Abstract
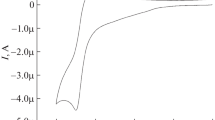
The electrochemical properties of N-substituted salts of 2,2'-bipyridine: N-methyl-2,2'-bipyridinium iodide and N,N '-dimethyl-2,2'-bipyridinium iodide were studied by cyclic voltammetry (CV). The electrochemical properties are greatly affected by the methyl substituents at the nitrogen atom in the ortho-bipyridine molecule. The conproportionation constants were calculated for N,N '-dimethyl-2,2'-bipyridinium iodide and made it possible to judge about the degree of electron localization in the systems.








Similar content being viewed by others
REFERENCES
R. S. Weber, ACS Catal. 9, 946 (2019). https://doi.org/10.1021/acscatal.8b04143
G. Chisholm, T. Zhao, and L. Cronin, in Storing Energy: With Special Reference to Renewable Energy Sources (Elsevier, Amsterdam, 2022), p. 559. https://doi.org/10.1016/B978-0-12-824510-1.00015-5
Z. Chen, W. Wei, L. Song, et al., Sustainable Horizons 1, 100002 (2022). https://doi.org/10.1016/j.horiz.2021.100002
Catalysts for Sustainable Hydrogen Production: Preparation, Applications and Process Integration (MDPI, 2022). https://doi.org/10.3390/books978-3-0365-3671-2
J. K. Nørskov, T. Bligaard, A. Logadottir, et al., J. Electrochem. Soc. 152 (3), 1 (2005). https://doi.org/10.1149/1.1856988
X. Gao and S. Kawi, in Heterogeneous Catalysis for Sustainable Energy, Ed. by Landong Li and J. S. J. Hargreaves (Wiley-VCH, Weinheim, 2022), p. 1. https://doi.org/10.1002/9783527815906.ch1
N. Queyriaux, D. Sun, J. Fize, et al., J. Am. Chem. Soc. 142, 274 (2020). https://doi.org/10.1021/jacs.9b10407
M. Wang, L. Chen, and L. Sun, Energy Environ. Sci. 5, 6763 (2012). https://doi.org/10.1039/c2ee03309g
Y. Huang, A. G. A. Mohamed, J. **e, et al., Nano Energy 82, 105745 (2021). https://doi.org/10.1016/j.nanoen.2021.105745
N. Dubouis and A. Grimaud, Chem. Sci. 10, 9165 (2019). https://doi.org/10.1039/C9SC03831K
J. A. Cracknell, K. A. Vincent, and F. A. Armstrong, Chem. Rev. 108, 2439 (2008). https://doi.org/10.1021/cr0680639
D. Merki and X. R. Hu, Energy Environ. Sci. 4, 3878 (2011). https://doi.org/10.1039/c1ee01970h
A. V. Dolganov, O. V. Tarasova, A. Y. Ivleva, et al., Int. J. Hydrogen Energy 42, 27084 (2017). https://doi.org/10.1016/j.ijhydene.2017.09.080
A. V. Dolganov, O. V. Tarasova, D. N. Moiseeva, et al., Int. J. Hydrogen Energy 41, 9312 (2016). https://doi.org/10.1016/j.ijhydene.2016.03.131
A. V. Dolganov, A. V. Balandina, D. B. Chugunov, A. S. Timonina, Yu. I. Lyukshina, A. A. Ahmatova, A. D. Yudina, V. V. Shindina, V. O. Zhirnova, L. A. Klimaeva, and A. K. Osipov, Russ. J. Gen. Chem. 90, 1229 (2020). https://doi.org/10.1134/S1070363220070099
A. V. Dolganov, B. S. Tanaseichuk, M. K. Pryanichnikova, et al., J. Phys. Org. Chem. 32, e3930 (2019). https://doi.org/10.1002/poc.3930
A. V. Dolganov, E. E. Muryumin, O. Y. Chernyaeva, et al., Mater. Chem. Phys. 224, 148 (2019). https://doi.org/10.1016/j.matchemphys.2018.12.006
A. V. Dolganov, B. S. Tanaseichuk, Y. V. Tsebulaeva, et al., Int. J. Electrochem. Sci. 11, 9559 (2016). https://doi.org/10.20964/2016.11.24
A. V. Dolganov, O. V. Tarasova, A. V. Balandina, O. Yu. Chernyaeva, V. Yu. Yurova, Yu. M. Selivanova, and A. D. Yudina, Russ. J. Org. Chem. 55, 938 (2019). https://doi.org/10.1134/S1070428019070030
A. V. Dolganov, B. S. Tanaseichuk, V. Yu. Yurova, et al., Int. J. Hydrogen Energy 44, 21495 (2019). https://doi.org/10.1016/j.ijhydene.2019.06.067
A. V. Dolganov, B. S. Tanaseichuk, D. N. Moiseeva, et al., Electrochem. Commun. 68, 59 (2016). https://doi.org/10.1016/j.elecom.2016.04.015
A. V. Dolganov, O. Y. Chernyaeva, S. G. Kostryukov, et al., Int. J. Hydrogen Energy 45, 501 (2020). https://doi.org/10.1016/j.ijhydene.2019.10.175
A. V. Dolganov, B. S. Tanaseichuk, O. V. Tarasova, O. Yu. Chernyaeva, Yu. M. Selivanova, A. D. Yudina, K. A. Grigor’yan, A. V. Balandina, and V. Yu. Yurova, Russ. J. Electrochem. 55, 807 (2019). https://doi.org/10.1134/S1023193519080056
O. Yu. Ganz, L. A. Klimaeva, D. B. Chugunov, A. V. Mazhorova, A. V. Dolganov, and A. V. Knyazev, Russ. J. Phys. Chem. A 96, 954 (2022). https://doi.org/10.1134/S0036024422050120
P. J. Stephens, F. J. Devlin, C. F. Chabalowski, et al., J. Phys. Chem. 98, 11623 (1994). https://doi.org/10.1021/j100096a001
R. Ditchfield, W. J. Hehre, and J. A. Pople, J. Chem. Phys. 54, 724 (1971). https://doi.org/10.1063/1.1674902
M. W. Schmidt, K. K. Baldridge, J. A. Boatz, et al., J. Comput. Chem. 14, 1347 (1993). https://doi.org/10.1002/jcc.540141112
M.-H. Baik and R. A. Friesner, J. Phys. Chem. A 106, 7407 (2002). https://doi.org/10.1021/jp025853n
Yun-Fang Gao, Li-Li Yu, Qing-Qing Lu, and M. A. Chun-An, J. Acta Phys. Chim. Sin. 25, 1421 (2009). https://doi.org/10.3866/PKU.WHXB20090735
M. B. Robin and P. Day, Adv. Inorg. Chem. Radiochem. 10, 247 (1968). https://doi.org/10.1016/S0065-2792(08)60179-X
P. Zanello, S. Tamburini, P. A. Vigato, et al., Coord. Chem. Rev. 77, 165 (1987). https://doi.org/10.1016/0010-8545(87)85034-8
Funding
This study was financially supported by the Ministry of Science and Higher Education of the Russian Federation (topic no. 121111000064-5). It was performed under the government contract at Mordovia National Research University (creation of new laboratories for young researchers).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by L. Smolina
Rights and permissions
About this article
Cite this article
Dolganov, A.V., Klimaeva, L.A., Muryumin, E.E. et al. Electrochemical Properties of N-Methyl-2,2'-bipyridinium Iodide and N,N'-Dimethyl-2,2'-bipyridinium Iodide. Russ. J. Phys. Chem. 97, 2307–2313 (2023). https://doi.org/10.1134/S0036024423100060
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024423100060