kalogera E, Ray U, et al. Inhibition of PFKFB3 induces cell death and synergistically enhances chemosensitivity in endometrial cancer. Oncogene. 2021;40(8):1409–24.
Mourksi NE, Dalban C, Colombe-Vermorel A, Odeyer L, Simioni V, Frenel JS, et al. Ribosome biogenesis-based predictive biomarkers in endocrine therapy (Anastrozole) combined with mTOR inhibitor (Vistusertib) in endometrial cancer: translational study from the VICTORIA trial in collaboration with the GINECO group. Mol Oncol. 2023;17(1):27–36.
van den Heerik A, Horeweg N, de Boer SM, Bosse T, Creutzberg CL. Adjuvant therapy for endometrial cancer in the era of molecular classification: radiotherapy, chemoradiation and novel targets for therapy. Int J Gynecol Cancer. 2021;31(4):594–604.
Trivedi DV, Nag S, Spudich A, Ruppel KM, Spudich JA. The myosin family of mechanoenzymes: from mechanisms to therapeutic approaches. Annu Rev Biochem. 2020;89:667–93.
Lin X, Li AM, Li YH, Luo RC, Zou YJ, Liu YY, et al. Silencing MYH9 blocks HBx-induced GSK3beta ubiquitination and degradation to inhibit tumor stemness in hepatocellular carcinoma. Signal Transduct Target Ther. 2020;5(1):13.
Li Y, Liu X, Lin X, Zhao M, **ao Y, Liu C, et al. Chemical compound cinobufotalin potently induces FOXO1-stimulated cisplatin sensitivity by antagonizing its binding partner MYH9. Signal Transduct Target Ther. 2019;4:48.
Liu Y, Jiang Q, Liu X, Lin X, Tang Z, Liu C, et al. Cinobufotalin powerfully reversed EBV-miR-BART22-induced cisplatin resistance via stimulating MAP2K4 to antagonize non-muscle myosin heavy chain IIA/glycogen synthase 3beta/beta-catenin signaling pathway. EBioMedicine. 2019;48:386–404.
Engevik KA, Engevik MA, Engevik AC. Bioinformatics reveal elevated levels of Myosin Vb in uterine corpus endometrial carcinoma patients which correlates to increased cell metabolism and poor prognosis. PLoS ONE. 2023;18(1): e0280428.
Tochimoto M, Oguri Y, Hashimura M, Konno R, Matsumoto T, Yokoi A, et al. S100A4/non-muscle myosin II signaling regulates epithelial-mesenchymal transition and stemness in uterine carcinosarcoma. Lab Invest. 2020;100(5):682–95.
Greener JG, Kandathil SM, Moffat L, Jones DT. A guide to machine learning for biologists. Nat Rev Mol Cell Biol. 2022;23(1):40–55.
Li YK, Zeng T, Guan Y, Liu J, Liao NC, Wang MJ, et al. Validation of ESM1 related to ovarian cancer and the biological function and prognostic significance. Int J Biol Sci. 2023;19(1):258–80.
Mo J, Ruan S, Yang B, ** Y, Liu K, Luo X, et al. A novel defined risk signature of endoplasmic reticulum stress-related genes for predicting the prognosis and immune infiltration status of ovarian cancer. J Zhejiang Univ Sci B. 2023;24(1):64–77.
Sheehy J, Rutledge H, Acharya UR, Loh HW, Gururajan R, Tao X, et al. Gynecological cancer prognosis using machine learning techniques: a systematic review of the last three decades (1990–2022). Artif Intell Med. 2023;139: 102536.
Homayoonfal M, Asemi Z, Yousefi B. Potential anticancer properties and mechanisms of thymoquinone in osteosarcoma and bone metastasis. Cell Mol Biol Lett. 2022;27(1):21.
Maleki Dana P, Sadoughi F, Asemi Z, Yousefi B. The role of polyphenols in overcoming cancer drug resistance: a comprehensive review. Cell Mol Biol Lett. 2022;27(1):1.
Arooj A, Rabail R, Naeem M, Goksen G, Xu B, Aadil RM. A comprehensive review of the bioactive components of sesame seeds and their impact on bone health issues in postmenopausal women. Food Funct. 2023;14(11):4966–80.
Rosalina R, Weerapreeyakul N. An insight into sesamolin: physicochemical properties, pharmacological activities, and future research prospects. Molecules. 2021;26(19):5849.
Srisongkram T, Weerapreeyakul N. Route of intracellular uptake and cytotoxicity of sesamol, sesamin, and sesamolin in human melanoma SK-MEL-2 cells. Biomed Pharmacother. 2022;146: 112528.
Lin X, Zheng X, Yang B, Chen J, Xu Q, Wang Q. Clinical significance and immune landscapes of stemness-related and immune gene set-based signature in oral cancer. Clin Transl Med. 2021;11(2): e343.
