Abstract
Killer cell immunoglobulin-like receptor genes, namely KIRs, cluster together within the 160 kb genomic DNA region. In this study, we used PCR-SSP approach and successfully identified the genotype of 17 KIR genes in 123 independent healthy donors residing in the Jiangsu province, China. All individuals were positive at the 7 genes. The observed carrier gene frequencies (OFs) of remaining 10 KIRs ranged from 14.63% (KIR2DS3) to 95.93% (KIR3DL1). We found 27 distinct genotypes excluding KIR1D. The most frequent occurred in 63 individuals (51.22%). The linkage disequilibrium analysis signified 29 positive and 6 negative relations in 45 pairwise comparisons. To study population differentiation, we drew a Heatmap based on the data of KIRs from 59 populations and conducted Hierarchical Clustering by Euclidean distances. We next validated our results by estimating pairwise DA distances and illustrating a Neighbor-Joining tree, as well as a MDS plot covering 3 additional Chinese Han groups. The phylogenetic reconstruction and cluster analysis strongly indicated a genetically close relationship between Eastern and Jilin Hans. In conclusion, the present study provided a meritorious resource of KIR genoty** for population genetics, and could be helpful to uncover the genetic mechanism of KIRs in immune disease in the future.
Similar content being viewed by others
Introduction
Natural killer (NK) cells, associated with the innate immune response, are considered as the first line of defense against both infected and malignantly transformed cells1. As a kind of bone marrow-derived lymphocyte, NK cells use specific cell surface receptors to distinguish healthy cells and diseased cells2. Like T cells, NK cells possess the qualified characteristics of the adaptive immune system, including the production of memory cells that persist following antigen invasion and the ability to create a secondary recall response3. Whether NK cells produce activation or inhibitory function depends on the varieties of their surface receptors. NK cells express three main families of receptors: (i) the highly polymorphic killer cell immunoglobulin-like receptors (KIRs) which specifically interact with classical MHC Class I molecules, (ii) the non-polymorphic C-type lectin (CD94/NKGs) receptors which bind to the non-classical MHC molecule HLA-E, and (iii) the immunoglobulin-like transcripts4. Killer cell immunoglobulin-like receptors (KIRs), defined as specific cell surface receptors, are a group of glycoproteins expressed on the surface of both NK cells and a few subsets of T cells. Upon the interaction with polymorphic human leukocyte antigen (HLA) class I molecules on the surface of target cells and other ligands, the KIRs participate in various immune responses to different infectious agents. Because of the high diversity of KIR genes, it is reasonable to hypothesize that the polymorphism of the KIRs in combination with HLA genes might affect predisposition to autoimmune disease. Recent genetic experiments demonstrated the associations between KIR and HLA genes with susceptibility to autoimmune diseases including Systemic Lupus Erythematosus (SLE), rheumatoid arthritis, systemic sclerosis and multiple sclerosis5,6,7,8,9,10. NK cells derived from human pulmonary artery hypertension (PAH) patients exhibited a lower level of the KIRs 2DL1/S1 and 3DL1 expression as well as a great disruption of 3DL1 receptor associated cytolytic function, suggesting a novel and substantive role for KIRs in the occurrence and development of immune associated vascular disease11.
