Abstract
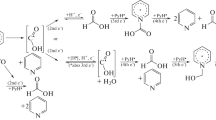
A study is performed of the electrocatalytic properties of 4,4'-bipyridine in the presence of acids of different natures (HBF4, HClO4, TsOH, CF3COOH). It is shown that pK acids are a key factor influencing the mechanism of the reactions taking place. The obtained electrochemical and electrocatalytic data are used to investigate the main mechanisms of the electrocatalytic production of molecular hydrogen, depending on the pK of the relevant acids. The same data are employed to calculate their thermodynamic and kinetic parameters.







Similar content being viewed by others
REFERENCES
J. A. Turner, Science (Washington, DC, U. S.) 305, 972 (2004). https://doi.org/10.1126/science.1103197
I. Dincer, Int. J. Hydrogen Energy 37, 1954 (2012). https://doi.org/10.1016/j.ijhydene.2011.03.173
S. A. Sherif, F. Barbir, and T. N. Veziroglu, Sol. Energy 78, 647 (2005). https://doi.org/10.1016/j.solener.2005.01.002
O. Pantani, E. Anxolabehere-Mallart, A. Aukauloo, et al., Electrochem. Commun. 9, 54 (2007). https://doi.org/10.1016/j.elecom.2006.08.036
J. P. Cao, T. Fang, L. Z. Fu, et al., Int. J. Hydrogen Energy 39, 10980 (2014). https://doi.org/10.1016/j.ijhydene.2014.05.082
C. Tang, R. Zhang, W. Lu, et al., Angew. Chem. Int. Ed. 37, 1127 (2016). https://doi.org/10.1002/anie.201608899
A. V. Dolganov, B. S. Tanaseichuk, V. Y. Yurova, et al., Int. J. Hydrogen Energy 44, 21495 (2019). https://doi.org/10.1016/j.ijhydene.2019.06.067
A. V. Dolganov, B. S. Tanaseichuk, D. N. Moiseeva, et al., Electrochem. Commun. 68, 59 (2016). https://doi.org/10.1016/j.elecom.2016.04.015
A. V. Dolganov, B. S. Tanaseichuk, M. K. Pryanichnikova, et al., J. Phys. Org. Chem. 32, 3930 (2019). https://doi.org/10.1002/poc.3930
V. Artero and J. M. Saveant, Energy Environ. Sci. 7, 3808 (2014). https://doi.org/10.1039/C4EE01709A
C. Costentin and J. M. Saveant, ChemElectroChem. 1, 1226 (2014). https://doi.org/10.1002/celc.201300263
P. S. Guin, S. Das, and P. C. Mandal, Int. J. Electrochem., 1 (2011).https://doi.org/10.4061/2011/816202
M. E. Tessensohn, H. Hirao, and R. D. Webster, J. Phys. Chem. 117, 1081 (2013). https://doi.org/10.1021/jp311007m
N. Elgrishi, B. D. McCarthy, E. S. Rountree, et al., ACS Catal. 6, 3644 (2016). https://doi.org/10.1021/acscatal.6b00778
J. A. S. Roberts and R. M. Bullock, Inorg. Chem. 52, 3823 (2013).https://doi.org/10.1021/ic302461q
Funding
This work was supported by the Russian Foundation for Basic Research, project no. 19-33-90094.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Klimaeva, L.A., Ganz, O.Y., Chugunov, D.B. et al. Electrocatalytic Activity of 4,4'-Bipyridine in the Production of Molecular Hydrogen in the Presence of Acids of Different Nature. Russ. J. Phys. Chem. 96, 958–963 (2022). https://doi.org/10.1134/S0036024422050156
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024422050156