Lin X, Chen J, Li X, Chen D, Luo K, Deng Y, et al. Dimeric oxyberberine CT4-1 targets LINC02331 to induce cytotoxicity and inhibit chemoresistance via suppressing Wnt/beta-catenin signaling in hepatocellular carcinoma. Arch Toxicol. 2023;97(6):1627–47.
Miranda A, Hamilton PT, Zhang AW, Pattnaik S, Becht E, Mezheyeuski A, et al. Cancer stemness, intratumoral heterogeneity, and immune response across cancers. Proc Natl Acad Sci U S A. 2019;116(18):9020–9.
Ronchetti L, Terrenato I, Ferretti M, Corrado G, Goeman F, Donzelli S, et al. Circulating cell free DNA and citrullinated histone H3 as useful biomarkers of NETosis in endometrial cancer. J Exp Clin Cancer Res. 2022;41(1):151.
Lin X, Liu J, Zou Y, Tao C, Chen J. Xanthotoxol suppresses non-small cell lung cancer progression and might improve patients’ prognosis. Phytomedicine. 2022;105: 154364.
Lin X, Wang F, Chen J, Liu J, Lin YB, Li L, et al. N(6)-methyladenosine modification of CENPK mRNA by ZC3H13 promotes cervical cancer stemness and chemoresistance. Mil Med Res. 2022;9(1):19.
Lin X, Chen J, Tao C, Luo L, He J, Wang Q. Osthole regulates N6-methyladenosine-modified TGM2 to inhibit the progression of rheumatoid arthritis and associated interstitial lung disease. MedComm (2020). 2023;4(2):e219.
Kalogera E, Roy D, Khurana A, Mondal S, Weaver AL, He X, et al. Quinacrine in endometrial cancer: repurposing an old antimalarial drug. Gynecol Oncol. 2017;146(1):187–95.
Han P, An N, Yang L, Ren XD, Lu SL, Ji H, et al. Molecular dynamics simulation of the interactions between sesamol and myosin combined with spectroscopy and molecular docking studies. Food Hydrocoll. 2022;131:107801.
Pian**g P, Thiantanawat A, Rangkadilok N, Watcharasit P, Mahidol C, Satayavivad J. Estrogenic activities of sesame lignans and their metabolites on human breast cancer cells. J Agric Food Chem. 2011;59(1):212–21.
Sobocan M, Smolle MA, Schatz C, Haybaeck J. The interplay of tumor stroma and translational factors in endometrial cancer. Cancers (Basel). 2020;12(8):2074.
Ma X, Zhao T, Yan H, Guo K, Liu Z, Wei L, et al. Fatostatin reverses progesterone resistance by inhibiting the SREBP1-NF-kappaB pathway in endometrial carcinoma. Cell Death Dis. 2021;12(6):544.
Miao Y, Konno Y, Wang B, Zhu L, Zhai T, Ihira K, et al. Integrated multi-omics analyses and functional validation reveal TTK as a novel EMT activator for endometrial cancer. J Transl Med. 2023;21(1):151.
Zhou Q, Li W, Kong D, Liu Z, Shi Z, Ma X, et al. DACH1 suppresses epithelial to mesenchymal transition (EMT) through Notch1 pathway and reverses progestin resistance in endometrial carcinoma. Cancer Med. 2019;8(9):4380–8.
Wang S, Ma Y, Hu Y, Zhao X, Li Y, Ouyang S, et al. FOXD3 confers chemo-sensitivity in ovarian cancer through a miR-335/DAAM1/myosin II axis-dependent mechanism. J Ovarian Res. 2023;16(1):8.
Gao S, Hu J, Wu X, Liang Z. PMA treated THP-1-derived-IL-6 promotes EMT of SW48 through STAT3/ERK-dependent activation of Wnt/beta-catenin signaling pathway. Biomed Pharmacother. 2018;108:618–24.
Zheng H, Song K, Fu Y, You T, Yang J, Guo W, et al. An absolute human stemness index associated with oncogenic dedifferentiation. Brief Bioinform. 2021;22(2):2151–60.
Yi M, Nissley DV, McCormick F, Stephens RM. ssGSEA score-based Ras dependency indexes derived from gene expression data reveal potential Ras addiction mechanisms with possible clinical implications. Sci Rep. 2020;10(1):10258.
Ang HL, Mohan CD, Shanmugam MK, Leong HC, Makvandi P, Rangappa KS, et al. Mechanism of epithelial-mesenchymal transition in cancer and its regulation by natural compounds. Med Res Rev. 2023;43(4):1141–200.
Sadrkhanloo M, Entezari M, Orouei S, Ghollasi M, Fathi N, Rezaei S, et al. STAT3-EMT axis in tumors: modulation of cancer metastasis, stemness and therapy response. Pharmacol Res. 2022;182: 106311.
Liang Y, Tan B, Du M, Wang B, Gao Y, Wang M. A tricarboxylic acid cycle-based machine learning model to select effective drug targets for the treatment of esophageal squamous cell carcinoma. Front Pharmacol. 2023;14:1195195.