The KIRs are encoded by a family of highly polymorphic genes clustered within the leukocyte receptor complex region on human chromosome 19q13.4. All of the KIR genes are encoded within a range of 160 kb genomic sequence, and they cluster together with a genetic distance shorter than 3 kb. As well, the KIR clusters have shown a good deal of gene duplications and unequal crossing over, which lead to a wide range of KIR gene combinations. KIR genes contain a tandem array of highly homologous genes, the number and type of which have the ability to show high diversity in different NK clone cells and individuals12. KIR gene nomenclature as defined by the World Health Organization (WHO) subcommittee is based on the structure of the encoded molecules. Accordingly, the number of extracellular immunoglobulin domain (D) could be double (2D) or triple (3D) and therefore the length of the intracytoplasmic tail would be short (S) or long (L). In terms of the order of KIR genes and the gene contents of 15 loci, KIR genes are divided into two haplotypes, A and B. The A haplotype contains at least six encoding inhibitory receptors (KIR3DL3, 2DL3, 2DL1, 2DL4, 3DL1 and 3DL2), one pseudogene (KIR3DP1) and one activating receptor gene (KIR2DS4)13. The haplotype B consists of a great variety of subtypes that differ from each other in the combination of stimulatory receptors (KIR2DS1, 2DS2, 2DS3, 2DS5, 3DS1, 2DL2 and 2DL5)14. On condition that the presence of KIR2DS1, 2DS2, 2DS3, 2DS5, 3DS1, 2DL2 and 2DL5 are observed, the genotype would be determined as including B. If all of the above-mentioned genes are absent, the genotype would be defined as AA. When one or more of KIR genes belonging to A group are absent, the genotype would be taken as BB. Otherwise, all the remaining genotypes are defined to be AB. Seven KIR genes belonging to B group are located centromeric or telomeric half in B cluster, B group is classified into 2 half groups: C4 half (KIR2DS2, 2DL2, 2DS3 and 2DL5) and T4 half (KIR3DS1, 2DL5, 2DS1 and 2DS5). Herein, the B group is divided into 4 subgroups: C4T4, C4Tx, CxT4, and CxTx. Seven KIRs (KIR2DL1, 2DL2, 2DL3, 2DL5, 3DL1, 3DL2 and 3DL3) have inhibitory functions while five KIRs (KIR2DS1, 2DS2, 2DS3, 2DS5 and 3DS1) exhibit active functions. KIR2DL4 has both inhibitory and active functions12. Among all KIR genes, four genes KIR2DL4, KIR3DL2, KIR3DL3 and KIR3DP1 are described as framework genes, and they are present in nearly all individuals15.
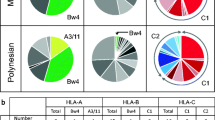
In the previous research, studies have shown the KIR gene diversity in different geographical populations16,17,18,19,20,21,22,23,24,25, Blood samples were collected from a total of 123 randomly selected healthy donors of Han ancestry living in Nan**g, Jiangsu province of China, membership of Eastern China. All participants provided their written informed consent and completed a basic health screen. Also, each participant was interviewed to ensure that no individuals have common ancestry going back at least three generations and their three generations are native of eastern coastal area of China. The whole-blood samples anti-coagulated with ethylene diamine tetraacetic acid (EDTA) were frozen at −80 °C until use. The study was conducted in accordance with the human and ethical research principles of Nan**g Medical University and approved by the ethics committee of Nan**g Medical University. According to the manufacturer’s instructions, we extracted genomic DNA from 300 μl peripheral blood containing ethylene diamine tetraacetic acid (EDTA), using TIANamp Genomic DNA Kit (TIANGEN, Bei**g, China). The quality and quantity of extracted DNA samples were determined by NanoDrop 2000 (Thermo Fisher Scientific, Waltham, USA). The optimal density values used to evaluate the concentration and purity of extracted DNA reflected by the A260/280 values (1.7 to 1.9). The concentration was adjusted to 20–40 ng/μl. KIR genes were genotyped for the presence or absence of the 17 KIR genes, including KIR2DL1, 2DL2, 2DL3, 2DL4, 2DL5, 3DL1, 3DL2, 3DL3, 2DS1, 2DS2, 2DS3, 2DS4, 2DS5, 3DS1 (in the full-length form), 1D (in the deleted form) and two pseudogenes, 3DP1(putative protein product) and 2DP1(no protein expression) using PCR-SSP method with a commercially available KIR GENOTYPING SSP KIT (Invitrogen, California, USA). The PCR amplification was performed with the PCR system in a reaction mixture volume of 10 μl consisting of 4 μl pre-aliquoted PCR buffer, 0.06 μl Taq polymerase, and 30 to 50 ng of template DNA. Temperature cycling conditions for PCR reactions were as follows: denaturation for 1 minute at 95 °C, followed by 30 cycles for 20 seconds at 94 °C, 20 seconds at 63 °C, 1.5 minutes at 72 °C, a elongation step for 10 minutes at 72 °C and finally hold in 4 °C. PCR products were visualized under ultraviolet light after electrophoresis in 1.5% agarose gel well mixed with 10% v/v ethidium bromide. Negative controls were performed for each gene while positive internal controls were performed for each lane. False reactions that yielded no internal control bands were repeated. The observed carrier frequencies (OFs) of the KIR genes were determined by the number of positive ty** reactions. Based on the assumption of Hardy-Weinberg equilibrium, we calculated the estimated gene frequencies (GFs) using the formula, GF = 1−(1−OF)1/2. The GF is determined by the OF of the KIR gene in all individuals. Package “pheatmap” (https://cran.r-project.org/web/packages/pheatmap/index.html) based on statistical software R version 3.2.5 (https://www.r-project.org/) was used to draw a Heatmap containing Eastern Chinese Han and 58 other populations with complete KIR genoty** files of 16 KIR genes exclusive of KIR1D which are Jilin Han27, Shaanxi Han18, Shenzhen Han28, Sichuan Han29, ** profiles observed on Killer cell immunoglobulin-like receptors (KIRs) in the Eastern Chinese Han(n = 123). Since KIR genes are close to each other by approximately 3 kb genomic DNA region, linkage disequilibrium should be taken into consideration, especially for those two nearest-neighbor KIRs (present or absent simultaneously). Therefore, we conducted the LD analysis, in which D was the parameter representing the test statistics linkage disequilibrium coefficient. P-value < 0.05 signified a strong genetic relation between two KIRs. Based on the genoty** profiles of 123 Eastern Chinese Hans, Table 3 and Fig. 2 listed the D coefficients and conspicuous differences of 45 pairs among 10 KIR genes except for 7 KIR genes which appeared in all individuals. The Linkage Disequilibrium comparisons were listed in an inverted triangle in Fig. 2. The results of LD analysis showed 35 strong relations in total, among which 6 were negative, all associated with KIR1D (1D-3DL1, 1D-2DS4, 1D-2DL5, 1D-3DS1, 1D-2DS1, and 1D-2DS3). The rest 29 relations were significantly positive on different levels. In terms of group classification, 1 relation was from A group (3DL1-2DS4) while 10 pairs linked A and B groups (3DL1-2DL5, 3DL1-3DS1, 3DL1-2DS1, 3DL1-2DS5, 3DL1-2DS3, 2DS4-2DL5, 2DS4-3DS1, 2DS4-2DS1, 2DS4-2DS5, and 2DS4-2DS3) and 18 linkage relations existed within B group. After further demarcation, 6 linkage relations (2DL5-2DL2, 2DS2-2DL2, 2DS3-2DL2, 2DS2-2DL5, 2DS3-2DL5, and 2DS2-2DS3) and 5 (3DS1-2DL5, 2DS1-2DL5, 2DS5-2DL5, 3DS1-2DS5, and 2DS1-2DS5) were found within C4 and T4 subgroups from B family respectively, consistent to the genomic location of the genes. As for 1D, a known 2DS4 variant, we illustrated a very close linkage relationship between 1D and KIR2DS4 which showed the linkage coefficient was −0.0245 (p < 0.001). Thus, the results supported that KIR1D is the variant of KIR2DS447. These findings are consistent to other published studies48,49,50,51. In order to investigate the potential role of KIRs in population genetics, we compared the KIR frequencies of Eastern Han and 58 other populations and performed population differentiation analysis. Because KIR1D was excluded from the database, we utilized the rest 16 genes for the Heatmap analysis. The 59 selected populations are from East Europe, West Europe, Africa, North America, Latin America, Asia and Pacific Ocean, which are almost on behalf of human populations all over the earth. In Fig. 3, the deeper color represented higher observed frequency. We found all KIR genes are present in Eastern Han population. However, KIR3DS1 was not detected in African populations such as Bantu NE, Bantu S, Mbuti Pygmies and San groups. Similarly, 2DS3 was missing in some populations from East Asia (Hezhen, Lahu, Mongola, and Tujia) and America (Karitiana, Pima, Surui, and Colombian). With the knowledge that EDAR variant emerged with ameliorative phenotypes in East Asian, especially Central Chinese by natural selection52, we speculated that KIR3DS1 was beginning to surface and KIR2DS3 disappeared gradually due to evolutionary pressure such as microbial infection, communicable disease or natural environment on the route of “out of Africa”. However, the potential causes remained to be discovered. The Hierarchical Clustering method was employed to reconstruct the cluster trees and molecular evolutionary structures