Chai N, **e P, Chen H, Li Y, Zhao Y, He J, et al. Elevated ADAM-like Decysin-1 (ADAMDEC1) expression is associated with increased chemo-sensitivity and improved prognosis in breast cancer patients. Ann Transl Med. 2023;11(1):14.
Li W, Wang S, Qiu C, Liu Z, Zhou Q, Kong D, et al. Comprehensive bioinformatics analysis of acquired progesterone resistance in endometrial cancer cell line. J Transl Med. 2019;17(1):58.
Huang Y, Hong W, Wei X. The molecular mechanisms and therapeutic strategies of EMT in tumor progression and metastasis. J Hematol Oncol. 2022;15(1):129.
Lee JH, Massague J. TGF-beta in developmental and fibrogenic EMTs. Semin Cancer Biol. 2022;86(Pt 2):136–45.
Lin X, Zuo S, Luo R, Li Y, Yu G, Zou Y, et al. HBX-induced miR-5188 impairs FOXO1 to stimulate beta-catenin nuclear translocation and promotes tumor stemness in hepatocellular carcinoma. Theranostics. 2019;9(25):7583–98.
Lin X, Luo L, Zou Y, Chen J. Cancer stemness-associated LINC02475 serves as a novel biomarker for diagnosis and prognosis prediction of hepatocellular carcinoma. Front Genet. 2022;13: 991936.
Tu Y, Tan L, Tao H, Li Y, Liu H. CETSA and thermal proteome profiling strategies for target identification and drug discovery of natural products. Phytomedicine. 2023;116: 154862.
Lucero B, Francisco KR, Liu LJ, Caffrey CR, Ballatore C. Protein-protein interactions: develo** small-molecule inhibitors/stabilizers through covalent strategies. Trends Pharmacol Sci. 2023;44(7):474–88.
Wu D, Li Y, Zheng L, **ao H, Ouyang L, Wang G, et al. Small molecules targeting protein-protein interactions for cancer therapy. Acta Pharm Sin B. 2023;13(10):4060–88.
Wang ZZ, Shi XX, Huang GY, Hao GF, Yang GF. Fragment-based drug discovery supports drugging “undruggable” protein-protein interactions. Trends Biochem Sci. 2023;48(6):539–52.
Kitipaspallop W, Sillapaprayoon S, Phuwapraisirisan P, Kim WK, Chanchao C, Pimtong W. Developmental effects of sesamolin on zebrafish (Danio rerio) embryos. Comp Biochem Physiol C Toxicol Pharmacol. 2022;256: 109319.
Wu D, Wang XP, Zhang W. Sesamolin exerts anti-proliferative and apoptotic effect on human colorectal cancer cells via inhibition of JAK2/STAT3 signaling pathway. Cell Mol Biol (Noisy-le-grand). 2019;65(6):96–100.
Baek SH, Kang MG, Park D. Inhibitory effect of sesamolin on melanogenesis in B16F10 cells determined by in vitro and molecular docking analyses. Curr Pharm Biotechnol. 2020;21(2):169–78.
Surcel A, Schiffhauer ES, Thomas DG, Zhu Q, DiNapoli KT, Herbig M, et al. Targeting mechanoresponsive proteins in pancreatic cancer: 4-hydroxyacetophenone blocks dissemination and invasion by activating MYH14. Cancer Res. 2019;79(18):4665–78.
Bryan DS, Stack M, Krysztofiak K, Cichon U, Thomas DG, Surcel A, et al. 4-Hydroxyacetophenone modulates the actomyosin cytoskeleton to reduce metastasis. Proc Natl Acad Sci U S A. 2020;117(36):22423–9.
Oboulbiga EB, Douamba Z, Compaore-Sereme D, Sempore JN, Dabo R, Semde Z, et al. Physicochemical, potential nutritional, antioxidant and health properties of sesame seed oil: a review. Front Nutr. 2023;10:1127926.
Zhong Z, Vong CT, Chen F, Tan H, Zhang C, Wang N, et al. Immunomodulatory potential of natural products from herbal medicines as immune checkpoints inhibitors: Hel** to fight against cancer via multiple targets. Med Res Rev. 2022;42(3):1246–79.
Acknowledgements
The authors sincerely compliment the staff of the pathology department for completing the project.
Funding
This study was funded by the Joint Funds for the Innovation of Science and Technology, Fujian Province (no. 2023Y9454), the Guangdong Basic and Applied Basic Research Foundation (no. 2021A1515111089), the Fujian Provincial Financial Health Provincial Special Subsidy Funds (No. BPB-2023ZXF), the Natural Science Foundation of Fujian Province (no. 2021J01233), the Medical Innovation Project of Fujian Provincial Health Commission (no. 2020CXA036), and the Fujian Province Finance Project (no. BPB-ZXF2021).