based on Euclidean distance. The populations and KIRs were listed corresponding to the population genetic and homologue analysis, respectively (Fig. 3). Firstly, we found that molecular evolutionary structure was divided into two clusters. The left was composed of KIR genes defined as B group, while the right consisted of A group and KIR2DP1. The results were consistent with the deduced classification clones from original research14. Interestingly, KIR2DS1 clustered with KIR3DS1, KIR2DL2 and 2DS2 were from the same branch, matching up with the classification of linkage subgroups (C4, T4). Secondly, from the phylogenetic structure, two main clusters existed according to the relative OFs of B group loci. The lower cluster differed from the top by higher OFs for most B haplotype KIRs (especially the lowest 5 populations from Oceania and Africa including Melanesian, Papuan, Mbuti Pygmies, Biaka Pygmies and San). The studied Eastern Han labelled with red arrow resided in the top cluster. Obviously, Han populations from HGDP-CEPH database including ** files of 16 KIR genes from a mathematics angle. In the validation study, we constructed another Neighbor-Joining tree (Fig. 4) by utilizing the observed frequency data of the Eastern Han, 52 populations from HGDP-CEPH, and reported 9 Han groups at 13 KIR loci (KIR2DL1, 2DL2, 2DL3, 2DL4, 2DL5, 3DL1, 3DL2, 3DL3, 2DS1, 2DS2, 2DS3, 2DS5 and 3DS1) with the purpose of revealing genetic relationships among these 62 populations. The results of interior branch test were shown in Figure S1 with the sum of branch length = 0.41784153. The values of DA and significant level were listed in Supplementary Table S1. Without correction, the p-value greater than 0.05 appeared 330 times and the rest 1,561 comparisons were found to be significantly different. The number of significances ranged from 13 in Pacific Melanesian to 60 in Bedouin, Balochi, Brahui, ** files, phylogenetic construction, evolutionary molecular analysis and sub-Han group comparison provided valuable resources for both enriching information pool of forensic and population genetics and benefiting in uncovering underlying genetic mechanisms underlying immune diseases in the future. The comprehensive and comparative analysis on KIRs revealed a unique genetic background of Eastern Chinese Han through phylogenetic construction, evolutionary molecular analysis, and sub-Han group comparison which provided valuable resources for both enriching information pool of forensic and population genetics.Methods
Study samples
DNA isolation
KIR PCR-SSP genoty**
Statistical analysis
Linkage disequilibrium (LD) analysis
Evolution significance of KIR cluster and Phylogenetic restructure
Phylogenetic structure and MDS analysis
Additional Information
How to cite this article: Yin, C. et al. Genetic polymorphism and evolutionary differentiation of Eastern Chinese Han: a comprehensive and comparative analysis on KIRs. Sci. Rep. 7, 42486; doi: 10.1038/srep42486 (2017).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Trinchieri, G. Biology of natural killer cells. Advances in Immunology 47, 187–376 (1989).
Lanier, L. L. NK cell recognition. Annual Review of Immunology 23, 225–274, doi: 10.1146/annurev.immunol.23.021704.115526 (2005).
Rajalingam, R. Overview of the killer cell immunoglobulin-like receptor system. Methods in Molecular Biology 882, 391–414, doi: 10.1007/978-1-61779-842-9_23 (2012).
Samaridis, J. & Colonna, M. Cloning of novel immunoglobulin superfamily receptors expressed on human myeloid and lymphoid cells: structural evidence for new stimulatory and inhibitory pathways. European Journal of Immunology 27, 660–665, doi: 10.1002/eji.1830270313 (1997).
Schleinitz, N., Vely, F., Harle, J. R. & Vivier, E. Natural killer cells in human autoimmune diseases. Immunology 131, 451–458, doi: 10.1111/j.1365-2567.2010.03360.x (2010).
Fogel, L. A., Yokoyama, W. M. & French, A. R. Natural killer cells in human autoimmune disorders. Arthritis Research & Therapy 15, 216, doi: 10.1186/ar4232 (2013).
Fusco, C. et al. KIRs and their HLA ligands in Remitting-Relapsing Multiple Sclerosis. Journal of Neuroimmunology 229, 232–237, doi: 10.1016/j.jneuroim.2010.08.004 (2010).
Pellett, F. et al. KIRs and autoimmune disease: studies in systemic lupus erythematosus and scleroderma. Tissue Antigens 69, 106–108, doi: 10.1111/j.1399-0039.2006.762_6.x (2007).
Hou, Y. F. et al. Disparate distribution of activating and inhibitory killer cell immunoglobulin-like receptor genes in patients with systemic lupus erythematosus. Lupus 19, 20–26, doi: 10.1177/0961203309345779 (2010).
McGeough, C. M. et al. Killer immunoglobulin-like receptor and human leukocyte antigen-C genotypes in rheumatoid arthritis primary responders and non-responders to anti-TNF-alpha therapy. Rheumatology International 32, 1647–1653, doi: 10.1007/s00296-011-1838-6 (2012).
Ormiston, M. L. et al. Impaired natural killer cell phenotype and function in idiopathic and heritable pulmonary arterial hypertension. Circulation 126, 1099–1109, doi: 10.1161/CIRCULATIONAHA.112.110619 (2012).
Uhrberg, M. et al. Human diversity in killer cell inhibitory receptor genes. Immunity 7, 753–763 (1997).
Martin, A. M., Freitas, E. M., Witt, C. S. & Christiansen, F. T. The genomic organization and evolution of the natural killer immunoglobulin-like receptor (KIR) gene cluster. Immunogenetics 51, 268–280 (2000).
Wilson, M. J., Torkar, M. & Trowsdale, J. Genomic organization of a human killer cell inhibitory receptor gene. Tissue Antigens 49, 574–579 (1997).
Al Omar, S. Y. et al. Genotypic diversity of the Killer Cell Immunoglobulin-like Receptors (KIR) and their HLA class I Ligands in a Saudi population. Genetics and Molecular Biology 39, 14–23, doi: 10.1590/1678-4685-GMB-2015-0055 (2016).
Zhu, B. F. et al. Diversity of killer cell immunoglobulin-like receptor genes in the Bai ethnic minority of Yunnan, China. Scandinavian Journal of Immunology 73, 284–292, doi: 10.1111/j.1365-3083.2011.02508.x (2011).
Zhu, B. F. et al. Killer cell immunoglobulin-like receptor gene diversity in the Tibetan ethnic minority group of China. Human Immunology 71, 1116–1123, doi: 10.1016/j.humimm.2010.07.010 (2010).
Wang, H. D. et al. Diversity distributions of killer cell immunoglobulin-like receptor genes and their ligands in the Chinese Shaanxi Han population. Human Immunology 72, 733–740, doi: 10.1016/j.humimm.2011.04.003 (2011).
Middleton, D., Meenagh, A., Moscoso, J. & Arnaiz-Villena, A. Killer immunoglobulin receptor gene and allele frequencies in Caucasoid, Oriental and Black populations from different continents. Tissue Antigens 71, 105–113, doi: 10.1111/j.1399-0039.2007.00973.x (2008).
Norman, P. J. et al. Natural killer cell immunoglobulin-like receptor (KIR) locus profiles in African and South Asian populations. Genes and Immunity 3, 86–95, doi: 10.1038/sj.gene.6363836 (2002).
Lee, Y. C., Chan, S. H. & Ren, E. C. Asian population frequencies and haplotype distribution of killer cell immunoglobulin-like receptor (KIR) genes among Chinese, Malay, and Indian in Singapore. Immunogenetics 60, 645–654, doi: 10.1007/s00251-008-0321-3 (2008).
Pincan, S. et al. Polymorphism of killer cell immunoglobulin-like receptors gene in Yunnan Han population, China. Chinese Journal of Blood Transfusion 21, 268–272 (2008).
Yawata, M. et al. Predominance of group A KIR haplotypes in Japanese associated with diverse NK cell repertoires of KIR expression. Immunogenetics 54, 543–550, doi: 10.1007/s00251-002-0497-x (2002).
Whang, D. H., Park, H., Yoon, J. A. & Park, M. H. Haplotype analysis of killer cell immunoglobulin-like receptor genes in 77 Korean families. Human Immunology 66, 146–154, doi: 10.1016/j.humimm.2004.10.013 (2005).
Zhang, L. et al. [Killer Ig-like receptor gene content diversity and haplotype analysis in Chinese Han population in Shanghai]. Zhonghua Yi Xue Yi Chuan Xue Za Zhi = Zhonghua Yixue yichuanxue Zazhi = Chinese Journal of Medical Genetics 20, 396–399 (2003).
Chi, Y. B. et al. The diversity of KIR gene content and haplotype analysis in **njiang Uygur and Yunnan Yi populations in China. Current Immunology 25, 204–207 (2005).
Han, Y., Yang, F., Lin, Q. F., **, C. R. & Jiao, L. X. Analysis of the expression of compound KIR-HLA in Northeast Han population. Chinese Journal of Blood Transfusion 28, 1340–1342 (2015).
Zhen, J. et al. Genetic profile of KIR and HLA in southern Chinese Han population. Human Immunology 75, 59–64, doi: 10.1016/j.humimm.2013.09.006 (2014).
Zhan, X. T. et al. Relationship between KIR and HLA Ligands in Han population of Sichuan Marrow Donor Program. Chinese Journal of Blood Transfusion 25, 327–332, doi: 10.13303/j.cjbt.issn.1004-549x.2012.04.023 (2012).
Lin, G. Y. et al. Genetic polymorphism at the KIR gene locus: determination of gene, genotype, and haplotype frequencies in the **njiang Han population. Genetics and Molecular Biology 14, 6213–6222, doi: 10.4238/2015.June.9.7 (2015).
Hollenbach, J. A., Nocedal, I., Ladner, M. B., Single, R. M. & Trachtenberg, E. A. Killer cell immunoglobulin-like receptor (KIR) gene content variation in the HGDP-CEPH populations. Immunogenetics 64, 719–737, doi: 10.1007/s00251-012-0629-x (2012).
Cann, H. M. et al. A human genome diversity cell line panel. Science 296, 261–262 (2002).
White, S. D. M. The hierarchy of correlation functions and its relation to other measures of galaxy clustering. Monthly Notices of the Royal Astronomical Society 186, 145–154 (1979).
Pando, J., Lipa, P., Greiner, M. & Fang, L.-Z. Probing Hierarchical Clustering by Scale-Scale Correlations of Wavelet Coefficients. The Astrophysical Journal 496, 1–10 (1998).
Lewontin, R. C. On measures of gametic disequilibrium. Genetics 120, 849–852 (1988).
Reich, D. E. et al. Linkage disequilibrium in the human genome. Nature 411, 199–204, doi: 10.1038/35075590 (2001).
Nei, M. Analysis of gene diversity in subdivided populations. Proceedings of the National Academy of Sciences USA 70, 3321–3323 (1973).
Nei, M., Tajima, F. & Tateno, Y. Accuracy of estimated phylogenetic trees from molecular data. II. Gene frequency data. Journal of Molecular Evolution 19, 153–170 (1983).
Takezaki, N. & Nei, M. Genetic distances and reconstruction of phylogenetic trees from microsatellite DNA. Genetics 144, 389–399 (1996).
Latter, B. D. Selection in finite populations with multiple alleles. 3. Genetic divergence with centripetal selection and mutation. Genetics 70, 475–490 (1972).
Middleton, D., Meenagh, A., Serrano-Vela, J. I. & Arnaiz-Villen, J. M. Different Evolution of Inhibitory and Activating Killer Immunoglobulin Receptors (KIR) in Worldwide Human Populations The Open Immunology Journal 1, 42–50, doi: 10.2174/1874226200801010042 (2008).
Wang, H. D. et al. Study of genetic diversity of killer cell immunoglobulin-like receptor loci in the Tujia ethnic minority. Human Immunology 77, 869–875, doi: 10.1016/j.humimm.2016.06.015 (2016).
Takezaki, N. & Nei, M. Empirical tests of the reliability of phylogenetic trees constructed with microsatellite DNA. Genetics 178, 385–392, doi: 10.1534/genetics.107.081505 (2008).
Tamura, K., Stecher, G., Peterson, D., Filipski, A. & Kumar, S. MEGA6: Molecular Evolutionary Genetics Analysis version 6.0. Molecular Biology and Evolution 30, 2725–2729, doi: 10.1093/molbev/mst197 (2013).
Saitou, N. & Nei, M. The neighbor-joining method: a new method for reconstructing phylogenetic trees. Molecular Biology and Evolution 4, 406–425 (1987).
Zhou, Q. et al. KIR diversity in three ethnic minority populations in China. Journal of Translational Medicine 13, 1–8, doi: 10.1186/s12967-015-0544-7 (2015).
Wu, X. et al. KIR2DS4 and Its Variant KIR1D Are Associated with Acute Graft-versus-Host Disease, Cytomegalovirus, and Overall Survival after Sibling-Related HLA-Matched Transplantation in Patients with Donors with KIR Gene Haplotype A. Biology of Blood and Marrow Transplantation 22, 220–225, doi: 10.1016/j.bbmt.2015.10.004 (2016).
Hsu, K. C. et al. Killer Ig-like receptor haplotype analysis by gene content: evidence for genomic diversity with a minimum of six basic framework haplotypes, each with multiple subsets. Journal of Immunology 169, 5118–5129 (2002).
Norman, P. J., Stephens, H. A., Verity, D. H., Chandanayingyong, D. & Vaughan, R. W. Distribution of natural killer cell immunoglobulin-like receptor sequences in three ethnic groups. Immunogenetics 52, 195–205 (2001).
Shilling, H. G. et al. Allelic polymorphism synergizes with variable gene content to individualize human KIR genotype. Journal of Immunology 168, 2307–2315 (2002).
Jiang, K., Zhu, F. M., Lv, Q. F. & Yan, L. X. Distribution of killer cell immunoglobulin-like receptor genes in the Chinese Han population. Tissue Antigens 65, 556–563, doi: 10.1111/j.1399-0039.2005.00412.x (2005).
Kamberov, Y. G. et al. Modeling recent human evolution in mice by expression of a selected EDAR variant. Cell 152, 691–702, doi: 10.1016/j.cell.2013.01.016 (2013).
Liu, H., Prugnolle, F., Manica, A. & Balloux, F. A geographically explicit genetic model of worldwide human-settlement history. American Journal of Human Genetics 79, 230–237, doi: 10.1086/505436 (2006).
This week in Science: Out of Africa Revisited. Science 308, 1, doi: 10.1126/science.308.5724.921g (2005).
Johanson, D. Origins of Modern Humans: Multiregional or Out of Africa? Action Bioscience (2001).
Stringer, C. Human evolution: Out of Ethiopia. Nature 423, 692–693, 695, doi: 10.1038/423692a (2003).
Yao, J. & Wang, B. J. Genetic Variation of 25 Y-Chromosomal and 15 Autosomal STR Loci in the Han Chinese Population of Liaoning Province, Northeast China. PloS one 11, e0160415, doi: 10.1371/journal.pone.0160415 (2016).
Danielson, E. N. Shanghai and the Yangzi Delta: From Past to Present. The New Yangzi River Trilogy, Vol. I Times Edtions ISBN: 9812325972, 2 (2004).
Ward, B. E. Through other eyes: an anthropologist’s view of Hong Kong. Chinese University Press (1989).
Watson, J. L. Emigration and the Chinese lineage: the Mans in Hong Kong and London. University of California Press (1975).
Wen, B. et al. Genetic evidence supports demic diffusion of Han culture. Nature 431, 302–305, doi: 10.1038/nature02878 (2004).
Chen, J. et al. Genetic structure of the Han Chinese population revealed by genome-wide SNP variation. American Journal of Human Genetics 85, 775–785, doi: 10.1016/j.ajhg.2009.10.016 (2009).
1000 Genomes Project, C. et al. A global reference for human genetic variation. Nature 526, 68–74, doi: 10.1038/nature15393 (2015).
Torres, A. R., Westover, J. B., Gibbons, C., Johnson, R. C. & Ward, D. C. Activating killer-cell immunoglobulin-like receptors (KIR) and their cognate HLA ligands are significantly increased in autism. Brain Behavior and Immunity 26, 1122–1127 (2012).
Alter, G. et al. Differential natural killer cell-mediated inhibition of HIV-1 replication based on distinct KIR/HLA subtypes. Journal of Experimental Medicine 204, 3027–3036, doi: 10.1084/jem.20070695 (2007).
Garcia-Leon, J. A. et al. Killer-cell immunoglobulin-like receptor expression on lymphocyte subsets in multiple sclerosis patients treated with interferon-beta: evaluation as biomarkers for clinical response. CNS Drugs 28, 559–570 (2014).
Acknowledgements
This project was supported by the funding for National Natural Science Foundation of China (81570378 and 81400033 to F.C.), the Science and Technology of Jiangsu Province China (BK20140902 to F.C.), and Jiangsu Specially-Appointed Professor program.
Author information
Authors and Affiliations
Contributions
C.Y. wrote the main manuscript text, and F.C. designed the study and did the manuscript modification. C.Y., L.H., H.H, Y.Y., Z.L., Q.J., X.K., Z.W., J.Y., J.Y. and B.Z. did the data processing and prepared the figures. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Yin, C., Hu, L., Huang, H. et al. Genetic polymorphism and evolutionary differentiation of Eastern Chinese Han: a comprehensive and comparative analysis on KIRs. Sci Rep 7, 42486 (2017). https://doi.org/10.1038/srep42486
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep42486
- Springer Nature Limited